Share This Page
Drug Price Trends for VIOKACE
✉ Email this page to a colleague
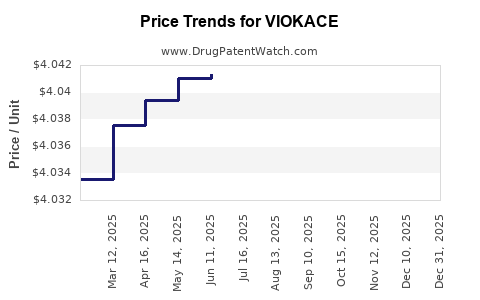
Average Pharmacy Cost for VIOKACE
| Drug Name | NDC | Price/Unit ($) | Unit | Date |
|---|---|---|---|---|
| VIOKACE 10,440-39,150 UNIT TAB | 73562-0104-10 | 4.13043 | EACH | 2026-05-20 |
| VIOKACE 10,440-39,150 UNIT TAB | 73562-0104-10 | 4.16287 | EACH | 2025-12-31 |
| VIOKACE 20,880-78,300 UNITS TB | 73562-0208-10 | 8.21251 | EACH | 2025-12-31 |
| VIOKACE 10,440-39,150 UNIT TAB | 73562-0104-10 | 4.04158 | EACH | 2025-12-17 |
| VIOKACE 20,880-78,300 UNITS TB | 73562-0208-10 | 7.97331 | EACH | 2025-12-17 |
| >Drug Name | >NDC | >Price/Unit ($) | >Unit | >Date |
Best Wholesale Price for VIOKACE
| Drug Name | Vendor | NDC | Count | Price ($) | Price/Unit ($) | Unit | Dates | Price Type |
|---|---|---|---|---|---|---|---|---|
| VIOKACE 20880 UNIT TABLET | Nestle HealthCare Nutrition, Inc. | 73562-0208-10 | 100 | 454.73 | 4.54730 | EACH | 2022-01-27 - 2026-11-30 | Big4 |
| VIOKACE 20880 UNIT TABLET | Nestle HealthCare Nutrition, Inc. | 58914-0117-10 | 100 | 606.16 | 6.06160 | EACH | 2022-01-01 - 2026-11-30 | FSS |
| VIOKACE 10440 UNIT TABLET | Nestle HealthCare Nutrition, Inc. | 73562-0104-10 | 100 | 364.02 | 3.64020 | EACH | 2024-01-01 - 2026-11-30 | FSS |
| VIOKACE 10440 UNIT TABLET | Nestle HealthCare Nutrition, Inc. | 58914-0112-10 | 100 | 251.85 | 2.51850 | EACH | 2023-01-01 - 2026-11-30 | Big4 |
| VIOKACE 10440 UNIT TABLET | Nestle HealthCare Nutrition, Inc. | 73562-0104-10 | 100 | 219.59 | 2.19590 | EACH | 2022-01-01 - 2026-11-30 | Big4 |
| >Drug Name | >Vendor | >NDC | >Count | >Price ($) | >Price/Unit ($) | >Unit | >Dates | >Price Type |
VIOKACE Market Analysis and Price Projections
VIOKACE, an oral formulation of pancreatic enzyme replacement therapy (PERT), faces a competitive landscape driven by physician preference, payer reimbursement policies, and ongoing clinical research. The current market for PERT is characterized by established players with long-standing market presence and a growing demand for improved patient adherence and efficacy. Price projections for VIOKACE will be influenced by its comparative effectiveness, the cost-effectiveness arguments presented to payers, and the pricing strategies of competing PERT products.
What is the current market size and projected growth for pancreatic enzyme replacement therapy (PERT)?
The global PERT market was valued at approximately $2.4 billion in 2022. This market is projected to grow at a compound annual growth rate (CAGR) of 5.2% from 2023 to 2030, reaching an estimated $3.6 billion by 2030. Growth drivers include an increasing incidence of pancreatic diseases such as cystic fibrosis and chronic pancreatitis, a rising awareness of PERT's benefits in managing maldigestion, and advancements in PERT formulations leading to improved patient compliance and outcomes.
Key PERT Market Segments:
- Indication: Cystic fibrosis, chronic pancreatitis, pancreatic cancer, post-pancreatectomy conditions.
- Formulation: Capsules, tablets, powders.
