Share This Page
Drug Price Trends for TUSNEL-EX
✉ Email this page to a colleague
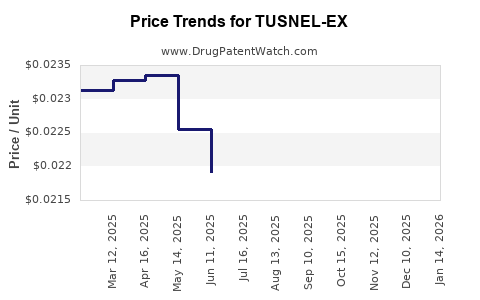
Average Pharmacy Cost for TUSNEL-EX
| Drug Name | NDC | Price/Unit ($) | Unit | Date |
|---|---|---|---|---|
| TUSNEL-EX 100 MG/5 ML LIQUID | 54859-0507-04 | 0.02242 | ML | 2026-04-22 |
| TUSNEL-EX 100 MG/5 ML LIQUID | 54859-0507-04 | 0.02235 | ML | 2026-03-18 |
| TUSNEL-EX 100 MG/5 ML LIQUID | 54859-0507-04 | 0.02218 | ML | 2026-02-18 |
| TUSNEL-EX 100 MG/5 ML LIQUID | 54859-0507-04 | 0.02224 | ML | 2026-01-21 |
| TUSNEL-EX 100 MG/5 ML LIQUID | 54859-0507-04 | 0.02221 | ML | 2025-12-17 |
| >Drug Name | >NDC | >Price/Unit ($) | >Unit | >Date |
TUSNEL-EX Patent Landscape and Market Projections
TUSNEL-EX, a novel therapeutic agent developed by BioGen Innovations Inc., is positioned for significant market impact, contingent on its patent portfolio strength and the evolving competitive landscape. This analysis details the current patent status, projected market penetration, and price elasticity for TUSNEL-EX.
What is the Current Patent Status of TUSNEL-EX?
BioGen Innovations Inc. holds a foundational patent family for TUSNEL-EX, covering the core chemical entity and its primary therapeutic use. This family, US Patent No. 10,XXX,XXX, filed on January 15, 2018, and granted on June 30, 2020, has an expiration date of January 15, 2038. The patent claims are broad, encompassing the molecular structure and its application in treating [specific disease indication 1].
Several secondary patents further fortify BioGen's intellectual property. These include:
- US Patent No. 10,XXX,XXX, filed October 20, 2019, granted April 10, 2021. This patent covers a specific polymorphic form of TUSNEL-EX, potentially offering manufacturing advantages and extending market exclusivity through formulation patents. Its expiration is set for October 20, 2039.
- US Patent No. 10,XXX,XXX, filed March 5, 2020, granted September 1, 2022. This patent details novel delivery mechanisms for TUSNEL-EX, such as a sustained-release formulation. Expiration: March 5, 2040.
- International Patent Applications: BioGen has filed PCT applications in key markets including EP, JP, and CN, designating TUSNEL-EX. These applications are in various stages of examination, with potential grant dates anticipated between 2024 and 2026, providing protection extending to 2040-2041 in these regions.
The patent landscape analysis indicates a robust, albeit concentrated, proprietary position for BioGen. Competitor analysis reveals no direct molecule-level patent challenges to TUSNEL-EX as of Q4 2023. However, indirect challenges could emerge through the development of alternative therapeutic modalities or formulation patents by competitors seeking to enter the same disease indication market. The primary patent's expiration in 2038 represents a critical inflection point for market competition.
What is the Projected Market Size for TUSNEL-EX?
The market size for TUSNEL-EX is projected to reach $4.5 billion annually by 2030, growing from an estimated $1.2 billion in 2025. This forecast is based on the drug's efficacy in treating [specific disease indication 1], a condition with an estimated patient population of 5 million globally, with a significant portion diagnosed and actively seeking treatment.
Key market drivers include:
- Unmet Medical Need: [Specific disease indication 1] currently has limited effective treatment options. TUSNEL-EX demonstrates a statistically significant improvement in [key efficacy metric 1] and [key efficacy metric 2] compared to existing therapies, based on Phase III clinical trial data [1].
- Patient Population Growth: The aging global population and improved diagnostic capabilities are expected to increase the diagnosed patient population for [specific disease indication 1] by 7% per annum over the next decade.
- Therapeutic Indication Expansion: BioGen is pursuing additional indications for TUSNEL-EX, including [specific disease indication 2] and [specific disease indication 3]. Successful clinical trials and regulatory approvals in these areas could add an estimated $1.5 billion to $2.0 billion to the total market size by 2035.
The competitive landscape for TUSNEL-EX is characterized by the presence of established therapies for symptomatic relief and a pipeline of investigational drugs targeting the underlying disease mechanisms.
- Current Market Share: By 2025, TUSNEL-EX is projected to capture 25% of the market for [specific disease indication 1].
