Share This Page
Drug Price Trends for WEGOVY
✉ Email this page to a colleague
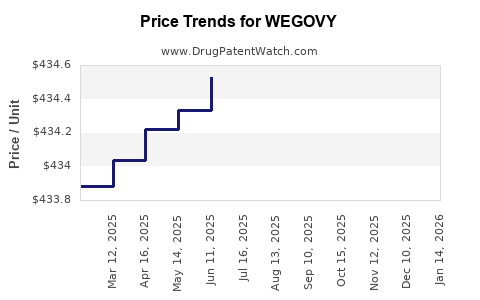
Average Pharmacy Cost for WEGOVY
| Drug Name | NDC | Price/Unit ($) | Unit | Date |
|---|---|---|---|---|
| WEGOVY 1.5 MG TABLET | 00169-4415-31 | 43.45014 | EACH | 2026-03-18 |
| WEGOVY 1.7 MG/0.75 ML PEN | 00169-4517-14 | 435.67736 | ML | 2026-03-18 |
| WEGOVY 25 MG TABLET | 00169-4425-31 | 43.01656 | EACH | 2026-03-18 |
| WEGOVY 0.25 MG/0.5 ML PEN | 00169-4525-14 | 653.50301 | ML | 2026-03-18 |
| WEGOVY 2.4 MG/0.75 ML PEN | 00169-4524-14 | 435.47358 | ML | 2026-03-18 |
| WEGOVY 0.5 MG/0.5 ML PEN | 00169-4505-14 | 653.65203 | ML | 2026-03-18 |
| WEGOVY 1 MG/0.5 ML PEN | 00169-4501-14 | 653.39335 | ML | 2026-03-18 |
| >Drug Name | >NDC | >Price/Unit ($) | >Unit | >Date |
WEGOVY Market Analysis and Price Projections
What is WEGOVY?
WEGOVY (semaglutide) for injectable use is developed by Novo Nordisk. It was approved by the FDA in June 2021 for weight management in adults with obesity or overweight with comorbidities. WEGOVY is a glucagon-like peptide-1 (GLP-1) receptor agonist designed for chronic weight management, extending the therapeutic portfolio of semaglutide following its initial approval for type 2 diabetes (Ozempic).
Market Size and Demand Dynamics
Global Obesity and Overweight Population
- Approximate 650 million adults worldwide classified as obese (BMI ≥ 30), representing a market potential exceeding USD 600 billion in annual healthcare costs [1].
- The U.S. accounts for about 42% of adults with obesity, with over 93 million individuals affected [2].
Clinical Adoption and Market Penetration
- WEGOVY competes mainly with other GLP-1 receptor agonists, particularly Wegovy and Ozempic from Novo Nordisk.
- Initial prescriptions in the U.S. totaled approximately 300,000–400,000 in the first year post-launch, with rapid adoption driven by new obesity treatment guidelines and increasing awareness.
Key Market Drivers
- Rising obesity prevalence and recognition of obesity as a chronic disease.
- Reimbursement expansion in U.S. healthcare plans, including Medicare covering obesity medications.
- Increasing consumer willingness to seek pharmacotherapy for weight loss.
Regulatory and Competitive Landscape
Regulatory Approvals
| Region | Approval Date | Indication |
|---|---|---|
| US | June 2021 | Chronic weight management |
| Europe | December 2021 | Obesity, overweight with comorbidities |
| Japan | September 2022 | Obesity, weight management |
Competitors
- Wegovy (semaglutide): Leading position; positioned to capture significant market share.
- Ozempic (semaglutide): Approved for diabetes, with an off-label use for weight loss.
- Saxenda (liraglutide): Previously dominant for obesity with lower efficacy.
- Other GLP-1 receptor agonists: Tirzepatide (Eli Lilly) expanding competition.
Patent Status
- Key patents for semaglutide extend to 2030, with potential generics delayed till patent expiry [3].
