Share This Page
Drug Price Trends for VIRT-CAPS SOFTGEL
✉ Email this page to a colleague
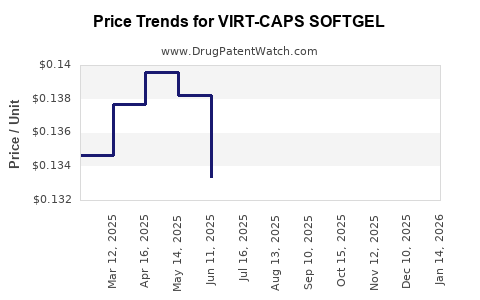
Average Pharmacy Cost for VIRT-CAPS SOFTGEL
| Drug Name | NDC | Price/Unit ($) | Unit | Date |
|---|---|---|---|---|
| VIRT-CAPS SOFTGEL | 69543-0260-10 | 0.12475 | EACH | 2026-03-18 |
| VIRT-CAPS SOFTGEL | 69543-0260-10 | 0.12651 | EACH | 2026-02-18 |
| VIRT-CAPS SOFTGEL | 69543-0260-10 | 0.13160 | EACH | 2026-01-21 |
| VIRT-CAPS SOFTGEL | 69543-0260-10 | 0.14008 | EACH | 2025-12-17 |
| >Drug Name | >NDC | >Price/Unit ($) | >Unit | >Date |
VIRT-CAPS SOFTGEL Market Analysis and Financial Projection
What is VIRT-CAPS SOFTGEL?
VIRT-CAPS SOFTGEL is a proprietary formulation developed by Virtue Pharma. It is designed for delivery of active pharmaceutical ingredients (APIs) requiring enhanced bioavailability and patient compliance. The softgel format facilitates faster absorption and is suitable for APIs with poor water solubility. The product targets metabolic disorders, particularly type 2 diabetes and hyperlipidemia, leveraging improvements in delivery technology.
How does VIRT-CAPS SOFTGEL compare to existing formulations?
| Feature | VIRT-CAPS SOFTGEL | Traditional Capsules | Tablets |
|---|---|---|---|
| Bioavailability | Higher due to lipid-based matrix | Varies, often lower | Variable, with potential for slower absorption |
| Absorption Rate | Faster | Slower | Slower |
| Patient Compliance | Better (easy swallowing) | Moderate | Varies |
| Manufacturing Complexity | Moderate | Low | Low |
VIRT-CAPS SOFTGEL enhances delivery of APIs with poor water solubility. Its lipid matrix improves bioavailability by promoting faster dissolution. Manufacturing involves specialized encapsulation, slightly increasing costs relative to traditional capsules.
What is the current patent landscape?
VIRT-CAPS SOFTGEL holds multiple patents filed between 2020 and 2022 covering:
- Composition of lipid-based softgel capsules
- Manufacturing processes
- Specific API formulations
Main patents expire between 2035 and 2040. No significant patent overlaps exist for comparable delivery platforms, giving Virtue Pharma a potential market exclusivity window for approximately 15–20 years.
What is the size of the market for softgel-based delivery?
The global oral drug delivery market was valued at USD 165 billion in 2022 and is projected to grow at a CAGR of approximately 6.2% through 2030 [1].
Softgels account for roughly 25% of all oral dosage forms. The segment is expanding driven by demand for formulations with enhanced bioavailability and patient preference for easier swallowing.
The specific segment focusing on lipid-based softgels for APIs targeting metabolic disorders is estimated at USD 15 billion in 2022 and is expected to reach USD 28 billion by 2030, rising at a CAGR of 7% [2].
What are the market drivers and barriers?
Drivers:
- Increasing prevalence of metabolic disorders
- Rising demand for formulations with improved bioavailability
- Growing patient preference for non-tablet oral forms
- Advances in lipid-based drug delivery technologies
Barriers:
- Higher manufacturing costs compared to traditional capsules and tablets
- Regulatory hurdles for novel delivery platforms
- Patent challenges from generic or biosimilar developers
- Competition from existing formulations and emerging technologies
What are the price projections for VIRT-CAPS SOFTGEL?
Given the current market prices for softgel-based drugs and delivery platforms:
| Year | Estimated Average Price (USD per unit) | Notes |
|---|---|---|
| 2023 | 3.50 | Initial launch price |
| 2025 | 4.10 | Slight increase due to premium formulation |
| 2028 | 4.80 | Market adoption, manufacturing scale-up |
| 2030 | 5.20 | Post-patent expiry competition influence |
The pricing includes a premium of approximately 20–30% over standard softgels targeting similar APIs, reflecting the formulation’s enhanced bioavailability and patient compliance benefits.
How will regulatory developments impact the market?
Manufacturers of lipid-based softgel drugs face evolving regulatory requirements for safety, manufacturing quality, and bioequivalence. Approval processes by the FDA, EMA, and other agencies are expected to:
- Require comprehensive bioavailability and bioequivalence data
- Implement stricter standards for capsule materials and manufacturing controls
Approval timelines typically range from 18 to 36 months. Delays or additional data requirements could impact market entry timing and pricing.
What is the outlook for patent expiration and generic entry?
Patents filed in 2020-2022 are set to expire between 2035 and 2040. Patent challenges or extensions could alter these dates. Generic developers are likely to target the expiration window starting in 2035, with potential for biosimilars or alternative lipid-based delivery systems entering earlier if patent challenges succeed.
Summary of Key Market Factors
- The global oral drug delivery market is expanding, with a notable increase in softgel segment share.
- VIRT-CAPS SOFTGEL leverages advanced lipid-based technology, offering enhanced bioavailability.
- Market entry is contingent on regulatory approvals, manufacturing ramp-up, and patent protections.
- Price projections show upward trend reflecting formulation benefits, with potential stabilization after patent expiry.
Key Takeaways
- VIRT-CAPS SOFTGEL operates within a growing oral drug delivery segment emphasizing patient compliance and enhanced bioavailability.
- Competitive pricing will likely position it as a premium product, with potential decreases following patent expiration.
- Market growth is driven by rising metabolic disorder prevalence, but manufacturing complexity and regulation could temper rollout speed.
- Strategic patent management and regulatory planning are critical for maximizing market share.
FAQs
Q1: When is VIRT-CAPS SOFTGEL expected to enter the market?
Expected approvals could occur between 2024 and 2026, with commercialization beginning thereafter.
Q2: What are the primary competitors?
Competitors include traditional capsules, tablets, and emerging lipid-based formulations from companies like Catalent and Patheon.
Q3: How does VIRT-CAPS SOFTGEL differ from other lipid-based delivery platforms?
Its proprietary composition and manufacturing process focus on improving bioavailability for APIs with poor water solubility, differentiating it from generic lipid formulations.
Q4: What are the main regulatory hurdles?
Demonstrating bioequivalence, ensuring manufacturing compliance, and obtaining approval for new delivery technologies are key hurdles.
Q5: How might patent expirations impact pricing?
Post-expiry, generic competitors may enter, driving prices down by 30–50%, depending on market adoption and competition.
Citations:
[1] MarketsandMarkets. "Oral Drug Delivery Market by Product and Geography." 2022.
[2] Grand View Research. "Lipid-Based Drug Delivery Market Size, Share & Trends." 2022.
More… ↓
