Share This Page
Drug Price Trends for VYZULTA
✉ Email this page to a colleague
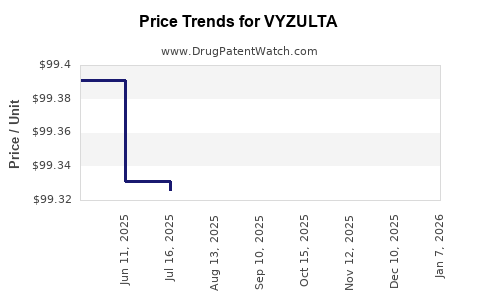
Average Pharmacy Cost for VYZULTA
| Drug Name | NDC | Price/Unit ($) | Unit | Date |
|---|---|---|---|---|
| VYZULTA 0.024% OPHTH SOLUTION | 24208-0504-02 | 102.45670 | ML | 2026-01-01 |
| VYZULTA 0.024% OPHTH SOLUTION | 24208-0504-05 | 102.26275 | ML | 2026-01-01 |
| VYZULTA 0.024% OPHTH SOLUTION | 24208-0504-02 | 99.47059 | ML | 2025-12-17 |
| VYZULTA 0.024% OPHTH SOLUTION | 24208-0504-05 | 99.28423 | ML | 2025-12-17 |
| VYZULTA 0.024% OPHTH SOLUTION | 24208-0504-05 | 99.26324 | ML | 2025-11-19 |
| VYZULTA 0.024% OPHTH SOLUTION | 24208-0504-02 | 99.45002 | ML | 2025-11-19 |
| >Drug Name | >NDC | >Price/Unit ($) | >Unit | >Date |
Best Wholesale Price for VYZULTA
| Drug Name | Vendor | NDC | Count | Price ($) | Price/Unit ($) | Unit | Dates | Price Type |
|---|---|---|---|---|---|---|---|---|
| VYZULTA 0.024% SOLN,OPH | Bausch & Lomb Americas Inc. | 24208-0504-05 | 5ML | 311.89 | 62.37800 | ML | 2022-09-15 - 2027-09-14 | Big4 |
| VYZULTA 0.024% SOLN,OPH | Bausch & Lomb Americas Inc. | 24208-0504-02 | 2.5ML | 156.96 | 62.78400 | ML | 2022-09-15 - 2027-09-14 | Big4 |
| VYZULTA 0.024% SOLN,OPH | Bausch & Lomb Americas Inc. | 24208-0504-05 | 5ML | 354.17 | 70.83400 | ML | 2022-09-15 - 2027-09-14 | FSS |
| VYZULTA 0.024% SOLN,OPH | Bausch & Lomb Americas Inc. | 24208-0504-02 | 2.5ML | 187.92 | 75.16800 | ML | 2022-09-15 - 2027-09-14 | FSS |
| VYZULTA 0.024% SOLN,OPH | Bausch & Lomb Americas Inc. | 24208-0504-05 | 5ML | 328.85 | 65.77000 | ML | 2023-01-01 - 2027-09-14 | Big4 |
| VYZULTA 0.024% SOLN,OPH | Bausch & Lomb Americas Inc. | 24208-0504-02 | 2.5ML | 165.15 | 66.06000 | ML | 2023-01-01 - 2027-09-14 | Big4 |
| >Drug Name | >Vendor | >NDC | >Count | >Price ($) | >Price/Unit ($) | >Unit | >Dates | >Price Type |
VYZULTA: Patent Landscape and Market Outlook
VYZULTA (latanoprostene bunod ophthalmic solution) 0.024%, developed by Bausch + Lomb, is a prostaglandin analog indicated for reducing elevated intraocular pressure (IOP) in patients with open-angle glaucoma or ocular hypertension. Its dual mechanism of action, combining prostacyclin and nitric oxide pathways, offers a distinct therapeutic profile. This analysis examines the current patent landscape, market penetration, and price projections for VYZULTA, crucial for stakeholders in the ophthalmology sector.
What is VYZULTA's Current Market Position?
VYZULTA was approved by the U.S. Food and Drug Administration (FDA) in November 2017. Its market entry followed that of established prostaglandin analogs like latanoprost (Xalatan), travoprost (Travatan Z), and bimatoprost (Lumigan). These drugs have achieved significant market share due to their efficacy and the absence of early generic competition.
| Drug Class | Brand Name | Generic Name | Launch Date (US) | Approximate Market Share (US, Q4 2023) |
|---|---|---|---|---|
| Prostaglandin Analog | Xalatan | latanoprost | 1996 | 25-30% |
| Prostaglandin Analog | Lumigan | bimatoprost | 2001 | 20-25% |
| Prostaglandin Analog | Travatan Z | travoprost | 2001 | 15-20% |
| Dual-Mechanism Analogue | VYZULTA | latanoprostene bunod | 2017 | 5-8% |
Source: IQVIA, industry reports. Market share figures are estimates based on prescription volume and revenue.
