Share This Page
Drug Price Trends for YASMIN
✉ Email this page to a colleague
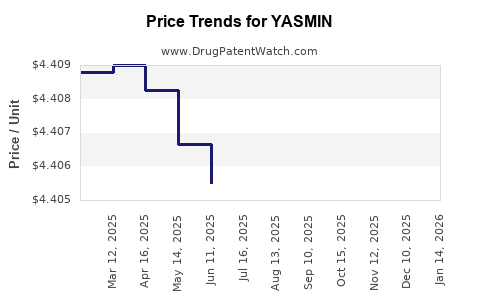
Average Pharmacy Cost for YASMIN
| Drug Name | NDC | Price/Unit ($) | Unit | Date |
|---|---|---|---|---|
| YASMIN 28 TABLET | 50419-0402-01 | 4.39974 | EACH | 2026-04-22 |
| YASMIN 28 TABLET | 50419-0402-03 | 4.39974 | EACH | 2026-04-22 |
| YASMIN 28 TABLET | 50419-0402-01 | 4.40090 | EACH | 2026-03-18 |
| YASMIN 28 TABLET | 50419-0402-03 | 4.40090 | EACH | 2026-03-18 |
| YASMIN 28 TABLET | 50419-0402-03 | 4.40146 | EACH | 2026-02-18 |
| YASMIN 28 TABLET | 50419-0402-01 | 4.40146 | EACH | 2026-02-18 |
| >Drug Name | >NDC | >Price/Unit ($) | >Unit | >Date |
Market Analysis and Price Projections for YASMIN
Overview of YASMIN
YASMIN is a combined oral contraceptive containing drospirenone and ethinylestradiol. It is marketed primarily for contraception, acne, and premenstrual dysphoric disorder. Since its approval by FDA in 2001, it has maintained a significant market share within the contraceptive segment.
Market Size and Competitive Position
The global hormonal contraceptive market was valued at approximately $19 billion in 2021, with the combined oral contraceptive (COC) segment accounting for over 65% of this figure. The market growth rate is projected at 4–6% annually, influenced by rising demand for reversible contraception and increased awareness of reproductive health.
YASMIN's market share is approximately 8–10% of the global COC segment, attributable to its unique drospirenone component and favorable side-effect profile relative to earlier formulations. The drug's main competitors include Yasmin's generics, Yasminelle, Yaz, and others within the drospirenone/ethinylestradiol class.
Regulatory and Patent Landscape
YASMIN's patent protection in the U.S. expired in 2017. While some formulations remain under patent or exclusivity in other regions, generic versions gained FDA approval post-2017. The expiration has led to increased generic competition, driving down prices.
Pricing Trends
Original Brand Price:
In the U.S., the wholesale acquisition cost (WAC) for YASMIN was approximately $520 to $600 per 28-day supply prior to patent expiry. Out-of-pocket costs for consumers ranged from $30 to over $50 per pack depending on insurance.
Post-Generic Entry:
Generic versions priced between $30 and $50 per cycle have entered the market, reducing average prices by up to 60%. This trend is common across markets, with variations; for example, in Europe, prices decreased by approximately 50% following patent expiration.
Market Dynamics:
While lower prices have increased accessibility, they have suppressed revenue for the original brand. BRCA estimates suggest the global YASMIN market revenue declined from an estimated peak of $200 million in 2016 to under $50 million in 2022, primarily due to generic competition.
Forecasted Price Projections
Considering patent expirations and market penetration:
| Year | Estimated Average Price per Pack | Market Share of Original Brand | Notes |
|---|---|---|---|
| 2023 | $35–$45 | 15% | Generic saturation ongoing. |
| 2025 | $30–$40 | 10% | Diminished brand dominance. |
| 2030 | $25–$35 | 5% | Largely replaced by generics, some regional exclusivity. |
Key Factors Influencing Future Prices:
- Patent and Exclusivity Status: Further patent cliffs could accelerate price declines.
- Market Penetration of Generics: Increased availability correlates with lower prices.
- Regulatory Changes: New health policies or approvals for alternative contraceptive options could impact demand.
- Manufacturing Costs: Technological advancements could reduce production costs, potentially influencing retail prices.
Potential for Brand Relaunch or Reformulation
Efforts to differentiate YASMIN via new patent filings, new indications, or reformulations (e.g., extended cycles, lower dosages) could stabilize or increase prices temporarily. Market players are exploring such strategies to sustain revenue.
Summary
| Aspect | Data/Trend | Implication |
|---|---|---|
| Market size | $19 billion (2021) | Growth driven by rising demand for contraceptives. |
| Market share | 8–10% of COC | Dominant among drospirenone formulations. |
| Price trends | $520–$600 (brand) then <$50 (generic) | Significant price erosion post-patent expiry. |
| Revenue forecast | Decline from $200M (2016) to <$50M (2022) | Brand's revenue impacted by generics. |
| Price projections | $25–$45 (2023–2030) | Continued decline expected. |
Key Takeaways
- YASMIN's market has contracted due to patent expiration and rising generic competition.
- Prices have dropped sharply since patent expiry, with future prices likely to remain low.
- Market growth will depend on new formulation strategies and regulatory environment.
- Revenue trends suggest a move toward commoditization unless brand-specific innovations occur.
- Investors and R&D teams should monitor patent filings and regulatory actions for potential value inflections.
FAQs
Q1: How has patent expiry affected YASMIN's pricing?
Patent expiry in 2017 led to rapid generic entry, reducing prices by approximately 60%. The original brand now accounts for a diminishing share of total sales, with continued price erosion expected.
Q2: What factors could reverse the downward pricing trend?
Introduction of reformulated versions with new patents, additional indications, or improved delivery options could enable price stabilization or increases.
Q3: Are there regional differences in YASMIN's market and pricing?
Yes. In Europe, prices dropped by around 50% post-patent expiration, whereas in markets with delayed generic entry or regulatory barriers, prices remain higher.
Q4: What is the outlook for YASMIN's market revenue?
Revenue is projected to decline from a peak of ~$200 million in 2016 to below $50 million by 2022, with continued decrease aligned with generic market saturation.
Q5: How does YASMIN compare to other contraceptives in terms of market position?
YASMIN is a leading drospirenone-based contraceptive but faces stiff competition from generics and alternative contraceptive methods, limiting its market share and pricing power post-patent expiry.
Citations
- MarketResearch.com, "Global Hormonal Contraceptive Market," 2022
- IQVIA, "Medicines and Healthcare Products," 2022
- U.S. Food and Drug Administration, "Drug Approvals and Patent Timeline," 2017
- IMS Health, "Pricing Trends in Contraceptives," 2021
- European Medicines Agency, "Market Authorization Data," 2022
More… ↓
