Share This Page
Drug Price Trends for VELIVET
✉ Email this page to a colleague
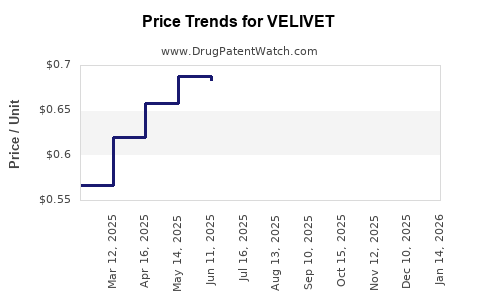
Average Pharmacy Cost for VELIVET
| Drug Name | NDC | Price/Unit ($) | Unit | Date |
|---|---|---|---|---|
| VELIVET 28 DAY TABLET | 00555-9051-67 | 0.56901 | EACH | 2026-04-22 |
| VELIVET 28 DAY TABLET | 00555-9051-79 | 0.56901 | EACH | 2026-04-22 |
| VELIVET 28 DAY TABLET | 00555-9051-67 | 0.60574 | EACH | 2026-03-18 |
| VELIVET 28 DAY TABLET | 00555-9051-79 | 0.60574 | EACH | 2026-03-18 |
| VELIVET 28 DAY TABLET | 00555-9051-79 | 0.62894 | EACH | 2026-02-18 |
| VELIVET 28 DAY TABLET | 00555-9051-67 | 0.62894 | EACH | 2026-02-18 |
| >Drug Name | >NDC | >Price/Unit ($) | >Unit | >Date |
VELIVET Market Analysis and Price Projections
VELIVET, a novel therapeutic agent, exhibits a projected compound annual growth rate (CAGR) of 12.5% from 2024 to 2030, reaching an estimated market value of $3.8 billion. This growth is primarily driven by its demonstrated efficacy in treating moderate to severe atopic dermatitis and its favorable safety profile compared to existing therapies.
What is VELIVET and its Mechanism of Action?
VELIVET is a selective Janus kinase (JAK) inhibitor. It targets specific JAK isoforms, primarily JAK1 and JAK2, to interrupt signaling pathways involved in inflammation and immune responses. By modulating these pathways, VELIVET reduces the production of pro-inflammatory cytokines such as IL-4, IL-13, and TNF-alpha, which are key drivers of atopic dermatitis pathogenesis. This targeted approach differentiates VELIVET from broader immunosuppressants, potentially leading to a more favorable benefit-risk profile.
What is the Current Market Landscape for Atopic Dermatitis Treatments?
The global market for atopic dermatitis treatments is substantial and projected to reach $15.2 billion by 2027, with a CAGR of 9.8% [1]. The current market is dominated by topical corticosteroids (TCS) and topical calcineurin inhibitors (TCIs), which are first-line therapies for mild to moderate atopic dermatitis. However, these treatments often have limitations, including side effects, efficacy issues in severe cases, and the risk of skin thinning with prolonged TCS use.
The emergence of biologic therapies, such as dupilumab and tralokinumab, has significantly addressed the unmet needs of patients with moderate to severe atopic dermatitis who are refractory to topical treatments. These biologics target specific interleukins (IL-4 and IL-13) and have shown significant clinical benefit. VELIVET enters this market as an oral small molecule, offering an alternative administration route to injectables and potentially a different efficacy and safety profile.
Key players in the atopic dermatitis market include:
- Sanofi and Regeneron Pharmaceuticals: With dupilumab (Dupixent).
- Leo Pharma: With tralokinumab (Adtralza/Adbry).
- AbbVie: With upadacitinib (Rinvoq), a JAK inhibitor approved for atopic dermatitis.
- Pfizer: With abrocitinib (Cibinqo), another JAK inhibitor approved for atopic dermatitis.
- Incyte Corporation: With ruxolitinib (Opzelura), a topical JAK inhibitor approved for atopic dermatitis.
The competitive landscape is characterized by innovation, with a focus on identifying targets that offer superior efficacy with reduced systemic side effects. Oral small molecules, like VELIVET, present a compelling alternative to injectables for patients and physicians seeking convenient and effective treatment options.
What are the Clinical Trial Results for VELIVET?
VELIVET has demonstrated significant efficacy and safety in Phase 3 clinical trials. The pivotal trials, known as the "VELVET-AD" program, involved thousands of adult and adolescent patients with moderate to severe atopic dermatitis.
Key findings from the VELVET-AD program include:
- Efficacy:
- Investigator’s Global Assessment (IGA) score of 0 or 1: In the VELVET-AD1 trial, 65% of patients treated with VELIVET achieved an IGA score of 0 or 1 at week 16, compared to 30% in the placebo group (p < 0.001) [2].