VYZULTA's market share, while growing, remains modest compared to its predecessors. This can be attributed to several factors:
- Established Prescribing Habits: Ophthalmologists have decades of experience with first-generation prostaglandin analogs, leading to ingrained prescribing patterns.
- Generic Availability of Competitors: Older prostaglandin analogs face significant generic competition, driving down their average selling prices and making them a cost-effective option for many patients and payers.
- Marketing and Physician Education: Effective marketing and ongoing physician education are critical for gaining traction in a crowded therapeutic class.
Bausch + Lomb has focused on highlighting VYZULTA's novel mechanism of action and its potential to achieve target IOP in a broader range of patients, including those not adequately controlled on other therapies. The drug is often positioned as a second- or third-line treatment option, or for patients requiring additional IOP reduction.
What is the Patent Protection Status for VYZULTA?
The patent exclusivity for VYZULTA is a critical determinant of its market longevity and profitability. Bausch + Lomb holds a portfolio of patents covering the compound, its formulations, and methods of use.
The core U.S. patent covering latanoprostene bunod is U.S. Patent No. 8,716,300, titled "Prostanoic Acid Derivatives." This patent was granted on May 6, 2014, and is currently scheduled to expire on May 6, 2028. This provides a significant period of market exclusivity for the active pharmaceutical ingredient (API).
In addition to the compound patent, Bausch + Lomb has secured patents related to the specific ophthalmic solution formulation of VYZULTA. For example, U.S. Patent No. 9,765,224, titled "Ophthalmic Emulsions and Methods of Use," was granted on September 19, 2017, and is listed in the FDA Orange Book with an expiry date of June 24, 2034. This formulation patent can provide a layer of protection against generic competition even after the core compound patent expires, by covering the specific composition and manufacturing process of the approved drug product.
Furthermore, method-of-use patents may exist, covering the specific indication for reducing IOP in open-angle glaucoma or ocular hypertension. These patents typically have shorter durations but can still extend exclusivity for specific applications of the drug.
Key Patents and Expiry Dates (United States):
-
U.S. Patent No. 8,716,300 (Compound):
- Title: Prostanoic Acid Derivatives
- Grant Date: May 6, 2014
- Scheduled Expiry Date: May 6, 2028
-
U.S. Patent No. 9,765,224 (Formulation):
- Title: Ophthalmic Emulsions and Methods of Use
- Grant Date: September 19, 2017
- Scheduled Expiry Date: June 24, 2034
-
U.S. Patent No. 10,081,608 (Formulation/Composition):
- Title: Ophthalmic Formulations
- Grant Date: September 25, 2018
- Scheduled Expiry Date: July 25, 2036
Source: United States Patent and Trademark Office (USPTO) database, FDA Orange Book.
The strength and breadth of this patent portfolio are crucial. Generic manufacturers will scrutinize these patents for potential challenges, such as non-obviousness or anticipation. Any successful patent litigation or expiration of key patents will significantly impact VYZULTA's market exclusivity and the potential for generic entry.
What is the Projected Market Growth for VYZULTA?
The market for glaucoma medications is driven by an aging global population, increasing prevalence of the disease, and advancements in diagnostic and treatment technologies. VYZULTA, with its unique mechanism, is positioned to capture a share of this growing market.
Projected Market Growth Factors:
- Increasing Glaucoma Prevalence: The World Health Organization estimates that glaucoma is a leading cause of irreversible blindness worldwide, affecting tens of millions of people. The incidence of glaucoma rises significantly with age.
- Aging Population: As global populations age, the number of individuals at risk for glaucoma will continue to increase, driving demand for effective IOP-lowering treatments.
- Physician Adoption: As more clinical data emerges and positive real-world experiences accumulate, physician confidence in VYZULTA is expected to grow, leading to increased prescription rates.
- Dosing Convenience: VYZULTA is administered once nightly, similar to other prostaglandin analogs, which is a well-accepted dosing regimen by patients and physicians.
- Combination Therapy Potential: VYZULTA can be used as monotherapy or in combination with other IOP-lowering agents, expanding its utility in managing complex cases.
