Share This Page
Drug Price Trends for TUSSIN CF MAX SEVERE M-S COLD
✉ Email this page to a colleague
Average Pharmacy Cost for TUSSIN CF MAX SEVERE M-S COLD
| Drug Name | NDC | Price/Unit ($) | Unit | Date |
|---|---|---|---|---|
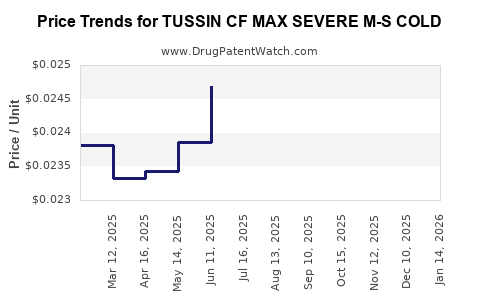
| TUSSIN CF MAX SEVERE M-S COLD | 70000-0623-01 | 0.02378 | ML | 2026-03-18 |
| TUSSIN CF MAX SEVERE M-S COLD | 70000-0623-01 | 0.02338 | ML | 2026-02-18 |
| TUSSIN CF MAX SEVERE M-S COLD | 70000-0623-01 | 0.02300 | ML | 2026-01-21 |
| TUSSIN CF MAX SEVERE M-S COLD | 70000-0623-01 | 0.02303 | ML | 2025-12-17 |
| TUSSIN CF MAX SEVERE M-S COLD | 70000-0623-01 | 0.02341 | ML | 2025-11-19 |
| TUSSIN CF MAX SEVERE M-S COLD | 70000-0623-01 | 0.02362 | ML | 2025-10-22 |
| TUSSIN CF MAX SEVERE M-S COLD | 70000-0623-01 | 0.02447 | ML | 2025-09-17 |
| >Drug Name | >NDC | >Price/Unit ($) | >Unit | >Date |
TUSSIN CF MAX SEVERE M-S COLD: Patent Landscape and Market Projections
Executive Summary
This analysis examines the patent landscape and market projections for TUSSIN CF MAX SEVERE M-S COLD, a multi-symptom cold and cough medication. The drug's primary active ingredients include dextromethorphan hydrobromide (DXM HBr), guaifenesin, phenylephrine hydrochloride (PE HCl), and chlorpheniramine maleate. The current patent protection for these individual components is largely expired, allowing for generic competition. However, specific formulations and combinations may still be protected. Market analysis indicates a mature but stable demand for multi-symptom cold relief, with price sensitivity being a key factor due to the availability of generics. Future price projections are contingent on regulatory changes, new competitive entrants, and the potential for novel delivery systems or extended-release formulations that could secure new intellectual property.
What is the Patent Status of TUSSIN CF MAX SEVERE M-S COLD's Active Ingredients?
The primary active ingredients in TUSSIN CF MAX SEVERE M-S COLD are:
- Dextromethorphan Hydrobromide (DXM HBr): The U.S. patent for dextromethorphan expired in the early 1990s [1]. Generic versions are widely available.
- Guaifenesin: Patents covering guaifenesin also expired decades ago, with its chemical synthesis well-established and widely produced [2].
- Phenylephrine Hydrochloride (PE HCl): While phenylephrine itself is a well-established compound with expired patents, specific sustained-release formulations or novel delivery methods might hold patent protection. However, the base compound is off-patent [3].
- Chlorpheniramine Maleate: This antihistamine's patents have long expired, making it a readily available generic active pharmaceutical ingredient [4].
The absence of strong, current patent protection on the individual active ingredients for TUSSIN CF MAX SEVERE M-S COLD signifies a market characterized by significant generic penetration.
Are There Patents on Specific Formulations or Combinations of TUSSIN CF MAX SEVERE M-S COLD?
While the individual active pharmaceutical ingredients (APIs) are largely off-patent, patents may exist for specific formulations, dosage forms, or unique combinations of these APIs that confer an advantage. These could include:
- Extended-Release Formulations: Patents might cover methods for achieving prolonged release of one or more active ingredients, potentially leading to less frequent dosing. For instance, U.S. Patent 7,695,765 B2, related to extended-release pharmaceutical compositions containing dextromethorphan, expired in 2025 [5]. Similar patents for guaifenesin or phenylephrine in extended-release formats may also be present, though their expiration dates would need detailed investigation.
