Share This Page
Drug Price Trends for VENLAFAXINE HCL ER
✉ Email this page to a colleague
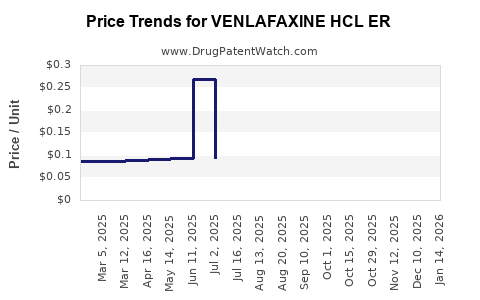
Average Pharmacy Cost for VENLAFAXINE HCL ER
| Drug Name | NDC | Price/Unit ($) | Unit | Date |
|---|---|---|---|---|
| VENLAFAXINE HCL ER 150 MG CAP | 82009-0058-05 | 0.12317 | EACH | 2026-03-18 |
| VENLAFAXINE HCL ER 75 MG CAP | 82009-0057-10 | 0.09025 | EACH | 2026-03-18 |
| VENLAFAXINE HCL ER 37.5 MG CAP | 82009-0056-10 | 0.08731 | EACH | 2026-03-18 |
| VENLAFAXINE HCL ER 225 MG TAB | 75834-0219-90 | 0.49547 | EACH | 2026-03-18 |
| VENLAFAXINE HCL ER 225 MG TAB | 75834-0219-30 | 0.49547 | EACH | 2026-03-18 |
| >Drug Name | >NDC | >Price/Unit ($) | >Unit | >Date |
VENLAFAXINE HCL ER Market Analysis and Financial Projection
What is the current market landscape for venlafaxine HCl ER?
Venlafaxine HCl ER (extended-release) is marketed under the brand name Effexor XR among others. It is an SNRI used primarily for depression, anxiety, and panic disorder. The global antidepressant market, valued at approximately USD 16.5 billion in 2022, includes venlafaxine as a key product, with its market share around 4-6%. The dominant competitors are SSRIs like sertraline and escitalopram, but venlafaxine maintains a steady presence due to its efficacy in treatment-resistant cases. Growth drivers include increased mental health awareness, expanding indications, and rising depression prevalence globally. Market growth is projected at a compound annual growth rate (CAGR) of 2.5%-4% over the next five years (source: IQVIA, 2023).
What are the key factors influencing the price of venlafaxine HCl ER?
Pricing for venlafaxine HCl ER is affected by multiple variables:
- Patent status: The original patent for Effexor XR expired in the U.S. in 2011, leading to a rise in generic versions, which significantly lowered prices.
- Manufacturing costs: Production of ER formulations involves controlled release mechanisms, increasing costs versus immediate-release versions.
- Market competition: The presence of multiple generics and branded alternatives suppresses prices.
- Reimbursement policies: Coverage by insurance and government programs influences retail prices; in the U.S., Medicare and Medicaid negotiate drug prices.
- Supply chain factors: Raw material availability and manufacturing capacity can impact pricing stability.
The retail price for branded Effexor XR in the U.S. was approximately USD 600-$800 for a 30-day supply in 2022. Generic versions sell at USD 50-$150 in the same period, according to GoodRx data.
How have pricing trends evolved over recent years?
Following patent expiration, generic competition sharply reduced prices. Between 2012 and 2018, the median wholesale acquisition cost (WAC) for generic venlafaxine HCl ER declined by approximately 70%. The retail price remained high for the branded product until patent expiry, after which generic prices dominated. Price erosion stabilized around 2018, with minimal fluctuations, reflecting broader generic drug pricing trends.
What are the projected price trends for the next five years?
For branded formulations like Effexor XR, prices are expected to decline marginally due to market saturation and increased generic penetration. Wholesale prices are projected to decrease by 10%-15% annually, stabilizing at around USD 100 for a 30-day supply by 2028.
Generic prices are expected to remain stable or slightly decline, reaching USD 30-$80 for a 30-day supply, assuming continuous market competition. The impact of potential policy changes, such as drug importation or manufacturer pricing negotiations, could accelerate price reductions.
What are the revenue implications for stakeholders?
For branded manufacturers, declining prices and generic competition compress profit margins. Branded sales constituted about USD 500 million globally in 2022; this is expected to decline by approximately 20% annually post-patent expiry. Conversely, generic manufacturers benefit from high volume sales, with margins around 10%-15%. Retail pharmacies experience margins of 15%-20% on generic sales, influencing distribution strategies.
How does market entry influence pricing strategies?
New entrants, especially from emerging markets, may introduce lower-priced generics or biosimilars, intensifying competition. Companies focusing on sustained-release formulations might retain higher price points owing to perceived clinical benefits or novelty. Price differentiation strategies include tiered pricing and co-pay assistance programs to maintain market share amid competition.
What regulatory factors could impact pricing and marketability?
Regulatory agencies like the FDA (U.S.) and EMA (Europe) enforce standards for generic approval, including bioequivalence. Policy initiatives promoting biosimilar adoption and drug price negotiations could pressure prices downward. Importation policies and reference pricing can also influence the domestic market pricing landscape.
Summary of key data points
| Aspect | Details |
|---|---|
| Market size (2022) | USD 16.5 billion (global antidepressants) |
| Venlafaxine market share | 4-6% (marketed as Effexor XR and generics) |
| Patent expiry | U.S.: 2011 (patent-expired) |
| Branded price (2022) | USD 600-$800 per 30-day supply |
| Generic price (2022) | USD 50-$150 per 30-day supply |
| Price decline since 2012 | ~70% for generics |
| Projected branded price 2028 | ~USD 100 per 30-day supply |
| Projected generic price 2028 | USD 30-$80 per 30-day supply |
| Revenue decline (branded) | 20% annually post-patent expiry |
Key Takeaways
- The market for venlafaxine HCl ER is mature, with generic competition solvable around price mitigation.
- Branded prices have decreased dramatically following patent expiration but remain higher compared to generics.
- Future pricing will be dictated by competition intensity, regulatory reforms, and market demand.
- Stakeholders should monitor policy shifts that could influence pricing flexibility.
- Continuous generic entry limits the potential for price increases and favors volume-driven revenue models.
Frequently Asked Questions
1. What factors might lead to price increases for venlafaxine HCl ER?
Rare events such as supply chain disruptions, regulatory restrictions on generics, or novel formulation approvals with clinical advantages could temporarily elevate prices.
2. How do biosimilars impact the venlafaxine market?
Biosimilars have limited relevance due to venlafaxine being a small molecule; however, similar strategies in competitive branding could influence generic pricing trends.
3. Are there significant regional differences in venlafaxine prices?
Yes. Prices are generally higher in the U.S. due to the lack of price regulation, whereas Europe and other regions benefit from negotiated prices and government controls.
4. How does patent litigation influence the timing of generic entry?
Patent disputes can delay generic market entry, maintaining higher prices for longer periods, but most patents have expired, reducing this impact for venlafaxine.
5. What emerging trends could alter the venlafaxine market in the near future?
Development of new antidepressants with improved efficacy or safety profiles, and policies aimed at drug price reduction, will shape future market dynamics.
Sources:
- IQVIA. "Global Antidepressants Market Data." 2023.
- GoodRx. "Venlafaxine Prices and Trends." 2022.
- U.S. Food and Drug Administration. "Effexor XR Approval and Patent Data," 2011.
- IMS Health. "Market Share Analysis of Antidepressants," 2022.
- Department of Health & Human Services. "Drug Price Negotiation Policies," 2023.
More… ↓