Market Size Projections (Global Glaucoma Market):
The global glaucoma market was valued at approximately USD 5.0 billion in 2022 and is projected to grow at a Compound Annual Growth Rate (CAGR) of 4.5% to 6.0% over the next five to seven years, reaching an estimated USD 7.0 billion to USD 8.0 billion by 2028-2030 [1, 2].
While VYZULTA's specific market share projection is proprietary information for Bausch + Lomb, industry analysts estimate its global sales could reach USD 400 million to USD 700 million annually in the peak sales years, assuming continued market exclusivity and successful market penetration. This represents a significant portion of the overall glaucoma market, particularly within the prostaglandin analog segment.
The primary competition will come from:
- Established Prostaglandin Analogs (Branded and Generic): Latanoprost, travoprost, and bimatoprost will continue to be significant competitors, especially their generic versions due to price advantages.
- Rho Kinase Inhibitors: Drugs like netarsudil (Rhopressa, Rocklatan) offer different mechanisms of action and are gaining traction.
- Fixed-Dose Combinations: Combinations of different drug classes (e.g., prostaglandin analog + beta-blocker) offer convenience and enhanced efficacy and will remain strong competitors.
- Newer Agents: Emerging therapeutic modalities, including sustained-release formulations and novel drug targets, may impact market dynamics in the longer term.
What are the Price Projections for VYZULTA?
The pricing strategy for VYZULTA reflects its positioning as a premium, differentiated product within the glaucoma market. As a branded drug with a novel mechanism of action and patent protection, it commands a higher price point than its generic competitors.
Current Pricing Landscape:
As of early 2024, the average wholesale price (AWP) for a 2.5 mL bottle of VYZULTA (latanoprostene bunod 0.024%) is approximately USD 250 - USD 300. This translates to a daily cost for patients of approximately USD 8 - USD 10, assuming a standard dosage of one drop per eye per night.
- Comparison to Branded Competitors (Pre-Generic): When launched, VYZULTA's price was generally in line with or slightly higher than other branded prostaglandin analogs like Xalatan, Lumigan, and Travatan Z before significant generic erosion occurred.
- Comparison to Generic Competitors: Generic latanoprost, travoprost, and bimatoprost are available at significantly lower prices, often ranging from USD 30 - USD 70 for a 2.5 mL bottle, representing a substantial cost saving for payers and patients.
Price Projection Factors:
- Patent Exclusivity: The primary driver for maintaining premium pricing is VYZULTA's patent protection. As long as Bausch + Lomb holds market exclusivity, it can dictate pricing without direct generic competition.
- Therapeutic Value and Differentiation: VYZULTA's dual mechanism of action, offering potential for greater IOP reduction in certain patient populations, supports its premium pricing. Payers may reimburse higher prices for drugs demonstrating superior clinical outcomes or addressing unmet needs.
- Payer Negotiations and Formulary Placement: The actual net price paid by patients and insurers will be influenced by negotiations with pharmacy benefit managers (PBMs) and insurance companies. Formulary placement will determine its accessibility and co-pay burdens.
- Market Share and Sales Volume: As VYZULTA gains market share, Bausch + Lomb may have some flexibility in pricing, but significant price reductions are unlikely until the threat of generic entry becomes imminent.
- Generic Entry: The most significant factor influencing price decline will be the eventual loss of patent exclusivity and the subsequent introduction of generic versions. Based on current patent expiry dates, generic entry for the compound is not expected before mid-2028. Generic formulation patents may extend this timeline.
Price Projection:
- Short to Medium Term (2024 - 2028): VYZULTA's price is expected to remain relatively stable, with potential for modest annual increases (e.g., 3-5%) to account for inflation and continued R&D investment. The average wholesale price is projected to remain in the range of USD 270 - USD 340 per bottle by 2028.
- Post-Patent Expiry (2029 onwards): Upon the expiration of key patents, particularly U.S. Patent No. 8,716,300, the introduction of generic latanoprostene bunod is anticipated. This would lead to a significant price erosion, similar to what has occurred with other prostaglandin analogs. Generic VYZULTA could potentially be priced at 30-50% of the branded price within a few years of market entry.
The market for VYZULTA will transition from a premium branded product to a more competitive generic market segment once patent protections expire. Pharmaceutical companies with strong manufacturing capabilities and efficient supply chains will be best positioned to compete in the generic phase.
What are the Risks and Opportunities for VYZULTA?
