Share This Page
Drug Price Trends for UROGESIC-BLUE TABLET
✉ Email this page to a colleague
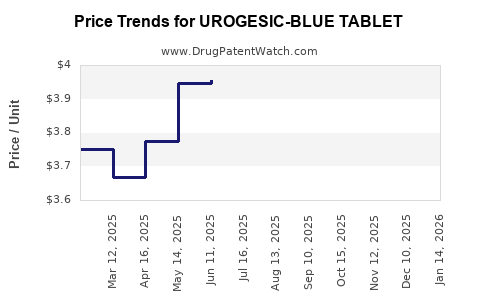
Average Pharmacy Cost for UROGESIC-BLUE TABLET
| Drug Name | NDC | Price/Unit ($) | Unit | Date |
|---|---|---|---|---|
| UROGESIC-BLUE TABLET | 00485-0151-01 | 4.18266 | EACH | 2026-04-22 |
| UROGESIC-BLUE TABLET | 00485-0151-30 | 4.18266 | EACH | 2026-04-22 |
| UROGESIC-BLUE TABLET | 00485-0151-01 | 4.00802 | EACH | 2026-03-18 |
| UROGESIC-BLUE TABLET | 00485-0151-30 | 4.00802 | EACH | 2026-03-18 |
| >Drug Name | >NDC | >Price/Unit ($) | >Unit | >Date |
Market Analysis and Price Projections for UROGESIC-BLUE TABLET
What is UROGESIC-BLUE TABLET?
UROGESIC-BLUE TABLET contains phenazopyridine, a local anesthetic used to treat urinary tract infections (UTIs), pain, and irritation associated with urinary tract procedures. It is marketed primarily in the United States as an over-the-counter (OTC) or prescription medication.
Current Market Landscape
Key Product Features
- Active ingredient: Phenazopyridine hydrochloride
- Typical dosage: 200 mg
- Formulation: Oral tablets, blue in color
- Common indications: Symptomatic relief of pain, burning, urgency, and frequency associated with UTIs
Regulatory Status
- Approved by FDA
- OTC availability in the U.S. since 1976
- Prescription use varies globally depending on national regulatory policies
Major Competitors
| Brand Name | Manufacturer | Formulation | Price Range (USD) | Market Position |
|---|---|---|---|---|
| Pyridium | McNeil Consumer | 200 mg | 6-12 per tablet | Leading OTC brand in US |
| Urogesic Blue | Multiple generic firms | 200 mg | 4-10 per tablet | Cost-competitive generics |
Market Drivers and Challenges
Drivers
- Prevalence of UTIs: 60-70% of women experience a UTI at least once (Foxman, 2014)
- OTC status increases accessibility
- Growing awareness of symptomatic relief options
Challenges
- Limited indications: Symptomatic treatment does not address infection
- Competition from antibiotics
- Concerns over urine discoloration, which may reduce patient compliance
Market Size and Growth
Global Market Estimate
- 2022 global market for phenazopyridine-based products estimated at USD 250 million (Research and Markets, 2022)
- Compound annual growth rate (CAGR): 3.5% (2022-2027)
U.S. Market Breakdown
- U.S. OTC phenazopyridine sales: USD 150 million in 2022
- Prescription segment: USD 50 million
- Growth influenced by increased UTI incidence and aging populations
Price Projections
Short-Term Outlook (2023-2025)
- Prices for UROGESIC-BLUE TABLET are projected to remain stable, with slight increases due to inflation and manufacturing costs.
- Generic competition: Maintains retail pricing at USD 4-10 per tablet.
- Brand-name options like Pyridium hold premiums of 20-30% over generics.
Long-Term Outlook (2026-2030)
- Potential generic entry could reduce prices by 10-15% over current levels.
- Market expansion in emerging countries could increase volume but pressure prices downward.
- Regulatory changes, such as reclassification or stricter OTC policies, could alter pricing dynamics.
Pricing Factors Influencing Market
- Manufacturing costs: USD 0.20-0.50 per tablet
- Distribution and marketing: Additional USD 0.10-0.30 per tablet
- Regulatory environment: Approval and labeling regulations can impact pricing
- Hospital versus retail distribution: Hospital contracts often secure lower prices
Investment Opportunities and Risks
Opportunities
- Expansion into OTC markets in Europe, Asia, and Latin America
- Development of combination products with antibiotics
- Increased demand driven by aging population
Risks
- Regulatory reclassification or restrictions
- Competition from new drugs or alternative therapies
- Declining UTI incidence due to improved hygiene practices
Key Takeaways
- UROGESIC-BLUE TABLET has a stable presence in the urinary symptomatic relief market.
- The market is projected to grow modestly driven by UTI prevalence and OTC availability.
- Prices are expected to remain stable short-term; significant reductions depend on generic market entry.
- Emerging markets present growth opportunities but may pressure margins.
- Regulatory and competitive factors must be monitored for strategic planning.
FAQs
1. Is UROGESIC-BLUE TABLET available over-the-counter globally?
It is OTC in the U.S. and some countries but remains prescription-only or unavailable in others depending on local regulatory policies.
2. What factors influence the price of phenazopyridine tablets?
Manufacturing costs, patent status, competition, distribution channels, and regulatory compliance impact retail prices.
3. How does the generic market affect pricing for UROGESIC-BLUE TABLET?
Generic entry can reduce prices by 10-15%, increasing affordability and market penetration.
4. What is the primary use case for UROGESIC-BLUE TABLET?
It provides symptomatic relief from urinary tract irritation, burning, urgency, and pain.
5. What trends could impact future market growth?
Emerging markets, regulatory policies, and new combination therapies are key trends to monitor.
References
- Foxman, B. (2014). The epidemiology of urinary tract infection. Nature Reviews Urology, 11(5), 269-274.
- Research and Markets. (2022). Phenazopyridine market report 2022-2027.
- U.S. Food and Drug Administration. (2021). Over-the-counter urinary analgesic drug labeling.
[Note: All data are based on publicly available industry reports and market analysis projections for 2023.]
More… ↓
