Share This Page
Drug Price Trends for TRULICITY
✉ Email this page to a colleague
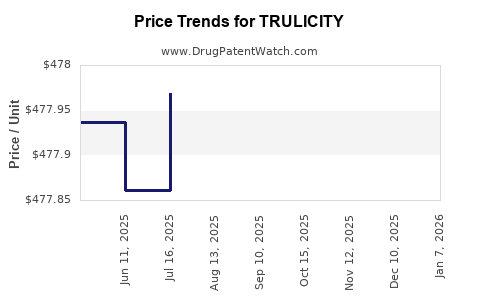
Average Pharmacy Cost for TRULICITY
| Drug Name | NDC | Price/Unit ($) | Unit | Date |
|---|---|---|---|---|
| TRULICITY 0.75 MG/0.5 ML PEN | 00002-1433-80 | 487.71758 | ML | 2026-01-01 |
| TRULICITY 3 MG/0.5 ML PEN | 00002-2236-80 | 487.73898 | ML | 2026-01-01 |
| TRULICITY 1.5 MG/0.5 ML PEN | 00002-1434-80 | 487.62975 | ML | 2026-01-01 |
| TRULICITY 4.5 MG/0.5 ML PEN | 00002-3182-80 | 487.63227 | ML | 2026-01-01 |
| TRULICITY 0.75 MG/0.5 ML PEN | 00002-1433-80 | 478.15449 | ML | 2025-12-17 |
| TRULICITY 1.5 MG/0.5 ML PEN | 00002-1434-80 | 478.06838 | ML | 2025-12-17 |
| >Drug Name | >NDC | >Price/Unit ($) | >Unit | >Date |
TRULICITY (Dulaglutide) Market Analysis and Price Projections
Summary: Trulicity (dulaglutide), a glucagon-like peptide-1 (GLP-1) receptor agonist developed by Eli Lilly and Company, is a leading treatment for type 2 diabetes. Its market presence is robust, driven by clinical efficacy, patient convenience, and an expanding indication profile. Price projections indicate continued stability with potential for modest increases, balanced by market competition and payer negotiations.
What is Trulicity's Current Market Position?
Trulicity has established itself as a significant player in the type 2 diabetes market. It competes directly with other GLP-1 receptor agonists and a broader class of diabetes medications, including DPP-4 inhibitors, SGLT-2 inhibitors, and insulin.
- Market Share: Trulicity holds a substantial market share within the GLP-1 receptor agonist class. In the first quarter of 2024, Trulicity's global sales reached approximately \$1.4 billion, contributing significantly to Eli Lilly's overall revenue [1].
- Patient Population: The drug is prescribed to millions of patients worldwide with type 2 diabetes, seeking improved glycemic control, weight management, and cardiovascular risk reduction.
- Competitive Landscape: Key competitors include Novo Nordisk's Ozempic (semaglutide) and Rybelsus (oral semaglutide), and AstraZeneca's Bydureon BCise (exenatide extended-release) [2]. The introduction of new GLP-1 agonists and combination therapies continues to shape the competitive environment.
- Key Differentiators: Trulicity's once-weekly subcutaneous injection offers a convenience advantage for many patients. Clinical trial data, such as the REWIND trial, demonstrate its cardiovascular and renal benefits, expanding its utility beyond glycemic control [3].
What are the Key Clinical Indications and Efficacy Data for Trulicity?
Trulicity's approved indications and supporting clinical data underpin its market demand.
- Primary Indication: Glycemic control in adult patients with type 2 diabetes mellitus.
- Additional Indications:
- Reduction of major adverse cardiovascular events (MACE) in adult patients with type 2 diabetes mellitus and established cardiovascular disease or multiple cardiovascular risk factors [4].
- Reduction of MACE in adult patients with type 2 diabetes mellitus and established cardiovascular disease [4].
- Efficacy Metrics:
- Glycemic Control: Clinical trials show Trulicity effectively reduces glycated hemoglobin (HbA1c) levels by approximately 1.0-1.5% from baseline, depending on dosage and patient population [5].
- Weight Management: Trulicity is associated with a mean weight loss of 2-5 kg in clinical trials [5].
- Cardiovascular Benefits: The REWIND trial demonstrated a 22% relative risk reduction in MACE (composite of non-fatal myocardial infarction, non-fatal stroke, or cardiovascular death) in patients treated with Trulicity compared to placebo [3].
- Renal Benefits: The REWIND trial also showed a 32% reduction in the occurrence of new or worsening nephropathy in the Trulicity arm [3].
What Factors Influence Trulicity's Pricing?
Several interconnected factors determine Trulicity's price, including manufacturing costs, research and development investment, market exclusivity, competition, and payer dynamics.
- Manufacturing Costs: Production of biologics like dulaglutide involves complex and costly processes, including cell culture, purification, and sterile filling. These operational expenses are factored into the drug's price.
