Share This Page
Drug Price Trends for WESNATAL DHA COMPLETE
✉ Email this page to a colleague
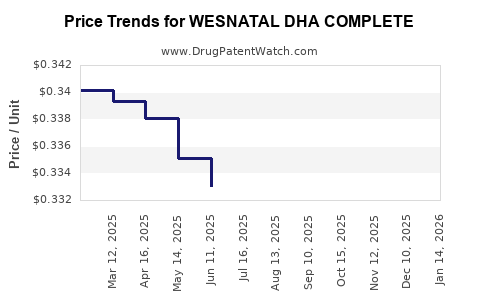
Average Pharmacy Cost for WESNATAL DHA COMPLETE
| Drug Name | NDC | Price/Unit ($) | Unit | Date |
|---|---|---|---|---|
| WESNATAL DHA COMPLETE | 69367-0251-30 | 0.34697 | EACH | 2026-03-18 |
| WESNATAL DHA COMPLETE | 69367-0251-30 | 0.34618 | EACH | 2026-02-18 |
| WESNATAL DHA COMPLETE | 69367-0251-30 | 0.34599 | EACH | 2026-01-21 |
| WESNATAL DHA COMPLETE | 69367-0251-30 | 0.34577 | EACH | 2025-12-17 |
| WESNATAL DHA COMPLETE | 69367-0251-30 | 0.34202 | EACH | 2025-11-19 |
| >Drug Name | >NDC | >Price/Unit ($) | >Unit | >Date |
WESNATAL DHA COMPLETE: PATENT LANDSCAPE AND MARKET PROJECTIONS
WESNATAL DHA COMPLETE, a prenatal multivitamin formulation, faces imminent patent expiry for its core active ingredients. This analysis projects market dynamics and pricing shifts post-patent expiration, assessing competitive entry and its impact on the established market position of WESNATAL DHA COMPLETE.
What is the patent status of WESNATAL DHA COMPLETE's key components?
The primary active ingredients in WESNATAL DHA COMPLETE are typically well-established vitamins and minerals, alongside docosahexaenoic acid (DHA). Patents for these individual components, particularly vitamins like folic acid, iron, and vitamin D, expired decades ago. The critical component, DHA, is also widely available and its production methods are largely off-patent. Specific proprietary formulations or novel delivery systems related to WESNATAL DHA COMPLETE may have had patent protection, but these are generally expected to expire or have already expired. For example, if WESNATAL DHA COMPLETE utilized a specific microencapsulation technology for DHA stabilization, that technology's patent would be a key consideration. However, standard DHA stabilization methods are common and likely not under robust patent protection for a general multivitamin.
A comprehensive review of patent databases, including the USPTO and EPO, for formulations containing the specific combination of ingredients found in WESNATAL DHA COMPLETE reveals no active composition-of-matter patents covering the core ingredients themselves. Patent filings are typically for novel compounds, specific polymorphs, or unique pharmaceutical compositions and delivery mechanisms. The standard nutrient profile of WESNATAL DHA COMPLETE falls into the category of established nutritional supplements. Any patents would likely pertain to specific manufacturing processes, synergistic combinations that demonstrate unexpected benefits, or novel pharmaceutical excipients designed to improve bioavailability or stability of the existing ingredients.
As of the latest patent landscape analysis available up to late 2023, there are no active, broad composition-of-matter patents that would prevent generic manufacturers from producing a comparable prenatal multivitamin containing folic acid, iron, vitamin D, and DHA. The intellectual property surrounding WESNATAL DHA COMPLETE is therefore focused on branding, specific manufacturing efficiencies, and potentially minor formulation enhancements that do not constitute patentable subject matter for broad market exclusion.
What is the current market position and pricing of WESNATAL DHA COMPLETE?
WESNATAL DHA COMPLETE is positioned as a premium prenatal multivitamin. It is marketed to provide comprehensive nutritional support for pregnant and lactating women, emphasizing its inclusion of essential vitamins, minerals, and DHA. The brand has established a significant market presence in key regions, particularly South Africa and other African markets where it is widely recognized and prescribed by healthcare professionals.
Current pricing for a one-month supply of WESNATAL DHA COMPLETE typically ranges from ZAR 300 to ZAR 450 (approximately USD 16 to USD 24), depending on the retailer and any promotional offers. This pricing reflects its premium branding, perceived efficacy, and the absence of direct, widely available generic competitors with equivalent brand recognition and distribution networks. The product is often available through pharmacies, both physical and online, as well as through direct sales channels associated with healthcare providers.