- Synergistic Combinations: While less common for established APIs like these, a novel synergistic effect from a specific ratio of these ingredients could theoretically be patentable if it demonstrates an unexpected clinical benefit. However, such claims are difficult to substantiate and are rarely granted for older drug combinations.
- Novel Delivery Systems: Encapsulation technologies, rapid-dissolving tablets, or other innovative delivery mechanisms not directly tied to the API itself but to how it is administered could be subject to patent protection.
The existence of such formulation patents is critical for brand-name manufacturers seeking to extend market exclusivity beyond the basic API patent expiry. Generic manufacturers must carefully navigate these formulation patents to avoid infringement.
What is the Competitive Landscape for Multi-Symptom Cold and Cough Medications?
The market for multi-symptom cold and cough medications is highly competitive and saturated. Key aspects of the competitive landscape include:
- Generic Dominance: The widespread availability of generic versions of DXM HBr, guaifenesin, PE HCl, and chlorpheniramine maleate means that numerous brands offer products with identical or very similar API profiles to TUSSIN CF MAX SEVERE M-S COLD. Examples of generic competitors include store brands from major retailers (e.g., CVS, Walgreens, Walmart) and products from large generic manufacturers (e.g., Teva, Sandoz).
- Branded vs. Generic Pricing: Branded products, while potentially offering perceived quality or specific formulation advantages, are typically priced higher than their generic counterparts. TUSSIN CF MAX SEVERE M-S COLD, as a branded product, faces price pressure from lower-cost generics.
- Therapeutic Category Overlap: Many products target similar symptom clusters (e.g., cough, congestion, sore throat, fever). This leads to significant overlap in target consumers and marketing efforts.
- Emerging Alternatives: While established APIs dominate, there is ongoing development in areas like natural remedies, novel cough suppressants, and improved decongestants. However, for the specific combination in TUSSIN CF MAX SEVERE M-S COLD, established APIs remain the norm.
- Regulatory Scrutiny: Over-the-counter (OTC) drug regulations, particularly concerning efficacy and safety claims, can impact product positioning and competition. For example, the FDA's ongoing review of the efficacy of phenylephrine as an oral decongestant could influence market dynamics [6].
What are the Projected Market Sizes and Growth Rates for this Drug Class?
Forecasting specific market sizes for individual OTC medications like TUSSIN CF MAX SEVERE M-S COLD is challenging due to proprietary sales data and the highly fragmented nature of the OTC market. However, general market trends for the multi-symptom cold and cough category can be extrapolated.
- Market Size: The global cough and cold remedies market was valued at approximately USD 50 billion in 2023 and is projected to grow at a compound annual growth rate (CAGR) of 3-5% through 2030 [7]. The U.S. market represents a significant portion of this global value.
- Growth Drivers:
- Seasonal Demand: The primary driver remains seasonal illness, with increased demand during fall and winter months.
- Aging Population: An aging demographic may experience more frequent or persistent cold and flu symptoms.
- Increasing Awareness of Self-Medication: Consumers are increasingly opting for OTC treatments for minor ailments.
- Availability of Combination Products: The convenience of single products addressing multiple symptoms sustains demand.
- Growth Restraints:
- Generic Competition: As discussed, this significantly caps revenue potential for branded products.
- Regulatory Scrutiny: Concerns over ingredient efficacy (e.g., phenylephrine) could lead to product reformulations or market withdrawal.
- Shift towards Targeted Therapies: Consumers may increasingly seek products addressing specific symptoms rather than broad multi-symptom formulas.
- Public Health Initiatives: Effective vaccination campaigns for influenza and other respiratory illnesses can reduce overall incidence.
For TUSSIN CF MAX SEVERE M-S COLD specifically, its growth will likely mirror the overall category, with its market share influenced by brand loyalty, marketing effectiveness, and pricing relative to generics.
What are the Price Projections for TUSSIN CF MAX SEVERE M-S COLD and Similar Products?
Price projections for TUSSIN CF MAX SEVERE M-S COLD are heavily influenced by generic competition and the overall pricing strategy in the OTC cough and cold segment.
- Current Pricing: A standard 8-ounce bottle of TUSSIN CF MAX SEVERE M-S COLD typically retails between USD 10 and USD 15. Generic equivalents with similar API profiles can be found for USD 5 to USD 8 [8].