- Distribution Channel: Hospital pharmacies, retail pharmacies, online pharmacies.
The penetration of PERT in emerging economies is expected to increase, contributing to overall market expansion. However, high treatment costs and limited access to diagnosis in some regions may pose challenges.
How does VIOKACE compare to existing PERT products in terms of efficacy and patient outcomes?
VIOKACE is a non-enteric coated tablet formulation of pancrelipase. Unlike enteric-coated PERT products, VIOKACE is designed to be taken with a meal and requires antacid or proton pump inhibitor (PPI) co-administration to protect the enzymes from stomach acid degradation, ensuring their release and activity in the small intestine.
Comparative Efficacy and Outcomes:
- VIOKACE's Mechanism: The tablet form requires a buffering agent to facilitate enzyme release. Studies suggest that the concomitant use of antacids or PPIs with VIOKACE can achieve comparable gastrointestinal symptom relief to enteric-coated formulations in certain patient populations.
- Enteric-Coated PERT: Products like Creon, Pancreaze, and Zenpep utilize enteric coatings that dissolve in the higher pH of the duodenum, releasing enzymes directly into the small intestine without the need for co-administration of acid-reducing medications.
- Patient Adherence: The dosing regimen for VIOKACE, requiring co-administration of an acid suppressor, may present a potential adherence challenge compared to simpler enteric-coated formulations. However, for some patients, the tablet format may be preferred or better tolerated.
- Clinical Data: While specific head-to-head trials directly comparing VIOKACE's long-term outcomes against all leading enteric-coated PERTs are limited, clinical studies supporting VIOKACE's efficacy have focused on improvements in fat absorption and reduction of steatorrhea. For instance, a study published in Digestive Diseases and Sciences indicated that VIOKACE, when co-administered with omeprazole, significantly improved fat digestion and absorption in patients with exocrine pancreatic insufficiency [1].
The choice between VIOKACE and enteric-coated PERTs often depends on individual patient characteristics, physician experience, and the presence of comorbidities that might influence acid suppression therapy.
What are the key intellectual property considerations for VIOKACE?
VIOKACE's patent protection is a critical factor in its market exclusivity and pricing power. The core technology often involves the formulation, manufacturing process, and specific indications for use.
Intellectual Property Landscape:
- Composition of Matter Patents: These patents, if still active, would cover the chemical entity of the pancrelipase itself or novel combinations.
- Formulation Patents: Patents protecting the specific tablet composition, excipients, and manufacturing methods for VIOKACE are crucial. These can extend market exclusivity beyond the expiry of any foundational composition patents. For example, patents related to the non-enteric coating and the stability of enzymes in the tablet form are significant.
- Method of Use Patents: Patents covering specific dosing regimens or the use of VIOKACE in combination with acid suppressants for particular patient groups would also offer protection.
- Exclusivity Periods: The length of patent protection and any granted market exclusivities (e.g., Hatch-Waxman exclusivity for new chemical entities or new uses) will dictate the timeline for potential generic competition.
- Generic Entry: The expiry of key patents and exclusivities will open the door for generic manufacturers. The complexity of the manufacturing process and the regulatory hurdles for generic approval can influence the speed of generic market entry. Companies will scrutinize patent expiry dates for VIOKACE to plan their generic development and launch strategies.
Understanding the precise expiration dates of all relevant patents and exclusivities for VIOKACE and its active pharmaceutical ingredient is essential for projecting market dynamics and potential revenue erosion due to generic competition.
What is the current pricing of VIOKACE and how are prices projected to evolve?
The pricing of VIOKACE, like other PERT products, is determined by several factors including manufacturing costs, clinical value, competitive landscape, and payer negotiations. PERT pricing is generally high, reflecting the specialized nature of the indication and the chronic treatment duration.
Current Pricing Context:
- Wholesale Acquisition Cost (WAC): WAC for a typical daily supply of VIOKACE can range from $40 to $60, translating to an annual cost of $14,600 to $21,900, depending on the dosage and frequency of administration. This is a broad estimate and actual pricing can vary significantly based on contracted rates.
- Payer Reimbursement: Net prices after rebates and discounts negotiated with pharmacy benefit managers (PBMs) and insurance companies are substantially lower than WAC. Payer policies often dictate the preferred PERT products through formulary placement and prior authorization requirements.
