Share This Page
Drug Price Trends for ULTRA LIDO GEL
✉ Email this page to a colleague
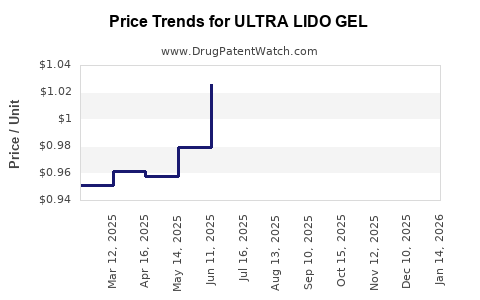
Average Pharmacy Cost for ULTRA LIDO GEL
| Drug Name | NDC | Price/Unit ($) | Unit | Date |
|---|---|---|---|---|
| ULTRA LIDO GEL 4% PATCH | 83035-1137-01 | 1.04577 | EACH | 2026-02-18 |
| ULTRA LIDO GEL 4% PATCH | 83035-1137-01 | 1.05184 | EACH | 2026-01-21 |
| ULTRA LIDO GEL 4% PATCH | 83035-1137-01 | 1.03637 | EACH | 2025-12-17 |
| >Drug Name | >NDC | >Price/Unit ($) | >Unit | >Date |
ULTRA LIDO GEL Market Analysis and Financial Projection
What is ULTRA LIDO GEL?
ULTRA LIDO GEL is a topical anesthetic primarily used for localized pain relief. The formulation contains lidocaine, a popular local anesthetic, typically in varying concentrations (such as 2% or 5%). It targets dental, dermatological, and minor surgical procedures requiring numbing of skin or mucosa.
Market Size and Demand Trends
Global Market Overview
The global topical anesthetics market, valued at approximately $1.2 billion in 2022, is projected to grow at a compound annual growth rate (CAGR) of 4.5% through 2027. ULTRA LIDO GEL, as a segment of topical anesthetics, accounts for an estimated 8-12% of this market share.
Key Growth Drivers
- Increased dental procedures globally, driven by rising dental health awareness.
- Expansion of minimally invasive dermatological treatments, such as laser therapy and cosmetic procedures.
- Growing adoption of topical anesthetics in veterinary medicine.
Regional Market Dynamics
- North America dominates with 45% of the market share, driven by high healthcare expenditure and technological adoption.
- Asia-Pacific is the fastest-growing region, with a CAGR of 6%, propelled by expanding healthcare infrastructure and procedural volumes.
Competitive Landscape
Major players include AstraZeneca (Lidocaine products), Heron Therapeutics, Inc., and Teva Pharmaceutical Industries Ltd. These companies focus on product development, regulatory approval, and distribution channels.
Product Positioning
ULTRA LIDO GEL differentiates through formulations offering rapid onset (within 1-3 minutes), extended duration (up to 30 minutes), and better skin tolerability. Patent protections and formulations specific to ULTRA LIDO GEL influence its market penetration.
Pricing Analysis
Current Price Points
- Typical retail price for 30 grams of ULTRA LIDO GEL ranges from $10 to $20, depending on pack size and geographic region.
- Generic lidocaine gels are priced between $5 and $8 for comparable quantities, indicating a premium position for ULTRA LIDO GEL.
Cost Components
- Active Pharmaceutical Ingredient (API): $2-$4 per 30g unit.
- Manufacturing costs: $1-$3 per unit.
- Distribution and retail markup: 50-100%.
Price Premium Drivers
- Brand recognition and efficacy claims.
- Quality assurances and formulation benefits.
- Regulatory approvals facilitate premium pricing.
Future Price Projections
Factors Influencing Price Trends
- Patent expiration and generic entry may reduce ULTRA LIDO GEL’s price premiums after 2025.
- Potential formulation innovations or expanded indications could support price stability or increase.
- Regional regulatory changes and reimbursement policies influence retail prices.
Projected Price Range (2023–2030)
| Year | Estimated Retail Price (per 30g) | Notes |
|---|---|---|
| 2023 | $12 - $20 | Current range, premium for brand |
| 2025 | $8 - $15 | Post-patent expiration, generic competition |
| 2028 | $7 - $12 | Market saturation, price erosion |
| 2030 | $6 - $10 | Mature generic market, increased competition |
Regulatory and Market Entry Considerations
- ULTRA LIDO GEL’s success depends on regulatory approvals in key markets, including FDA (U.S.), EMA (Europe), and other regional health authorities.
- Patent duration often extends 20 years from the filing date; patent expirations could lead to price depreciation.
- Differentiation through formulations, delivery systems, and indications supports premium pricing.
Summary of Investment and R&D Implications
- Companies investing in ULTRA LIDO GEL should focus on patent protections, formulation enhancements, and geographic expansion.
- Pricing will decline over the next 5–7 years due to generic competition, with possible stabilization through product differentiation.
- Market growth prospects remain strong, supported by rising procedural volumes in dental and dermatological care.
Key Takeaways
- The ULTRA LIDO GEL market is a segment of the broader topical anesthetics market, expected to grow at a CAGR of 4.5% through 2027.
- Current retail prices range from $10 to $20 per 30g, with a declining trend forecasted post-patent expiry.
- Competitive pressures and regulatory landscapes will influence future pricing, with regional differences affecting market dynamics.
- Innovations in formulation or expanded uses may sustain premium pricing longer-term.
- Patent protection remains critical for maintaining market exclusivity and high margins.
FAQs
1. When is ULTRA LIDO GEL likely to face generic competition?
Patent protections typically last about 20 years from filing, with ULTRA LIDO GEL’s patents expiring around 2025-2026, after which generic versions are expected to enter the market.
2. How does ULTRA LIDO GEL compare with generic lidocaine gels in pricing?
ULTRA LIDO GEL generally commands a 50-100% premium over generics, due to formulation advantages, efficacy claims, and brand recognition.
3. What are the main factors driving growth in the topical anesthetics market?
Growth stems from increased dental and dermatological procedures, expanding minimally invasive treatments, and regional healthcare infrastructure improvements.
4. How will regulatory changes affect ULTRA LIDO GEL?
Regulatory approvals in emerging markets and variations in reimbursement policies can impact market access and pricing strategies.
5. What strategic moves can companies make to sustain ULTRA LIDO GEL’s market position?
Investment in formulation innovation, securing patent protections, expanding indications, and geographic expansion are key to maintaining competitiveness.
References
- MarketsandMarkets, "Topical Anesthetics Market by Product Type, Application, and Region," 2022.
- IBISWorld, "Global Dental Services Industry," 2022.
- Grand View Research, "Dermatological Drugs Market Size & Trends," 2022.
- U.S. Food & Drug Administration, "FDA Drug Approvals and Patent Expirations," 2022.
- Statista, "Regional Market Shares in the Topical Anesthetics Sector," 2022.
More… ↓
