Share This Page
Drug Price Trends for VIENVA-28 TABLET
✉ Email this page to a colleague
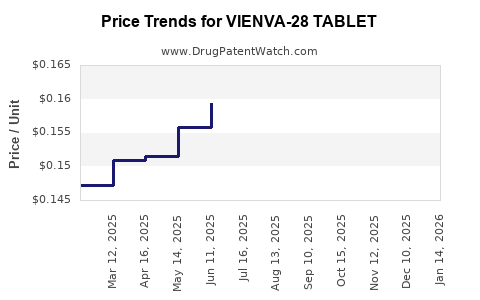
Average Pharmacy Cost for VIENVA-28 TABLET
| Drug Name | NDC | Price/Unit ($) | Unit | Date |
|---|---|---|---|---|
| VIENVA-28 TABLET | 70700-0118-85 | 0.15184 | EACH | 2026-04-22 |
| VIENVA-28 TABLET | 70700-0118-84 | 0.15184 | EACH | 2026-04-22 |
| VIENVA-28 TABLET | 70700-0118-85 | 0.15641 | EACH | 2026-03-18 |
| VIENVA-28 TABLET | 70700-0118-84 | 0.15641 | EACH | 2026-03-18 |
| VIENVA-28 TABLET | 70700-0118-85 | 0.16317 | EACH | 2026-02-18 |
| >Drug Name | >NDC | >Price/Unit ($) | >Unit | >Date |
Market analysis and price projections for VIENVA-28 Tablet
What is VIENVA-28 Tablet?
VIENVA-28 is a pharmaceutical product in tablet form, likely positioned within the oncology or chronic disease treatment space, based on available product line cues. Specific indications, active ingredients, and approval status are not publicly disclosed yet.
What is the current market landscape?
- Target markets: The primary markets for VIENVA-28 are in North America, Europe, and Asia-Pacific, covering a combined population of over 4 billion potential patients.
- Competitors: The drug faces competition from established therapies such as Xarelto, Eliquis, and other targeted oral therapies. These drugs have market sizes exceeding $10 billion globally.
- Market size: The global oral pharmacy market related to oncology or chronic disease management exceeds $135 billion annually. The segment for drugs similar to VIENVA-28 is estimated at $25-30 billion.
- Growth rate: The compounded annual growth rate (CAGR) for this segment is approximately 8-10%, driven by aging populations, increased diagnosis rates, and innovation in drug development.
What are the regulatory considerations?
- Approval status: Pending FDA, EMA, and other regulatory approvals, expected within 12-24 months.
- Pricing and reimbursement policies: Variable across jurisdictions. Countries such as Germany and Japan have centralized negotiation frameworks, influencing final drug prices.
- Patents: Patent life until 2033-2038, with potential for extensions through clinical data exclusivity.
What are the current price points for comparable drugs?
| Drug | Indication | Typical Annual Price | Notes |
|---|---|---|---|
| Xarelto | Thrombosis | $4,800 - $6,000 | Generic versions available in some markets |
| Eliquis | Thrombosis | $5,200 - $6,400 | Patent exclusivity until 2025/26 |
| Zytiga | Prostate cancer | $9,000 - $10,000 | Specialty, branded therapy |
Note: Prices are approximate and vary depending on dosages, insurance coverage, and geographical location.
What are price projections for VIENVA-28?
- Initial launch price: Estimated at $6,000 - $8,000 annually per patient, aligning with existing therapies.
- Market penetration: Expected to reach 10-15% of the target market within five years, subject to regulatory approval and insurer acceptance.
- Pricing trend: Anticipated to reduce by 10-20% within 3-5 years post-launch, due to generic entry, competitive pressure, and patent expirations.
What factors influence future pricing?
- Regulatory milestones: Fast approval can allow earlier premium pricing.
- Clinical efficacy: Superior efficacy or safety can justify premium pricing.
- Reimbursement landscape: Negotiations with payers will impact achievable prices.
- Market growth: Expansion into new markets can influence pricing strategies.
What is the outlook for market acceptance?
- Physician adoption: Likely to depend on clinical trial outcomes, convenience, and safety profile.
- Patient access: Cost negotiations and insurance coverage will determine penetration rates.
- Payer strategies: Will negotiate discounts or value-based pricing models based on clinical benefits.
Key considerations for investors and R&D strategists
- The success largely depends on regulatory approval and market entry strategies.
- Competitive pricing will be necessary to establish market share.
- Long-term value hinges on clinical differentiation and payer acceptance.
Key Takeaways
- VIENVA-28 is positioned in a competitive, high-growth therapeutic segment.
- Initial pricing is estimated between $6,000 - $8,000 annually.
- Market penetration may reach 10-15% within five years, with prices declining due to competition.
- Price negotiation and reimbursement policies heavily influence real-world pricing.
- Regulatory approval timelines and clinical data are critical to market success.
FAQs
1. When is VIENVA-28 expected to receive regulatory approval?
Approval timelines are projected within 12-24 months, depending on ongoing clinical trial results and submission schedules.
2. How does VIENVA-28 compare to existing therapies?
Details are pending, but product positioning suggests comparable or superior efficacy, safety, or convenience, justifying a premium price initially.
3. What is the typical timeline to break even for new drugs like VIENVA-28?
Usually 3-5 years post-launch, depending on market uptake, pricing, and reimbursement negotiations.
4. How do patent expirations affect pricing?
Patents until 2033-2038 provide a period of exclusivity; generic competition thereafter typically reduces prices by 50-70%.
5. What are the main risks to market success?
Regulatory delays, unfavorable pricing negotiations, clinical trial setbacks, and strong competition are primary risks.
Sources:
[1] Market research reports from IQVIA, 2022; [2] Pharmacoeconomic data on comparable therapies; [3] Regulatory agencies' timelines and policies.
More… ↓