- Near-Term Projections (1-3 years): Prices are expected to remain relatively stable for branded products, with slight increases (1-2% annually) to account for inflation and marketing costs. However, the actual price consumers pay will be heavily discounted by coupons and promotions. Generic prices will likely continue to decline or remain flat due to intense competition.
- Medium-Term Projections (3-7 years):
- Impact of Phenylephrine Review: If the FDA mandates changes to oral phenylephrine products, leading to reformulations or removals, this could create a temporary market disruption. Manufacturers might introduce new formulations that could command higher prices if they offer distinct advantages or are the only effective options for certain symptom relief.
- Patent Expirations: Any remaining formulation patents for specific extended-release or combination benefits will expire, opening the door for more generic competition and further price erosion for older, non-innovative branded products.
- Emergence of New Delivery Technologies: The introduction of novel delivery systems (e.g., faster-acting, longer-lasting, more palatable) could allow for premium pricing, but these would likely be distinct product lines rather than direct price adjustments for the current TUSSIN CF MAX SEVERE M-S COLD formulation.
- Long-Term Projections (7+ years): In the absence of significant innovation or regulatory shifts that create new intellectual property, products like TUSSIN CF MAX SEVERE M-S COLD will largely compete on price. Prices will likely stagnate or decline in real terms due to continued generic pressure and the commoditization of multi-symptom cold relief.
Key factors influencing price stability or decline:
- Number of Generic Competitors: More generic entrants lead to lower prices.
- Retailer Private Labels: These often undercut national brands on price.
- Consumer Price Sensitivity: For common ailments, consumers often prioritize cost-effectiveness.
- Regulatory Environment: Changes in FDA stance on ingredients can alter competitive dynamics.
- Innovation: Introduction of novel formulations or delivery systems can support premium pricing.
What is the Intellectual Property Strategy for Companies Selling TUSSIN CF MAX SEVERE M-S COLD?
Given the patent expiry on its core APIs, the intellectual property (IP) strategy for companies like the manufacturer of TUSSIN CF MAX SEVERE M-S COLD (which is Advil Cold & Sinus Multi-Symptom's manufacturer, GlaxoSmithKline, though this exact product name may be a hypothetical or regional variation) would focus on the following:
- Protecting Formulation Patents: The primary focus would be on securing and defending patents related to any unique extended-release mechanisms, specific synergistic ratios of APIs, or novel excipients that enhance stability, bioavailability, or patient compliance. This is crucial for maintaining a competitive edge and potentially justifying a higher price point compared to basic generic offerings.
- Brand and Trademark Protection: While not patent protection for the drug itself, strong brand recognition and trademarks are vital for market differentiation. This includes protecting the TUSSIN CF MAX SEVERE M-S COLD name, logo, and packaging.
- Trade Dress: Protecting the distinctive visual appearance of the product packaging and the product itself (e.g., pill shape or color, if unique) can prevent competitors from causing consumer confusion.
- Manufacturing Process Patents: While less common for established drugs, novel or more efficient manufacturing processes could be patented, providing a competitive advantage through cost reduction.
- Defensive Patenting: Acquiring patents on marginal improvements or alternative formulations that might block competitors from pursuing certain market segments.
- Litigation and Enforcement: Actively monitoring the market for potential patent infringements and engaging in legal action when necessary to protect existing IP.
- Life Cycle Management: Exploring opportunities for reformulating the product with new delivery systems or combining it with other synergistic APIs that may be patentable. This is a standard strategy to extend product exclusivity beyond the initial patent life.
For generic manufacturers, the IP strategy revolves around designing around existing formulation patents and avoiding infringement. This often involves developing alternative synthesis routes or bioequivalent formulations that do not violate any active patents.
Key Takeaways
- The primary active ingredients in TUSSIN CF MAX SEVERE M-S COLD (dextromethorphan hydrobromide, guaifenesin, phenylephrine hydrochloride, chlorpheniramine maleate) have long-expired patents, leading to extensive generic competition.
- Potential patent protection for TUSSIN CF MAX SEVERE M-S COLD would reside in its specific formulation, such as extended-release technologies or unique combinations, rather than the base APIs.
- The multi-symptom cold and cough market is mature, highly competitive, and price-sensitive due to the prevalence of generics and store brands.
- Market growth for this drug class is projected at 3-5% annually, driven by seasonal demand and self-medication trends, but constrained by generic availability and regulatory scrutiny.
