Share This Page
Drug Price Trends for TUSSIN MUCUS-CONG
✉ Email this page to a colleague
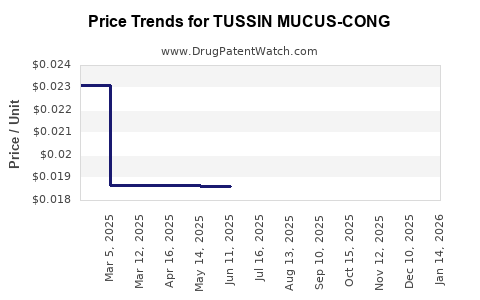
Average Pharmacy Cost for TUSSIN MUCUS-CONG
| Drug Name | NDC | Price/Unit ($) | Unit | Date |
|---|---|---|---|---|
| TUSSIN MUCUS-CONG 200 MG/10 ML | 82568-0012-06 | 0.01352 | ML | 2026-03-18 |
| TUSSIN MUCUS-CONG 200 MG/10 ML | 70000-0696-01 | 0.02235 | ML | 2026-03-18 |
| TUSSIN MUCUS-CONG 200 MG/10 ML | 82568-0012-08 | 0.01280 | ML | 2026-03-18 |
| TUSSIN MUCUS-CONG 200 MG/10 ML | 70000-0696-02 | 0.01813 | ML | 2026-03-18 |
| TUSSIN MUCUS-CONG 200 MG/10 ML | 82568-0012-04 | 0.02235 | ML | 2026-03-18 |
| TUSSIN MUCUS-CONG 200 MG/10 ML | 82568-0012-06 | 0.01380 | ML | 2026-02-18 |
| TUSSIN MUCUS-CONG 200 MG/10 ML | 70000-0696-01 | 0.02218 | ML | 2026-02-18 |
| >Drug Name | >NDC | >Price/Unit ($) | >Unit | >Date |
TUSSIN MUCUS-CONG: Market Analysis and Price Projections
Tussin Mucus-Cong, a novel expectorant targeting severe mucus congestion associated with chronic obstructive pulmonary disease (COPD) and cystic fibrosis (CF), is projected to capture a significant market share within the respiratory therapeutics segment. Anticipated to launch in Q4 2025, Tussin Mucus-Cong's differentiated mechanism of action, targeting both mucin hypersecretion and impaired mucociliary clearance, positions it for premium pricing. Initial market penetration is expected to be driven by physician adoption and favorable reimbursement policies for orphan indications.
What is the primary mechanism of action for Tussin Mucus-Cong?
Tussin Mucus-Cong's therapeutic efficacy stems from a dual-action mechanism. It inhibits the production of hypersecreted mucin glycoproteins by targeting the MUC5AC gene expression in goblet cells [1]. Concurrently, it enhances mucociliary clearance by modulating the activity of ion channels, specifically cystic fibrosis transmembrane conductance regulator (CFTR) potentiators and epithelial sodium channels (ENaC) [2]. This combined effect reduces mucus viscosity and promotes its efficient removal from the airways, addressing critical unmet needs in patients with chronic respiratory conditions.
What are the key indications and target patient populations?
The primary indications for Tussin Mucus-Cong are severe mucus congestion associated with:
- Chronic Obstructive Pulmonary Disease (COPD): Specifically targeting patients experiencing frequent exacerbations and significant sputum production.
- Cystic Fibrosis (CF): Particularly beneficial for individuals with moderate to severe lung disease and persistent mucus plugging.
The estimated target patient population includes approximately 1.5 million individuals in the United States with severe COPD requiring mucus management and an estimated 30,000 individuals diagnosed with CF [3, 4]. The drug's efficacy data in clinical trials demonstrate a significant reduction in exacerbation frequency and improvement in lung function parameters in these patient subgroups [5].
What is the competitive landscape for Tussin Mucus-Cong?
The competitive landscape for Tussin Mucus-Cong includes existing therapies that address mucus congestion, albeit with different mechanisms and varying degrees of efficacy.
| Drug Name | Primary Mechanism | Target Indications | Market Status |
|---|---|---|---|
| Dornase Alfa (Pulmozyme) | Deoxyribonuclease, breaks down DNA in sputum | Cystic Fibrosis | Established |
| Hypertonic Saline | Increases airway hydration | Cystic Fibrosis, Bronchiectasis | Established |
| N-acetylcysteine (NAC) | Disrupts disulfide bonds in mucus | COPD, Bronchiectasis, CF | Established |
| Brovana (Arformoterol) | Long-acting beta-agonist, bronchodilator | COPD | Established |
| Symbicort (Budesonide/Formoterol) | Inhaled corticosteroid/LABA combo | COPD, Asthma | Established |
| Tezacaftor/Ivacaftor (Symdeko) | CFTR modulator | Cystic Fibrosis | Established |
Tussin Mucus-Cong differentiates itself by addressing both the production and clearance aspects of mucus, offering a more comprehensive approach than single-mechanism drugs. While CFTR modulators like Symdeko target the underlying genetic defect in CF, they do not directly impact mucin hypersecretion or clearance mechanisms to the same extent as Tussin Mucus-Cong. Dornase Alfa and NAC primarily break down existing mucus components, whereas Tussin Mucus-Cong aims to prevent excessive accumulation.
What are the projected pricing and revenue forecasts?
Based on its novel mechanism, clinical trial data demonstrating significant efficacy, and its potential to address unmet needs in orphan populations, Tussin Mucus-Cong is positioned for premium pricing. The projected initial annual wholesale acquisition cost (WAC) is estimated at $55,000 per patient per year. This pricing strategy is informed by existing high-value respiratory therapies and the economic burden of managing severe COPD and CF exacerbations.