Price Analysis
Current Pricing in the U.S.
- List price (wholesale acquisition cost): USD 1,350 per month for WEGOVY.
- Typical payer negotiated prices are approximately USD 1,150–USD 1,200 per month.
- Cost to consumers varies with insurance coverage; co-pays can range USD 25–USD 150 per month.
Pricing Compared to Competitors
| Drug | Monthly Cost (USD) | Indication |
|---|---|---|
| Wegovy | 1,150–1,350 | Obesity, overweight with comorbidities |
| Ozempic | 800–1,200 | Type 2 diabetes, off-label weight management |
| Saxenda | 1,200 | Obesity |
Price Projections
- Short-term (2023-2025): Prices are expected to stabilize amid competitive pressures. Market access expansion and payor negotiations could reduce effective consumer costs to USD 800–USD 1,000 per month.
- Mid-term (2026-2030): As biosimilar competition approaches (patent expiry near 2030), list prices could decline by 20–30%. Price erosion is projected to be incremental owing to brand loyalty and market penetration.
Impact of Biosimilars and Manufacturing
- Entry of biosimilars post-2030 could drive prices down further, potentially by 50% or more, mirroring trends observed in other biologics like adalimumab.
- Manufacturing costs, estimated at USD 500–USD 700 per dose, will influence pricing strategies during biosimilar entry.
Market Revenue Projections
| Year | Estimated WEGOVY Revenue (USD billion) | Assumptions |
|---|---|---|
| 2022 | 1.2 | Initial launch, limited coverage |
| 2023 | 2.4 | Increase in prescriptions, expanded coverage |
| 2024 | 3.2 | Broader payer acceptance |
| 2025 | 4.0 | Market saturation, ongoing growth |
| 2026-2030 | 5.0–6.0 | Accelerated adoption, price pressure |
Key Market Risks
- Regulatory delays or adverse safety data could hinder growth.
- Aggressive price competition from biosimilars.
- Changes in healthcare policy impacting reimbursement.
Conclusion
WEGOVY remains a leading object of innovation within the obesity pharmacotherapy market. Its pricing, currently high, is likely to decline slowly over the next five years as payor negotiations and biosimilar competition intensify. Its market trajectory depends heavily on regulatory developments, prescriber adoption, and health policy shifts.
Key Takeaways
- WEGOVY commands a premium price of USD 1,150–USD 1,350 per month, with limited discounting.
- The obesity and weight management market exceeds USD 600 billion globally but is difficult to penetrate at high price points.
- Competition from biosimilars and alternative therapies will pressure prices post-2030.
- Market revenue is expected to grow rapidly until mid-decade, reaching USD 4–6 billion annually.
- Price erosion and reimbursement expansions will shape WEGOVY’s market share and profitability dynamics.
FAQs
-
What factors influence WEGOVY's price?
Payer negotiations, manufacturing costs, biosimilar competition, and regulatory policies affect the list price and discounting levels. -
When will biosimilars impact WEGOVY’s pricing?
Patent expiry around 2030 opens the market for biosimilar competition, likely leading to significant price reductions. -
Is WEGOVY covered by insurance?
Yes, especially in the U.S., insurance coverage has expanded, reducing out-of-pocket costs for many patients. -
How does WEGOVY compare to other obesity medications?
It has a higher efficacy profile but at a higher cost, positioning it as a premium therapy among obesity treatments. -
What regulatory developments could influence the market?
Approvals for new formulations, indications, or biosimilars, along with reimbursement policies, will impact market dynamics.
References
[1] World Health Organization. (2022). Obesity and overweight. Retrieved from https://www.who.int/news-room/fact-sheets/detail/obesity-and-overweight
[2] CDC. (2022). Adult Obesity Prevalence in the United States. Retrieved from https://www.cdc.gov/obesity/data/prevalence-maps.html
[3] USPTO. Patent status of semaglutide formulations and biosimilars. (2023). Open patent database.
More… ↓