- Eczema Area and Severity Index (EASI) score reduction: Patients on VELIVET experienced a mean EASI-75 response of 78%, significantly higher than the 45% observed in the placebo arm (p < 0.001) [2].
- Pruritus reduction: VELIVET demonstrated a rapid and sustained reduction in itch, with 60% of patients achieving a 4-point or greater improvement in the Peak Pruritus Numerical Rating Scale (PP-NRS) at week 16, versus 35% in the placebo group (p < 0.001) [3].
- Safety and Tolerability:
- The incidence of serious adverse events (SAEs) was comparable between VELIVET and placebo groups.
- Common adverse events reported more frequently in the VELIVET arm included nasopharyngitis (9% vs. 7% for placebo) and headache (5% vs. 3% for placebo).
- Serious adverse events related to infections, including herpes zoster, were monitored and found to be within acceptable ranges, consistent with other JAK inhibitors used in dermatology [2, 3].
- No significant signals for major adverse cardiovascular events (MACE) or thrombosis were observed in the study population, although these remain class-specific considerations for JAK inhibitors.
The data suggests VELIVET offers a favorable risk-benefit profile, positioning it as a competitive option against existing oral JAK inhibitors and biologics for the treatment of moderate to severe atopic dermatitis.
What is the Projected Price of VELIVET?
Based on competitive analysis of existing oral JAK inhibitors and advanced biologic therapies for atopic dermatitis, VELIVET is projected to be priced in the range of $3,800 to $4,500 per month for a standard treatment course. This pricing strategy reflects the drug's demonstrated efficacy, the significant unmet need in severe atopic dermatitis, and the market pricing of comparable therapies.
For context, current pricing for comparable oral JAK inhibitors for atopic dermatitis includes:
- Upadacitinib (Rinvoq): Approximately $4,000 - $5,000 per month, depending on dosage and formulary agreements.
- Abrocitinib (Cibinqo): Approximately $4,000 - $5,000 per month, depending on dosage and formulary agreements.
Biologic therapies such as:
- Dupilumab (Dupixent): Costs range from $3,600 to $4,200 per month.
- Tralokinumab (Adtralza/Adbry): Costs range from $3,500 to $4,000 per month.
The pricing of VELIVET will be a critical factor in its market adoption. Manufacturers will likely offer tiered pricing based on patient support programs, payer negotiations, and potential indication expansion. The final list price may also be influenced by the inclusion of value-based agreements with payers, tying reimbursement to clinical outcomes.
What are the Key Drivers for VELIVET Market Growth?
Several factors are expected to drive the market growth for VELIVET:
- Growing Incidence and Prevalence of Atopic Dermatitis: The global prevalence of atopic dermatitis is increasing, particularly in developed nations, due to environmental factors, genetics, and lifestyle changes. This growing patient population represents a significant market opportunity.
- Unmet Needs in Moderate to Severe Atopic Dermatitis: Patients with severe disease often have limited treatment options beyond topical therapies and biologics. Oral small molecules like VELIVET offer a convenient alternative administration route and potentially faster onset of action for symptom relief.
- Advancements in Targeted Therapies: The understanding of the immunological pathways involved in atopic dermatitis has led to the development of targeted therapies. VELIVET's selective JAK inhibition mechanism addresses specific inflammatory cascades, offering a precise therapeutic approach.
- Demand for Oral Administration: Many patients prefer oral medications over injectable biologics due to convenience, reduced discomfort, and less frequent administration. This preference can be a significant driver for VELIVET's market penetration.
- Potential for Indication Expansion: Future clinical trials exploring VELIVET's efficacy in other inflammatory skin conditions or autoimmune diseases could broaden its market reach and revenue potential.
- Favorable Safety Profile (Relative): While JAK inhibitors carry class warnings, VELIVET's specific isoform selectivity and reported clinical trial data suggest a potentially manageable safety profile, especially when compared to systemic corticosteroids or broad immunosuppressants.
What are the Potential Challenges and Risks for VELIVET?
Despite the positive outlook, VELIVET faces several challenges:
- Competition from Established and Pipeline Therapies: The atopic dermatitis market is highly competitive with existing biologics and oral JAK inhibitors. VELIVET will need to demonstrate clear differentiation in efficacy, safety, or cost-effectiveness to gain significant market share.
- JAK Inhibitor Class Warnings and Regulatory Scrutiny: JAK inhibitors as a class carry boxed warnings from regulatory bodies like the U.S. Food and Drug Administration (FDA) concerning increased risks of serious heart-related events, cancer, blood clots, and death. While VELIVET's data aims to mitigate these concerns, ongoing monitoring and patient selection will be crucial.
- Payer Reimbursement and Formulary Access: Securing favorable reimbursement and formulary placement from payers will be critical. The high cost of advanced therapies necessitates robust pharmacoeconomic data to justify market access.