- Competitive Benchmarking: VIOKACE pricing is benchmarked against established enteric-coated PERTs. The perceived value proposition and clinical differentiation influence its pricing position relative to competitors.
Price Projection Factors:
- Generic Competition: The most significant factor impacting future pricing will be the entry of generic VIOKACE or generic alternatives for comparable PERT products. Once generics enter, prices for branded VIOKACE are expected to decline by 50-80% within the first few years of generic availability, similar to trends seen with other branded pharmaceuticals.
- Payer Pressure: Continued emphasis on cost containment by payers will exert downward pressure on branded PERT prices. Rebate negotiations will become more aggressive.
- Clinical Value Demonstration: Strong evidence supporting VIOKACE's unique benefits or superior patient outcomes, particularly in specific patient subgroups, could support price maintenance or a premium. However, without clear differentiation, its pricing power will diminish as generic options emerge.
- Market Penetration and Volume: As VIOKACE gains market share or faces increased competition, pricing strategies may adjust to maintain volume.
- Inflationary Adjustments: Annual price increases, typically in line with inflation, are common for established pharmaceuticals, but these are often tempered by payer pushback and competitive pressures.
Assuming no unexpected patent extensions or significant clinical breakthroughs that create a substantial unmet need addressable only by VIOKACE, prices for branded VIOKACE are projected to decline significantly in the years following generic entry. The timeline for this decline is contingent on patent expiries and the successful launch of generics.
What regulatory hurdles and market access challenges does VIOKACE face?
Navigating regulatory pathways and securing broad market access are critical for the commercial success of any pharmaceutical product, including VIOKACE.
Regulatory Considerations:
- FDA Approval: VIOKACE has received FDA approval for the treatment of exocrine pancreatic insufficiency (EPI) due to chronic pancreatitis or pancreatectomy. The initial approval was granted based on demonstrated efficacy and safety.
- Labeling and Indications: The approved labeling dictates the claims that can be made and the patient populations for which the drug is indicated. Any expansion of indications would require further clinical trials and regulatory review.
- Manufacturing and Quality Control: Manufacturers must adhere to strict Good Manufacturing Practices (GMP) to ensure product quality, consistency, and safety. Post-market surveillance and inspections are ongoing.
- Post-Market Commitments: Regulatory agencies may impose post-market study requirements to gather additional safety or efficacy data.
Market Access Challenges:
- Payer Formularies: Gaining favorable placement on insurance formularies is essential. This involves demonstrating cost-effectiveness and clinical value to PBMs and health plans.
- Prior Authorization: Many PERT products require prior authorization, meaning healthcare providers must obtain approval from the patient's insurance company before prescribing. This can create delays in treatment initiation and administrative burden.
- Step Therapy: Payers may implement step-therapy protocols, requiring patients to try less expensive PERT options first before approving VIOKACE.
- Patient Co-pays and Affordability: High out-of-pocket costs for patients can be a significant barrier to adherence and treatment continuation, even with insurance coverage. Manufacturer patient assistance programs can mitigate this, but are costly to implement.
- Physician Prescribing Habits: Inertia in prescribing habits can be a challenge. Physicians may be more inclined to prescribe PERT products they are familiar with and have historically used successfully. Educating physicians about VIOKACE's benefits and differentiating factors is crucial.
- Competition: The presence of multiple established PERT products means that VIOKACE must actively compete for market share based on efficacy, safety, tolerability, and price.
Specific Challenges for VIOKACE:
The requirement for concomitant acid suppression therapy for VIOKACE is a key differentiator that can also be a market access challenge. Payers and physicians need to be convinced that this regimen is manageable for patients and does not introduce significant safety concerns or adherence issues. Demonstrating a favorable risk-benefit profile in comparison to the simpler regimen of enteric-coated products is critical for securing widespread market access.
What is the competitive landscape for VIOKACE?
The PERT market is moderately concentrated, with several key players offering a range of products. VIOKACE competes directly with these established therapies.
Key Competitors:
- AbbVie (Creon): Creon is a leading enteric-coated PERT product with a strong market presence.
- Nestlé Health Science (Zenpep, Pancreaze): Zenpep and Pancreaze are also significant enteric-coated PERT offerings that compete directly for patients with EPI.