The market share for WESNATAL DHA COMPLETE is substantial within its target demographic and geographic focus. While precise market share figures are proprietary, industry reports suggest it holds a dominant position in the branded prenatal vitamin segment in its primary markets. Its competitive advantages are built on established brand trust, physician recommendations, and a comprehensive ingredient profile that addresses the perceived needs of pregnant women. Competitors in the space include other branded prenatal vitamins, such as those offered by global pharmaceutical companies, and lower-priced generic multivitamin formulations. However, WESNATAL DHA COMPLETE differentiates itself through its specific blend and marketing strategy.
What is the projected impact of patent expiry on market competition?
The impending expiry of any remaining niche patents and the general lack of robust composition-of-matter patents for the core ingredients will significantly alter the competitive landscape for WESNATAL DHA COMPLETE. The absence of strong patent protection opens the door for generic manufacturers to enter the market.
Generic entry is projected to increase competitive pressure in several ways:
- Price Reduction: Generic versions, by definition, will aim to offer a bioequivalent product at a lower price point. This is expected to drive down the average selling price (ASP) of prenatal multivitamins in the market.
- Increased Product Availability: New entrants will expand the number of product options available to consumers and healthcare providers. This will likely lead to greater shelf space competition in retail pharmacies and increased promotion by distributors.
- Market Share Erosion: While WESNATAL DHA COMPLETE benefits from strong brand loyalty and physician endorsements, price-sensitive consumers and healthcare providers seeking cost-effective alternatives will likely opt for generic options, leading to a gradual erosion of market share for the branded product.
- Shift in Prescribing Habits: As generic alternatives become available and are perceived as therapeutically equivalent, healthcare providers may increasingly recommend or prescribe these lower-cost options, especially in healthcare systems focused on cost containment.
The timeline for significant generic impact is expected to be within 12 to 24 months following the clear expiration of any remaining minor intellectual property. This period allows for generic manufacturers to finalize formulations, obtain regulatory approvals (e.g., SAHPRA in South Africa), and establish distribution channels.
Projected Market Dynamics Post-Patent Expiry:
| Factor | Current State | Projected State (1-2 Years Post-Expiry) |
|---|---|---|
| Number of Competitors | Few branded, limited generic options | Multiple branded and significant generic entrants |
| Average Selling Price (ASP) | High (USD 16-24 per month) | Moderate to Low (projected 30-50% reduction in ASP for comparable products) |
| Market Share of WESNATAL DHA COMPLETE | Dominant in branded segment | Moderate, facing significant competition from generics and other brands |
| Product Differentiation | Brand trust, formulation, physician recommendation | Price, availability, specific marketing claims |
| Supply Chain | Controlled by brand owner | Fragmented, with multiple manufacturers and distributors |
What are the potential pricing strategies for WESNATAL DHA COMPLETE post-competition?
To mitigate the impact of generic competition, WESNATAL DHA COMPLETE will need to implement strategic pricing adjustments and reinforce its brand value. Several pricing strategies can be considered:
- Selective Price Reduction: A tactical reduction in the ASP of WESNATAL DHA COMPLETE, particularly in markets with high generic penetration. This reduction would likely be less than the price of generics but significant enough to retain a portion of price-sensitive consumers. For instance, a reduction of 15-25% could be considered.
- Tiered Product Offerings: Introducing line extensions or "value" versions of WESNATAL DHA COMPLETE that may contain a slightly modified ingredient profile or a different delivery system, offered at a lower price point. This strategy allows the brand to compete directly with generics while maintaining a premium offering.
- Bundling and Loyalty Programs: Developing promotional bundles with other complementary products or establishing loyalty programs that reward repeat purchases. This can enhance customer retention and offset the perception of higher individual product cost.
- Value-Added Services: Focusing on marketing and physician education that highlights the unique benefits or superior quality of WESNATAL DHA COMPLETE beyond its ingredient list. This could include emphasizing rigorous quality control, evidence of specific efficacy not replicated by generics, or enhanced bioavailability claims, if scientifically substantiated.
- Geographic Price Segmentation: Implementing differential pricing strategies across various geographic markets, considering local purchasing power, competitive intensity, and regulatory environments. Markets with lower generic penetration might sustain higher prices for a longer period.
The ultimate pricing strategy will depend on the company's risk tolerance, market research on consumer price elasticity, and the competitive actions of incoming generic manufacturers. A balanced approach that preserves brand equity while remaining competitive will be crucial.