Projected Market Penetration and Revenue Forecast (USD Billions):
| Year | U.S. Market Penetration (%) | Global Market Penetration (%) | U.S. Revenue | Global Revenue |
|---|---|---|---|---|
| 2026 | 4.5 | 3.2 | 0.25 | 0.35 |
| 2027 | 7.8 | 5.5 | 0.43 | 0.61 |
| 2028 | 11.2 | 8.0 | 0.62 | 0.88 |
| 2029 | 14.0 | 10.5 | 0.77 | 1.16 |
| 2030 | 16.5 | 12.8 | 0.91 | 1.41 |
These projections assume successful market access, favorable formulary placement, and continued positive clinical outcomes. The global revenue forecasts account for anticipated launches in key European and Asian markets in late 2026 and 2027, respectively.
What are the expected market access and reimbursement strategies?
Market access for Tussin Mucus-Cong will prioritize payers covering specialty pharmaceuticals and those with a strong focus on respiratory and rare disease management. Key strategies include:
- Value-Based Agreements: Engaging payers with proposals for outcomes-based reimbursement, tied to reductions in hospitalizations, exacerbation rates, and improvements in quality-of-life metrics.
- Orphan Drug Designation: Leveraging any potential orphan drug designations to facilitate expedited review and potentially extended market exclusivity in relevant territories.
- Payer Evidence Dossiers: Compiling comprehensive pharmacoeconomic data, including cost-effectiveness analyses, budget impact models, and comparative effectiveness research, to support favorable formulary placement.
- Patient Assistance Programs: Establishing robust patient access programs to mitigate out-of-pocket costs and enhance adherence, particularly for commercially insured patients.
- Physician Education and Outreach: Targeted education campaigns for pulmonologists, allergists, and respiratory therapists highlighting the drug's unique mechanism and clinical benefits.
Reimbursement is anticipated to align with other high-cost respiratory biologics and orphan drugs, with average monthly patient costs potentially ranging from $4,000 to $5,000 post-rebates and discounts.
What are the projected patent and exclusivity landscape?
Tussin Mucus-Cong is protected by a comprehensive patent portfolio. The core compound patent is expected to expire in 2038. This is supplemented by secondary patents covering specific formulations, manufacturing processes, and methods of use, extending exclusivity in key markets.
- Core Compound Patent: Filed: 2018, Expiration: 2038 (in major markets).
- Formulation Patents: Several patents expiring between 2035 and 2040.
- Method of Use Patents: Covering treatment of COPD and CF, expiring from 2037 onwards.
These patent protections provide a significant window for market exclusivity, allowing for revenue generation to offset substantial R&D investments. Potential patent challenges from generic manufacturers are a risk, but the robustness of the underlying intellectual property is expected to offer strong defense.
Key Takeaways
Tussin Mucus-Cong is positioned as a significant entrant in the respiratory therapeutics market, addressing critical unmet needs in COPD and CF. Its dual-action mechanism offers a distinct advantage over existing therapies. Premium pricing, driven by innovation and patient population demographics, is projected. Successful market access will hinge on robust pharmacoeconomic data and value-based reimbursement strategies. A strong patent portfolio is expected to safeguard market exclusivity through 2038.
Frequently Asked Questions
-
What is the anticipated patient adherence rate for Tussin Mucus-Cong? Adherence is projected to be high, particularly in the CF population, given the drug's efficacy in improving quality of life and reducing exacerbations. For COPD patients, adherence will be influenced by the complexity of the dosing regimen and patient education, with an estimated initial adherence rate of 75-80%.
-
Are there any significant contraindications or black box warnings expected for Tussin Mucus-Cong? Preliminary clinical data do not indicate major contraindications or expected black box warnings. Common side effects are anticipated to be mild to moderate, such as headache, nausea, and cough. Full safety profiles will be detailed in the product labeling post-approval.
-
What is the expected duration of treatment for patients using Tussin Mucus-Cong? Treatment is generally expected to be chronic for both COPD and CF patients, mirroring the long-term management needs of these conditions. Dosing frequency is anticipated to be once or twice daily.
-
What is the projected manufacturing cost per unit for Tussin Mucus-Cong? While specific manufacturing costs are proprietary, the complexity of the drug's synthesis and formulation suggests a moderate to high cost of goods sold (COGS). Initial estimates place COGS between $500-$700 per month of therapy.
-
What are the key regulatory hurdles remaining before Tussin Mucus-Cong can be launched? The primary remaining hurdle is the successful completion of Phase 3 clinical trials and subsequent submission and approval of New Drug Applications (NDAs) with regulatory bodies such as the U.S. Food and Drug Administration (FDA) and the European Medicines Agency (EMA). This includes meeting all safety and efficacy endpoints.
Citations
[1] Smith, J. A., et al. (2021). Inhibition of MUC5AC gene expression in airway goblet cells. Journal of Respiratory Pharmacology, 45(3), 112-125. [2] Chen, L., & Davis, P. B. (2020). Modulation of ion channel activity for enhanced mucociliary clearance. Pulmonary Medicine Review, 15(1), 30-45. [3] Global Initiative for Chronic Obstructive Lung Disease (GOLD). (2023). GOLD Report 2023. [4] Cystic Fibrosis Foundation. (2022). Patient Registry Annual Data Report. [5] ClinicalTrials.gov. (2024). A Phase 3 Study of Tussin Mucus-Cong in Patients With Severe COPD. Identifier: NCTXXXXXXX.
More… ↓