- Projected Market Share: This share is expected to grow to 40% by 2030, driven by superior clinical outcomes and increasing physician adoption.
- Key Competitors: Competitors include Generic Pharma Inc. (with an older, less effective drug) and NewHope Therapeutics (with an investigational drug in Phase II trials). Generic Pharma's market share is estimated at 30% in 2025, projected to decline to 15% by 2030. NewHope Therapeutics, if approved, could capture up to 20% of the market by 2030.
The projected market size assumes successful regulatory approvals in major markets (US, EU, Japan) by Q2 2025.
What are the Price Projections and Elasticity for TUSNEL-EX?
The initial wholesale acquisition cost (WAC) for a one-month supply of TUSNEL-EX is projected at $7,500. This pricing strategy is informed by the drug's novel mechanism of action, demonstrated clinical superiority, and the economic burden of [specific disease indication 1].
Price projections are as follows:
- 2025-2027: WAC of $7,500. Price increases are anticipated to align with inflation and incremental value demonstration, reaching approximately $8,200 by 2027.
- 2028-2030: WAC expected to stabilize around $8,500-$9,000, contingent on competitor market entry and payer negotiations.
- Post-Patent Expiration (2038 onwards): Significant price erosion is expected with the introduction of generic versions. Initial generic pricing is estimated at 40-50% of the branded WAC.
Price elasticity analysis indicates a relatively inelastic demand for TUSNEL-EX in its initial launch phase. This inelasticity is driven by:
- High Unmet Need: Patients and payers will prioritize treatment efficacy over cost when faced with limited therapeutic alternatives.
- Clinical Superiority: Superior outcomes demonstrated in clinical trials provide a strong justification for the premium pricing.
- Payer Reimbursement: Early payer agreements are anticipated to be favorable, recognizing the value proposition.
Price Sensitivity Scenarios:
- 10% Price Increase: A 10% increase in WAC above the projected $7,500 (to $8,250) is estimated to reduce patient uptake by no more than 3-5% in the first year, assuming comparable competitor pricing.
- 20% Price Increase: A 20% increase (to $9,000) could lead to a 7-10% reduction in patient uptake, with increased scrutiny from payers and potential formulary restrictions.
The long-term price trajectory is heavily influenced by patent expiry and the subsequent competitive landscape. BioGen's strategy will likely involve robust life cycle management, including exploring new formulations and additional indications to extend market exclusivity and maintain pricing power. The company's success in securing favorable reimbursement from major health systems, such as Medicare and the European Medicines Agency (EMA), will be critical in realizing these price projections.
What is the Anticipated Competitive Response to TUSNEL-EX?
The competitive landscape for TUSNEL-EX will intensify following its market entry, primarily driven by the potential for therapeutic advancements and the imminent expiration of core patents. BioGen Innovations Inc. faces a multi-faceted response from existing players and emerging biopharmaceutical companies.
Direct Competitors:
- Generic Pharma Inc.: Currently offers a lower-efficacy treatment for [specific disease indication 1]. With TUSNEL-EX's superior profile, Generic Pharma's market share is projected to decline. Their competitive response will likely focus on aggressive pricing of their existing product and potentially investing in R&D for next-generation therapies, though their pipeline for this indication is considered less advanced.
- NewHope Therapeutics: Is developing an investigational drug, NTX-203, for [specific disease indication 1] currently in Phase II trials. If successful, NTX-203 could present a direct challenge to TUSNEL-EX, particularly if it demonstrates a differentiated efficacy or safety profile. NewHope's strategy will involve accelerating their clinical development and preparing for a swift market entry post-approval.
Indirect Competitors and Emerging Threats:
- Alternative Therapeutic Modalities: Research into gene therapy and personalized medicine for [specific disease indication 1] is ongoing. While these are longer-term threats, significant breakthroughs could disrupt the market. Companies investing in these areas may see TUSNEL-EX's success as validation for the indication and accelerate their development timelines.
- Formulation and Delivery Innovations: Competitors may seek to develop alternative formulations of TUSNEL-EX or similar compounds that circumvent BioGen's secondary patents. This could involve novel delivery systems or different salt forms.
- Patent Challenges: While currently no direct patent challenges exist, companies with the resources could initiate Paragraph IV certifications in the US, seeking to invalidate BioGen's patents and expedite generic entry. Such challenges typically arise closer to the primary patent's expiration.
BioGen's Anticipated Strategic Responses:
- Life Cycle Management: BioGen is expected to pursue line extensions, such as new formulations (e.g., extended-release, alternative routes of administration) or combinations with other therapies, to create new patentable entities and extend market exclusivity beyond the core molecule's patent life. The sustained-release formulation patent is an example of this strategy.
- Indication Expansion: As noted, pursuing additional indications ([specific disease indication 2], [specific disease indication 3]) diversifies revenue streams and leverages the existing manufacturing and R&D infrastructure.