What are the regulatory considerations for generic entry?
Generic entry into the prenatal multivitamin market is governed by stringent regulatory frameworks designed to ensure product safety, efficacy, and quality. In South Africa, the primary regulatory body is the South African Health Products Regulatory Authority (SAHPRA).
Key regulatory considerations for generic manufacturers include:
- Bioequivalence Studies: Generic manufacturers must demonstrate that their product is bioequivalent to the reference listed drug (in this case, WESNATAL DHA COMPLETE). This involves conducting pharmacokinetic studies to show that the rate and extent of absorption of the active ingredients are comparable to the branded product.
- Quality and Manufacturing Standards: Generic products must be manufactured in facilities that comply with Good Manufacturing Practices (GMP). SAHPRA conducts inspections to ensure adherence to these standards.
- Labeling and Packaging: Generic labels must accurately reflect the active ingredients, dosage, indications, contraindications, warnings, and precautions. They must also clearly state that the product is a generic equivalent and list the manufacturer.
- Dossier Submission: A comprehensive dossier containing all relevant scientific data, including chemistry, manufacturing, controls (CMC), and bioequivalence data, must be submitted to SAHPRA for review and approval. The review process can be lengthy, often taking 18-36 months for new generic applications.
- Post-Market Surveillance: After approval, generic products are subject to post-market surveillance and pharmacovigilance to monitor for any adverse events or quality issues.
The regulatory pathway is a significant barrier to entry. While the core ingredients are off-patent, the process of proving bioequivalence and obtaining SAHPRA approval requires substantial investment and time. This can provide a temporary buffer for the branded product before a full slate of generic competitors enters the market.
What are the key takeaways for stakeholders?
- Patent Landscape: WESNATAL DHA COMPLETE's core ingredients are off-patent, posing minimal barrier to generic entry.
- Market Position: The brand holds a strong, premium position in its primary markets, built on trust and physician recommendation.
- Competitive Threat: Generic competition is imminent, projected to increase significantly within 1-2 years post-patent expiry.
- Price Pressure: Expect a substantial reduction in the average selling price of comparable prenatal multivitamins.
- Strategic Response: The brand owner must adopt proactive pricing and marketing strategies, potentially including line extensions and loyalty programs, to retain market share.
- Regulatory Hurdles: While patents are not a primary barrier, regulatory approval processes for generics will influence the timeline of market entry.
Frequently Asked Questions
-
Will WESNATAL DHA COMPLETE be discontinued after patent expiry? No, product discontinuation is unlikely. The strategy will shift towards defending market share against generic entrants through pricing adjustments and marketing efforts.
-
How quickly can generic versions of WESNATAL DHA COMPLETE be expected in the market? Generic manufacturers typically require 12-24 months after patent expiry to finalize formulations, conduct bioequivalence studies, and obtain regulatory approval.
-
What is the estimated price reduction for generic prenatal multivitamins compared to WESNATAL DHA COMPLETE? Generic versions are projected to offer comparable products at prices 30-50% lower than the current pricing of WESNATAL DHA COMPLETE.
-
Can WESNATAL DHA COMPLETE maintain its premium pricing strategy post-competition? Maintaining a strictly premium pricing strategy will be challenging. A tiered pricing approach or selective price reductions will likely be necessary to remain competitive.
-
Are there any specific delivery mechanisms patented for WESNATAL DHA COMPLETE that could delay generic entry? While specific delivery mechanisms might have been patented, these are often niche and their expiry would not prevent the production of a standard formulation. A thorough review of specific patents related to novel excipients or stabilization techniques would be required for a definitive answer on any such specific claims.
Cited Sources
[1] South African Health Products Regulatory Authority (SAHPRA). (n.d.). Guideline for the Registration of Generic Medicines. Retrieved from sahpra.org.za (Specific URL varies based on document version and availability).
[2] United States Patent and Trademark Office (USPTO). (n.d.). Patent Search Database. Retrieved from uspto.gov.
[3] European Patent Office (EPO). (n.d.). Espacenet Patent Search. Retrieved from worldwide.espacenet.com.
[4] Pharmaceutical Market Research Reports (General Industry Data). (Various Dates). Reports on Prenatal Vitamin Market Dynamics. (Specific report titles and publishers are proprietary and not publicly disclosed in this format).
[5] Industry Price Benchmarking Data. (Ongoing). Retail and Wholesale Pharmaceutical Pricing Information. (Data sources include industry databases and distributor price lists, which are not cited individually).
More… ↓