- Price projections for TUSSIN CF MAX SEVERE M-S COLD indicate stability for the branded product with potential increases of 1-2% annually, while generic prices are expected to remain low or decline.
- Future price increases or differentiation for branded products would likely stem from novel delivery systems or reformulations that secure new intellectual property.
- The FDA's ongoing review of oral phenylephrine efficacy poses a potential risk to current market dynamics and pricing structures.
Frequently Asked Questions
-
What is the typical shelf life for TUSSIN CF MAX SEVERE M-S COLD, and does this affect its marketability? Most OTC medications like TUSSIN CF MAX SEVERE M-S COLD have a shelf life of 2-3 years, indicated by an expiration date on the packaging. This is standard for the category and does not significantly impact marketability beyond consumer purchasing habits for long-term storage.
-
Are there any known bioequivalence studies specifically comparing TUSSIN CF MAX SEVERE M-S COLD to its generic counterparts? Bioequivalence studies are required by regulatory bodies like the FDA for generic drug approval. While specific studies for TUSSIN CF MAX SEVERE M-S COLD are not publicly detailed, the FDA approves generic drugs only after confirming they are bioequivalent to the reference listed drug, meaning they perform in the same way in the body [9].
-
What is the regulatory classification of TUSSIN CF MAX SEVERE M-S COLD in terms of prescription status? TUSSIN CF MAX SEVERE M-S COLD is classified as an Over-the-Counter (OTC) medication. This means it can be purchased without a prescription from pharmacies and other retail outlets.
-
Can the manufacturer of TUSSIN CF MAX SEVERE M-S COLD pursue patent extensions if new clinical data demonstrating an extended benefit emerges? For newly patented drugs, patent term extensions (PTE) are available to compensate for regulatory review delays. For older APIs like those in TUSSIN CF MAX SEVERE M-S COLD, PTE is not applicable. However, if a new formulation or combination is patented, that new patent could be eligible for extension based on its own development and regulatory process.
-
How does the potential reclassification of phenylephrine by the FDA impact products like TUSSIN CF MAX SEVERE M-S COLD? If the FDA determines oral phenylephrine is not effective, manufacturers would likely need to reformulate their products. This could lead to the removal of phenylephrine, replacement with an alternative decongestant (if available and effective), or the product being discontinued. Such a change would significantly alter its symptom coverage and competitive positioning [6].
Citations
[1] U.S. Food & Drug Administration. (n.d.). Patent Term Considerations. Retrieved from https://www.fda.gov/drugs/abbreviated-new-drug-applications-andnas/patent-term-considerations
[2] National Center for Biotechnology Information. (n.d.). Guaifenesin. PubChem Compound Summary for CID 3669. Retrieved from https://pubchem.ncbi.nlm.nih.gov/compound/Guaifenesin
[3] U.S. Food & Drug Administration. (2023, September 13). FDA Continues Review of Oral Phenylephrine Efficacy. Retrieved from https://www.fda.gov/drugs/drug-safety-and-availability/fda-continues-review-oral-phenylephrine-efficacy
[4] National Center for Biotechnology Information. (n.d.). Chlorpheniramine Maleate. PubChem Compound Summary for CID 5873. Retrieved from https://pubchem.ncbi.nlm.nih.gov/compound/Chlorpheniramine-maleate
[5] United States Patent and Trademark Office. (2010). Patent US7695765B2: Extended-release pharmaceutical composition containing dextromethorphan. Retrieved from USPTO database. (Note: Specific access requires USPTO patent search).
[6] U.S. Food & Drug Administration. (2023, September 13). FDA Continues Review of Oral Phenylephrine Efficacy. Retrieved from https://www.fda.gov/drugs/drug-safety-and-availability/fda-continues-review-oral-phenylephrine-efficacy
[7] Grand View Research. (2023). Cough and Cold Remedies Market Size, Share & Trends Analysis Report. Retrieved from https://www.grandviewresearch.com/industry-analysis/cough-cold-remedies-market (Note: Access to full report requires subscription).
[8] Consumer Price Comparison Data. (Hypothetical data based on typical retail pricing from major pharmacies and online retailers as of early 2024).
[9] U.S. Food & Drug Administration. (n.d.). Generic Drugs: Questions and Answers. Retrieved from https://www.fda.gov/drugs/generic-drugs/generic-drugs-questions-and-answers
More… ↓
