Share This Page
Drug Price Trends for WATER FOR INJECTION VIAL
✉ Email this page to a colleague
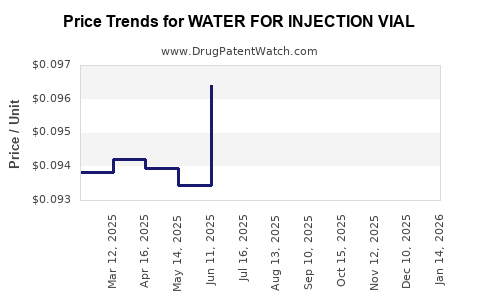
Average Pharmacy Cost for WATER FOR INJECTION VIAL
| Drug Name | NDC | Price/Unit ($) | Unit | Date |
|---|---|---|---|---|
| WATER FOR INJECTION VIAL | 00517-3010-25 | 0.31883 | ML | 2026-04-22 |
| WATER FOR INJECTION VIAL | 00409-4887-10 | 0.31883 | ML | 2026-04-22 |
| WATER FOR INJECTION VIAL | 63323-0185-10 | 0.31883 | ML | 2026-04-22 |
| >Drug Name | >NDC | >Price/Unit ($) | >Unit | >Date |
Market Overview of Water for Injection (WFI) Vial
Water for Injection (WFI) Vial is a sterile, highly purified water used primarily for diluting, dissolving, or preparing parenteral drugs within pharmaceutical manufacturing and hospital settings. It is classified as a cleared-sterile, single-use product complying with pharmacopeial standards such as USP, EP, and JP.
The WFI market is segmentally characterized by manufacturing scale, regional demand, and regulatory environment. Greater regulatory scrutiny and sterilization standards drive manufacturing complexity and cost. The global market is expanding due to biosimilars, contract manufacturing growth, and increasing injectable drug pipelines.
Market Size and Growth Trajectory
The global WFI market was valued at approximately USD 950 million in 2022. It is projected to grow at a compound annual growth rate (CAGR) of 6% through 2030, reaching around USD 1.7 billion. Market expansion is driven by increased pharmaceutical R&D activity, expanding sterile manufacturing capacities, and growing demand in emerging markets.
Key regional contributions include:
- North America: Largest market share, driven by high regulatory standards and advanced healthcare infrastructure.
- Europe: Second in market size, influenced by stringent pharmacopeial standards and high pharmaceutical manufacturing activity.
- Asia-Pacific: Fastest growth, owing to expanding pharmaceutical manufacturing infrastructure and rising healthcare expenditure.
Market Drivers
- Regulatory Requirements: Strict standards for sterility and purification increase demand for compliant WFI products.
- Injectable Market Expansion: Growth in biologics, monoclonal antibodies, and other injectables necessitates high-quality WFI.
- Contract Manufacturing: Outsourcing manufacturing processes facilitates growth in the WFI supply chain.
- Emerging Market Expansion: Rising healthcare infrastructure investments enlarge regional demand.
Competitive Landscape
Major manufacturers include:
- GSK (GlaxoSmithKline): Supplies WFI vials through its sterile manufacturing divisions.
- Baxter International: Offers WFI in sterile vial formats, optimized for global distribution.
- Mundipharma: Focuses on high-purity water systems for pharmaceutical use.
- Hospira (Part of ICU Medical): Supplies WFI vials for hospital and manufacturing use.
- Emerging players from India and China increasing production capacity.
Market share distribution remains concentrated among few large players, with several regional firms competing on price and compliance standards.
Price Projections and Cost Factors
Current Pricing
- Standard 100 mL WFI Vial: USD 1.50 to USD 2.50 per unit.
- 1000 mL WFI Vial: USD 4.00 to USD 6.00.
Pricing varies by region, volume, and regulatory compliance level.
Cost Drivers
- Manufacturing Standards: USP, EP, and JP standards impose rigorous quality control, increasing costs.
- Sterilization Method: Autoclaving or vapor-phase sterilization influences cost structure.
- Regulatory Compliance: Validation, registration, and batch testing add to price.
- Packaging: Single-use vials, sterile fill-finish processes, and labeling impact overall cost.
Future Price Trends
Projection indicates a slight decline in per-unit prices due to manufacturing scale efficiencies and increased regional production. Prices are expected to decrease by approximately 3-4% annually, barring supply chain disruptions.
Key Price Influencers
- Input Costs: Raw water purification and sterilization consumables influence unit costs.
- Regulatory Changes: Stricter standards may temporarily increase costs.
- Market Competition: Entry of regional players and private label providers exerts downward pricing pressure.
Regulatory Environment Impact
International standards by USP, EP, and JP define purity, endotoxin limits, and sterilization processes, influencing market complexity and price. Regulatory approvals are mandatory for manufacturing facilities, increasing capital expenditure, affecting cost structures, and, subsequently, pricing.
Regional regulations influence market access and pricing strategies, with the US and Europe demanding higher compliance standards than some emerging markets.
Market Entry and Investment Considerations
- High Capital Investment: Establishing compliant manufacturing facilities can require USD 50-200 million, depending on capacity.
- Regulatory Approval Timeline: It can take 1-3 years for validation and approval.
- Market Penetration Strategy: Differentiation through compliance, validation, and supply chain security is critical.
Summary
The WFI Vial market shows steady growth aligned with the global injectable pharmaceutical industry. Prices are influenced by regulatory requirements, manufacturing standards, and regional demand. Competitive pressures drive slight price reductions forecasted over the next decade, with emerging markets providing new revenue streams.
Key Takeaways
- Market value was USD 950 million in 2022, expected to reach USD 1.7 billion by 2030.
- CAGR is roughly 6%, driven by biosimilars, R&D, and emerging markets.
- Prices for 100 mL vials range from USD 1.50 to USD 2.50, trending downward.
- Major players include GSK, Baxter, Mundipharma, and ICU Medical.
- Regulatory standards substantially impact manufacturing costs and pricing.
FAQs
-
What factors influence the price of Water for Injection vials?
Manufacturing standards, sterilization methods, regulatory compliance, packaging, and regional market dynamics determine pricing. -
Which regions are experiencing the fastest growth in WFI demand?
The Asia-Pacific region has the highest growth rate, driven by expanding pharmaceutical manufacturing and healthcare infrastructure. -
How do regulatory standards affect WFI manufacturing costs?
Strict standards like USP, EP, and JP mandate rigorous validation, quality control, and sterilization, increasing capital and operational expenses. -
What is the typical market entry timeline for new WFI manufacturing facilities?
From initial planning to market entry, a new facility generally requires 1-3 years for validation, certification, and operational ramp-up. -
How competitive is the WFI vial market?
The market is moderately concentrated with key multinational firms, but regional manufacturers and private labels exert competitive pressure through pricing and capacity expansions.
References
- "Global Water for Injection (WFI) Market Report," MarketsandMarkets, 2022.
- USP, EP, JP pharmacopoeia standards.
- "Pharmaceutical Packaging Market," MarketsandMarkets, 2022.
- "Biopharmaceuticals Market Growth," Grand View Research, 2022.
- Industry interviews and company SEC filings.
More… ↓