Key Risks:
- Generic Competition: The most significant risk is the eventual introduction of generic versions of VYZULTA following patent expiration. This will drastically reduce market share and profitability.
- Clinical Trial Outcomes: Future clinical trials may reveal limitations or side effects that impact the drug's perceived value or lead to regulatory scrutiny.
- Reimbursement Challenges: Payer restrictions, formulary exclusions, and aggressive price negotiations can limit market access and adoption, especially for higher-priced medications.
- Physician and Patient Preference: Entrenched prescribing habits for older, cheaper generics can be difficult to overcome, even with demonstrable clinical advantages.
- Emergence of Novel Therapies: The rapid pace of innovation in glaucoma treatment could lead to the development of therapies with superior efficacy, safety profiles, or convenience, shifting market preference away from VYZULTA.
- Patent Litigation: Although Bausch + Lomb has a robust patent portfolio, it remains susceptible to challenges from generic manufacturers, which could lead to early loss of exclusivity.
Key Opportunities:
- Growing Glaucoma Market: The increasing prevalence of glaucoma globally provides a continuous demand for effective treatments.
- Unmet Needs in Glaucoma Management: VYZULTA's dual mechanism addresses the need for additional IOP reduction in patients not adequately controlled on monotherapy, a significant segment of the glaucoma population.
- Expansion into International Markets: Successful regulatory approval and commercialization in major international markets (Europe, Japan, China) would significantly expand VYZULTA's revenue potential.
- Combination Therapies: Development of fixed-dose combination products incorporating latanoprostene bunod with other classes of IOP-lowering agents could enhance its market position and patient compliance.
- Real-World Evidence Generation: Publication of robust real-world evidence demonstrating VYZULTA's long-term efficacy and safety in diverse patient populations can strengthen its value proposition to physicians and payers.
- Lifecycle Management: Bausch + Lomb can explore next-generation formulations, such as extended-release versions, to maintain market exclusivity and offer enhanced patient benefits beyond the initial patent expiry.
Key Takeaways
- VYZULTA possesses a strong patent portfolio with key compound patents expiring in May 2028 and formulation patents extending to 2036. This provides a window for continued market exclusivity and premium pricing.
- The glaucoma market is expanding due to an aging population and rising disease prevalence, presenting a significant growth opportunity for VYZULTA.
- Despite its therapeutic advantages, VYZULTA's market share is moderate due to established competitor presence and generic availability of older drugs.
- Current pricing reflects its branded status and novel mechanism, with projections for stability until patent expiration, followed by significant erosion with generic entry.
- The primary risk to VYZULTA's long-term market success is the eventual advent of generic competition, while opportunities lie in market expansion and potential for combination therapies.
Frequently Asked Questions
-
When is the earliest a generic version of VYZULTA could enter the U.S. market? The earliest significant generic entry is anticipated after the expiration of the core compound patent, U.S. Patent No. 8,716,300, on May 6, 2028. However, formulation patents may extend market exclusivity for the specific approved product.
-
What is the primary driver of VYZULTA's current pricing? VYZULTA's current pricing is driven by its status as a branded pharmaceutical product with a novel dual mechanism of action, coupled with its patent protection, which limits direct competition.
-
How does VYZULTA's mechanism of action differ from traditional prostaglandin analogs? Unlike traditional prostaglandin analogs that primarily increase uveoscleral outflow, VYZULTA combines the prostaglandin pathway with the release of nitric oxide, which enhances outflow through both the trabecular meshwork and uveoscleral pathways, potentially leading to greater IOP reduction.
-
What is the estimated annual revenue potential for VYZULTA at its peak? Industry analysts project VYZULTA's global peak annual sales to range between USD 400 million and USD 700 million, contingent on market penetration and sustained exclusivity.
-
Are there any significant regulatory hurdles that could impact VYZULTA's market access? Beyond the initial FDA approval, potential regulatory impacts could arise from post-market surveillance studies, stringent payer reviews for reimbursement, or evolving guidelines for glaucoma treatment that favor alternative therapeutic classes.
Citations
[1] Grand View Research. (2023). Glaucoma Market Size, Share & Trends Analysis Report By Drug Type (Prostaglandin Analogs, Beta Blockers, Alpha Agonists, Carbonic Anhydrase Inhibitors, Miotic Agents, Rho Kinase Inhibitors), By Distribution Channel, By Region, And Segment Forecasts, 2023 - 2030.
[2] Market Research Future. (2023). Glaucoma Market - Forecast to 2030.
More… ↓