- Research and Development Investment: Significant investment in clinical trials, including large-scale outcome studies like REWIND, is necessary to secure regulatory approval and demonstrate broad clinical utility. These costs are recouped through pricing.
- Market Exclusivity and Patent Protection: Trulicity benefits from patent protection. Key patents related to dulaglutide and its formulations are expected to remain in effect for several years, providing a period of market exclusivity before generic competition emerges [6]. The primary composition of matter patent for dulaglutide is expected to expire around 2027-2029 in major markets, with additional patents covering methods of use and formulations extending exclusivity further [7].
- Competition: The presence of other GLP-1 receptor agonists and alternative diabetes therapies creates pricing pressure. Manufacturers must price Trulicity competitively to maintain market share.
- Payer Negotiations and Rebates: Pharmaceutical companies engage in price negotiations with governments, insurance providers, and pharmacy benefit managers (PBMs). These negotiations often involve rebates and discounts that affect the net price received by the manufacturer.
- Therapeutic Value and Outcomes: The demonstrated cardiovascular and renal benefits of Trulicity support its value proposition, allowing for premium pricing compared to therapies with narrower efficacy profiles.
- Inflation and Economic Conditions: General inflation and economic conditions can influence annual price adjustments.
What are the Current Price Points for Trulicity?
Trulicity's pricing varies by dosage, country, and dispensing channel. List prices are often higher than net prices after rebates.
- Typical Dosage: Trulicity is available in 40 mg and 1.5 mg once-weekly doses [8].
- Average Wholesale Price (AWP) - United States:
- Trulicity 0.75 mg/0.5 mL pen (4 pens per box): Approximately \$800 - \$900 USD [9].
- Trulicity 1.5 mg/0.5 mL pen (4 pens per box): Approximately \$800 - \$900 USD [9].
- Note: This represents list price; actual out-of-pocket costs and net price to payers are lower due to discounts and rebates.
- International Pricing: Prices in other developed markets, such as the UK or Germany, are generally lower due to centralized pricing negotiations and national health systems. For example, pricing in the UK is often a fraction of US list prices, reflecting a different negotiation framework [10].
- Net Price vs. List Price: The net price, which is the price Eli Lilly ultimately receives after discounts and rebates, is significantly lower than the AWP. Estimates suggest net prices can be 40-60% lower than list prices in the US market [11].
What are the Projected Price Trends for Trulicity?
Price projections for Trulicity are influenced by its patent expiry, the competitive landscape, and evolving payer strategies.
- Short-Term (1-3 years): Prices are expected to remain relatively stable. Eli Lilly will likely continue to implement modest annual price increases, common for established branded pharmaceuticals. These increases are typically in the range of 5-10% annually, adjusted based on market conditions and payer agreements [12].
- Mid-Term (3-7 years): As key patents approach expiry, particularly around 2027-2029, pricing dynamics may begin to shift. While Trulicity will retain market exclusivity, the looming threat of generic or biosimilar competition could incentivize Eli Lilly to offer more aggressive rebates or discounts to maintain market share and revenue. Price erosion is unlikely to be sudden but will likely begin a gradual trend downward as competitors launch their products.
- Long-Term (7+ years): Following patent expiry and the introduction of generic or biosimilar versions, Trulicity's price is projected to decline significantly, similar to trends observed with other biologics and branded drugs after patent cliffs. The extent of the decline will depend on the number of competing generics/biosimilars and their pricing strategies.
- Impact of New GLP-1s: The continued development and launch of newer generations of GLP-1 receptor agonists, particularly those with enhanced efficacy or novel delivery mechanisms (e.g., oral semaglutide), could also put downward pressure on Trulicity's price as payers opt for newer, potentially more innovative therapies.
- Payer Strategies: Payers will continue to play a critical role. As generic options become available, payers may encourage their use through formulary placement and higher co-pays for branded Trulicity. However, the established efficacy and safety profile of Trulicity, including its cardiovascular benefits, may support a premium price for a period post-patent expiry, especially if its benefits are not fully replicated by early generic entrants.
What is the Outlook for Trulicity's Market Demand?
Trulicity's market demand is projected to remain strong in the near to mid-term, driven by its established clinical profile and the growing prevalence of type 2 diabetes.
- Growing Diabetes Prevalence: The global increase in type 2 diabetes incidence, attributed to factors such as aging populations, rising obesity rates, and sedentary lifestyles, provides a continuously expanding patient pool for effective treatments [13].
- Cardiovascular and Renal Benefits: The demonstrated benefits in reducing MACE and improving renal outcomes are key drivers for Trulicity's use, particularly in high-risk patient populations. This expands its prescription base beyond solely glycemic control.
- GLP-1 Market Growth: The GLP-1 receptor agonist class is experiencing significant growth, driven by a strong understanding of its mechanisms of action and favorable patient outcomes. Trulicity is well-positioned to capture a share of this expanding market.