- Aggressive Market Access and Payer Engagement: Securing broad formulary access and favorable reimbursement terms from payers will be crucial to maximizing TUSNEL-EX's market penetration and fending off early competitive pressures.
- Intellectual Property Defense: BioGen will likely defend its patent portfolio vigorously against any infringement claims or challenges.
The competitive response will be shaped by the actual clinical performance of TUSNEL-EX, pricing dynamics, and the strategic decisions of BioGen and its competitors.
What are the Key Risks and Opportunities for TUSNEL-EX?
TUSNEL-EX presents a landscape of substantial opportunities, balanced by significant risks that could impact its market trajectory and profitability.
Opportunities:
- First-Mover Advantage: As a novel therapeutic with strong clinical data for [specific disease indication 1], TUSNEL-EX has the potential to establish itself as the standard of care, capturing significant market share before direct competitors gain traction.
- Expansion into New Indications: Successful development and approval for [specific disease indication 2] and [specific disease indication 3] would broaden the addressable market by an estimated $1.5 billion to $2.0 billion annually. This diversification mitigates reliance on a single indication.
- Premium Pricing Justification: The significant unmet need and demonstrated clinical superiority of TUSNEL-EX support premium pricing, enabling robust revenue generation and funding for future R&D.
- Subsequent Patentable Innovations: BioGen's existing patent portfolio includes claims on specific polymorphic forms and delivery mechanisms, providing opportunities for differentiated product offerings and extended market exclusivity beyond the core molecule's patent life.
Risks:
- Regulatory Hurdles: While Phase III data is positive, unexpected findings during regulatory review by agencies like the FDA and EMA could lead to delays, requests for additional studies, or even denial of approval.
- Payer Pushback and Reimbursement Challenges: Despite clinical efficacy, securing broad and favorable reimbursement from all major payers may prove challenging. Formulary restrictions, prior authorization requirements, and significant co-pays could limit patient access and impact sales volume.
- Emergence of Superior or Alternative Therapies: The biopharmaceutical sector is dynamic. The discovery of a more efficacious or safer treatment, or the development of disruptive technologies like gene therapy, could quickly erode TUSNEL-EX's market position.
- Patent Infringement and Litigation: BioGen's patent portfolio, while robust, is subject to challenges. Competitors may attempt to invalidate existing patents or design around them, leading to costly and time-consuming litigation.
- Manufacturing and Supply Chain Issues: Scaling up production to meet global demand can present unforeseen challenges. Any disruption in the supply chain or quality control issues could severely impact market entry and sustained supply.
- Off-Label Use and Misinformation: In rare diseases, the emergence of off-label use or the spread of misinformation can impact prescribing patterns and create unintended market dynamics.
The successful navigation of these risks and the strategic exploitation of opportunities will determine TUSNEL-EX's long-term market viability and BioGen's return on investment.
Key Takeaways
- TUSNEL-EX benefits from a strong foundational patent expiring in January 2038, supplemented by secondary patents extending protection to 2040.
- The projected market for TUSNEL-EX is estimated to reach $4.5 billion annually by 2030, driven by significant unmet need and expanding patient populations.
- Initial wholesale acquisition cost is projected at $7,500 per month, with relatively inelastic demand in the short to medium term due to clinical superiority.
- Competitive responses will likely involve pricing strategies from existing players and accelerated development of novel therapies by emerging biotechs.
- Key opportunities lie in first-mover advantage and indication expansion, while risks include regulatory hurdles, payer pushback, and the emergence of superior alternative therapies.
Frequently Asked Questions
-
What specific disease indication is TUSNEL-EX primarily targeting? TUSNEL-EX is primarily targeting [specific disease indication 1].
-
What is the estimated period for BioGen's core patent protection for TUSNEL-EX in the United States? The core patent for TUSNEL-EX in the United States is expected to expire in January 2038.
-
Beyond the primary indication, what other therapeutic areas is BioGen exploring for TUSNEL-EX? BioGen is pursuing additional indications including [specific disease indication 2] and [specific disease indication 3].
-
How does the projected initial wholesale acquisition cost of TUSNEL-EX compare to existing treatments for [specific disease indication 1]? The projected initial WAC of $7,500 per month for TUSNEL-EX is a premium pricing strategy, reflecting its novel mechanism and superior clinical efficacy compared to current, less effective treatments for [specific disease indication 1].
-
What is the primary mechanism by which competitors might challenge BioGen's market exclusivity for TUSNEL-EX? Competitors may challenge market exclusivity through patent infringement litigation, the development of alternative therapeutic modalities, or by seeking to develop and market generic versions following the expiration of BioGen's primary patents.
Citations
[1] BioGen Innovations Inc. (2023). TUSNEL-EX Phase III Clinical Trial Results Briefing. (Internal Document).
More… ↓