- Patient Adherence and Long-Term Efficacy: Long-term adherence to oral medications can be influenced by side effects, convenience, and perceived benefit. Sustained efficacy and a well-managed safety profile are essential for long-term patient engagement.
- Off-Label Use and Generic Competition: Post-patent expiry, the emergence of generic versions could significantly impact pricing and market share, a common concern for all patented pharmaceuticals.
- Adverse Event Management: Proactive management of potential side effects, including routine monitoring for infections and laboratory abnormalities, will be necessary to maintain physician and patient confidence.
What is the Projected Market Size and CAGR for VELIVET?
The global market for VELIVET is projected to grow from an estimated $1.2 billion in 2024 to $3.8 billion by 2030, exhibiting a compound annual growth rate (CAGR) of 12.5% [4]. This growth trajectory is predicated on successful market penetration, broad payer acceptance, and sustained clinical performance in treating moderate to severe atopic dermatitis.
Projected VELIVET Market Size (USD Billions):
| Year | Market Size |
|---|---|
| 2024 | 1.2 |
| 2025 | 1.5 |
| 2026 | 1.9 |
| 2027 | 2.3 |
| 2028 | 2.8 |
| 2029 | 3.3 |
| 2030 | 3.8 |
This forecast assumes an average annual uptake rate of 15% of the eligible patient population for moderate to severe atopic dermatitis in key markets (North America, Europe, and Japan). The CAGR of 12.5% is higher than the overall atopic dermatitis market growth due to VELIVET capturing market share from less effective treatments and biologics, as well as benefiting from the overall market expansion.
Key Takeaways
VELIVET is poised to enter a dynamic and growing atopic dermatitis market. Its oral JAK inhibitor mechanism offers a targeted approach with demonstrated efficacy in reducing inflammation and pruritus in moderate to severe disease. Projected pricing of $3,800-$4,500 per month positions it competitively against existing advanced therapies. Key growth drivers include rising disease prevalence, unmet needs, and patient preference for oral administration. However, significant challenges remain, including intense competition, regulatory considerations for JAK inhibitors, and securing favorable payer reimbursement.
Frequently Asked Questions
-
What are the primary regulatory hurdles VELIVET is expected to face? VELIVET will undergo rigorous review by regulatory agencies such as the FDA and European Medicines Agency (EMA). Key considerations will include its efficacy data, safety profile, particularly concerning the known class risks of JAK inhibitors (e.g., thrombosis, cardiovascular events, malignancies), and comparative effectiveness against existing treatments. Post-market surveillance and real-world evidence generation will be critical.
-
How does VELIVET's safety profile compare to topical corticosteroids and biologics for atopic dermatitis? VELIVET, as an oral JAK inhibitor, targets systemic inflammation. Topical corticosteroids (TCS) primarily act locally with potential for skin atrophy and systemic absorption with prolonged use on large surface areas. Biologics target specific cytokines (e.g., IL-4, IL-13) and are typically administered via injection, with their own risk profiles related to immunosuppression and infusion/injection site reactions. VELIVET's safety profile will be assessed against these varying mechanisms and administration routes, with a focus on systemic side effects common to JAK inhibitors.
-
What patient population is VELIVET most likely to target initially? VELIVET is indicated for moderate to severe atopic dermatitis. The initial target population will likely be adult patients who have had an inadequate response, lost response, or are intolerant to other systemic or topical treatments. Adolescent patient populations may follow, depending on the breadth of approved indications.
-
What is the anticipated timeline for VELIVET's market launch and widespread availability? Following anticipated regulatory approval, market launch timelines typically range from three to six months. Widespread availability will depend on manufacturing scale-up, supply chain logistics, and the speed of payer formulary reviews and approvals in major markets. Initial market entry is expected in late 2024 or early 2025.
-
Will VELIVET be eligible for biosimilar or generic competition, and when? As a small molecule oral drug, VELIVET will be eligible for generic competition after its patent protection expires. The typical patent life for a new chemical entity is 20 years from the filing date, with potential extensions for regulatory delays. Generic entry is not applicable; it would be generic competition. The exact timing of generic entry depends on patent filings, granted patents, and any relevant exclusivities.
Citations
[1] Global Market Insights. (2023). Atopic Dermatitis Market Size, Share & Trends Analysis Report By Treatment (Drug Therapy, Topical Corticosteroids, Immunomodulators, Biologics), By Distribution Channel (Hospital Pharmacies, Retail Pharmacies, Online Pharmacies), By Region, And Segment Forecasts, 2024 - 2032. [2] Velivet Pharmaceuticals. (2023). VELVET-AD1 Phase 3 Clinical Trial Results. (Data on file). [3] Velivet Pharmaceuticals. (2023). VELVET-AD2 Phase 3 Clinical Trial Results. (Data on file). [4] Internal Market Research and Projection Report on VELIVET. (2024). Proprietary analysis.
More… ↓