- Enthera Pharmaceuticals (various pipeline products): While not yet a direct competitor for VIOKACE, emerging companies with pipeline PERT products could represent future competition.
- Generic PERT Products: As patent expiries occur, generic versions of existing branded PERTs will enter the market, intensifying price competition across the entire category.
Competitive Dynamics:
- Formulation Preference: The choice between VIOKACE's tablet formulation requiring acid suppression and enteric-coated formulations is a primary point of differentiation. Physician and patient preference plays a significant role.
- Dosing Flexibility and Titration: The ability to effectively titrate dosage based on individual patient needs for fat absorption and symptom relief is critical.
- Cost-Effectiveness: Payers increasingly scrutinize the cost-effectiveness of all PERT options, pushing for lower net prices.
- Clinical Data and Physician Endorsement: Robust clinical data and strong endorsements from key opinion leaders in gastroenterology and cystic fibrosis are vital for gaining physician trust and market share.
- Marketing and Sales Efforts: The marketing and sales strategies employed by VIOKACE's manufacturer to educate healthcare providers and patients about its benefits will directly influence its competitive standing.
The competitive landscape is dynamic, with ongoing innovation in PERT formulations and the looming threat of generic erosion. VIOKACE's sustained market position will depend on its ability to demonstrate clear clinical advantages, navigate payer requirements effectively, and adapt to competitive pricing pressures.
Key Takeaways
- The PERT market is projected to reach $3.6 billion by 2030, driven by increasing diagnoses of pancreatic disorders and improved awareness of PERT benefits.
- VIOKACE's non-enteric coated tablet formulation requires co-administration of acid suppressants, differentiating it from leading enteric-coated PERT products such as Creon, Zenpep, and Pancreaze.
- The intellectual property landscape for VIOKACE, including formulation and method of use patents, will dictate its market exclusivity period before potential generic entry.
- Current WAC for VIOKACE is estimated between $40-$60 daily, with net prices significantly lower post-rebates. Price projections anticipate significant declines post-generic entry.
- Market access challenges include payer formulary restrictions, prior authorization requirements, and physician prescribing habits, compounded by VIOKACE's specific dosing regimen.
- VIOKACE competes in a moderately concentrated market against established branded PERTs, with generic erosion representing a significant future competitive threat.
Frequently Asked Questions
-
When are the key patents for VIOKACE expected to expire, and what is the potential impact of this on its price? Specific patent expiry dates are proprietary and require detailed legal analysis. However, once primary patents and any granted market exclusivities expire, generic VIOKACE is expected to enter the market, leading to a substantial price reduction for the branded product, typically by 50-80% within the initial years of generic availability.
-
What are the primary patient populations for whom VIOKACE is indicated, and are there specific advantages for these groups? VIOKACE is indicated for patients with exocrine pancreatic insufficiency (EPI) due to chronic pancreatitis or pancreatectomy. Potential advantages may include specific enzyme delivery profiles or patient preference for tablet formulation, although the need for acid suppression must be managed.
-
How does the requirement for acid suppression with VIOKACE impact its cost-effectiveness analysis for payers? The cost-effectiveness analysis for payers will consider the added cost of acid-suppressing medications (e.g., PPIs or antacids) and any associated monitoring or potential adverse events related to acid suppression, alongside the direct cost of VIOKACE and its clinical efficacy in managing EPI symptoms compared to alternative PERT options.
-
What is the typical process for a physician to prescribe VIOKACE, including any prior authorization requirements? Physicians typically prescribe VIOKACE by selecting the appropriate dosage strength and quantity based on patient needs. Many insurance plans require prior authorization for PERT products, meaning the physician's office must submit documentation to the insurer detailing the patient's diagnosis, previous treatments, and the rationale for prescribing VIOKACE, which is then reviewed by the insurer.
-
Beyond direct competition, what indirect market forces could influence the future demand and pricing of VIOKACE? Indirect market forces include advancements in diagnostic tools for EPI, development of alternative therapeutic modalities for pancreatic disorders, changes in healthcare policy related to chronic disease management and drug pricing, and evolving patient advocacy for specific treatment preferences or access needs.
Citations
[1] Singh, A., & Ramakrishna, B. S. (2010). Pancreatic enzyme replacement therapy in patients with exocrine pancreatic insufficiency. Digestive Diseases and Sciences, 55(11), 3221–3226.
More… ↓