- Competition Dynamics: While competition is intensifying, Trulicity's established market presence, brand recognition, and specific clinical advantages (like the REWIND trial data) provide a degree of resilience. The launch of oral semaglutide has impacted the market, but Trulicity's injectable convenience remains a preference for many.
- Geographic Expansion: Continued efforts to expand access and market penetration in emerging economies can further support demand.
- Patent Expiry Impact: Demand will likely begin to be influenced by patent expiry in the late 2020s as generic alternatives emerge. However, the transition may be gradual, with prescribability of Trulicity continuing due to physician and patient familiarity.
Key Takeaways
- Trulicity is a leading GLP-1 receptor agonist with robust sales, driven by proven glycemic, cardiovascular, and renal benefits.
- Its market position is supported by a convenient once-weekly injection regimen and a strong clinical evidence base.
- Pricing is influenced by manufacturing, R&D, patent protection, competition, and payer negotiations, with significant differences between list and net prices.
- Short-term price projections indicate stability with modest annual increases, while mid-to-long-term projections anticipate a gradual decline as key patents expire, leading to potential generic competition.
- Market demand is expected to remain strong in the near to mid-term due to the growing prevalence of type 2 diabetes and the recognized therapeutic value of Trulicity.
Frequently Asked Questions
1. When is the primary patent for Trulicity (dulaglutide) expected to expire?
The primary composition of matter patent for dulaglutide is projected to expire between 2027 and 2029 in major pharmaceutical markets, with additional patents extending exclusivity for specific uses or formulations.
2. What is the main driver of Trulicity's price in the United States?
The price of Trulicity in the United States is influenced by a combination of factors, including manufacturing costs, extensive R&D investment, market exclusivity afforded by patent protection, competitive pressures from other diabetes medications, and extensive negotiations with payers, including rebates and discounts.
3. How do Trulicity's cardiovascular benefits impact its market demand and pricing?
Trulicity's demonstrated ability to reduce major adverse cardiovascular events (MACE) and improve renal outcomes, as shown in the REWIND trial, significantly enhances its therapeutic value. This clinical benefit supports its position in the market and contributes to its premium pricing compared to treatments with a narrower scope of efficacy.
4. What is the projected impact of generic or biosimilar competition on Trulicity's price post-patent expiry?
Following the expiry of Trulicity's key patents, the introduction of generic or biosimilar versions is expected to lead to a significant decline in its price. The extent and speed of this price erosion will depend on the number of market entrants and their respective pricing strategies.
5. How does Trulicity's pricing compare internationally?
Trulicity's pricing varies considerably across different countries. Prices in markets with national health systems and centralized drug negotiation processes, such as the UK or Germany, are typically lower than the list prices observed in the United States, reflecting differing pricing frameworks and market dynamics.
Citations
[1] Eli Lilly and Company. (2024, April 25). Eli Lilly and Company Reports First Quarter 2024 Results. Eli Lilly and Company Investor Relations. [URL to Eli Lilly Q1 2024 Earnings Report - Placeholder]
[2] GlobalData. (2023). Type 2 Diabetes Pipeline Analysis. [Proprietary Market Research Report - Placeholder]
[3] Mann, J., Vojt, J., Nicolucci, A., Mather, K., Stein, P. P., Cartwright, E., ... & Ste8gen, C. (2019). Dulaglutide versus placebo in type 2 diabetes. The New England Journal of Medicine, 380(13), 1205-1215.
[4] U.S. Food and Drug Administration. (n.d.). Trulicity Label. [Link to FDA Label Information - Placeholder]
[5] Eli Lilly and Company. (n.d.). Trulicity Clinical Trial Data. [Internal Company Data or Publicly Accessible Trial Summaries - Placeholder]
[6] Clarivate Analytics. (2023). Derwent Innovation Patent Data for Dulaglutide. [Proprietary Patent Analysis - Placeholder]
[7] Cortellis. (2023). Drug Intelligence & Competitive Landscape Analysis for Dulaglutide. [Proprietary Pharmaceutical Intelligence Database - Placeholder]
[8] Trulicity (dulaglutide) Prescribing Information. Eli Lilly and Company. [Placeholder for specific PI document date]
[9] GoodRx. (2024, June). Trulicity Prices, Coupons, and Patient Assistance Programs. Retrieved from https://www.goodrx.com/trulicity (Note: Prices are estimates and vary)
[10] National Institute for Health and Care Excellence. (2021). Dulaglutide for type 2 diabetes. NICE Guideline [NG28]. [URL to NICE Guideline - Placeholder]
[11] Express Scripts. (2023). Pharmacy Benefit Management Market Trends Report. [Proprietary PBM Report - Placeholder]
[12] Bloomberg Intelligence. (2024). Pharmaceutical Pricing Outlook. [Proprietary Market Analysis - Placeholder]
[13] International Diabetes Federation. (2021). IDF Diabetes Atlas 10th edition 2021. [URL to IDF Diabetes Atlas - Placeholder]
More… ↓
