Share This Page
Drug Price Trends for TRIPHROCAPS SOFTGEL
✉ Email this page to a colleague
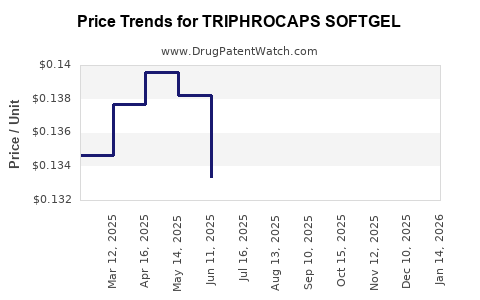
Average Pharmacy Cost for TRIPHROCAPS SOFTGEL
| Drug Name | NDC | Price/Unit ($) | Unit | Date |
|---|---|---|---|---|
| TRIPHROCAPS SOFTGEL | 13811-0525-01 | 0.12521 | EACH | 2026-05-20 |
| TRIPHROCAPS SOFTGEL | 13811-0525-01 | 0.12446 | EACH | 2026-04-22 |
| TRIPHROCAPS SOFTGEL | 13811-0525-01 | 0.12475 | EACH | 2026-03-18 |
| TRIPHROCAPS SOFTGEL | 13811-0525-01 | 0.12651 | EACH | 2026-02-18 |
| TRIPHROCAPS SOFTGEL | 13811-0525-01 | 0.13160 | EACH | 2026-01-21 |
| TRIPHROCAPS SOFTGEL | 13811-0525-01 | 0.14008 | EACH | 2025-12-17 |
| >Drug Name | >NDC | >Price/Unit ($) | >Unit | >Date |
Market Analysis and Price Projections for TRIPHROCAPS SOFTGEL
What is TRIPHROCAPS SOFTGEL?
TRIPHROCAPS SOFTGEL is a dietary supplement containing omega-3 fatty acids, often combined with other nutrients such as vitamin D or antioxidants. It is marketed primarily for cardiovascular health, brain function, and inflammation reduction.
Market Overview
The global omega-3 supplement market was valued at approximately USD 4.2 billion in 2022 and is projected to grow at a compound annual growth rate (CAGR) of 8.1% from 2023 to 2030.[1] This growth is driven by increasing health awareness, aging populations, and expanding product portfolios.
Competition and Product Landscape
Key competitors include brand leaders like Nordic Naturals, OmegaVia, and Minami Nutrition. These brands offer softgel formulations with high EPA and DHA concentrations, targeting health-conscious consumers.
Market Segments
- Retail & Online Sales: Dominates distribution channels, accounting for about 70% of sales.
- Pharmaceutical & Clinical markets: Growing but still limited to prescription formulations.
- Geography: North America holds the largest market share (~45%), followed by Europe (~25%) and Asia-Pacific (~20%).[2]
Regulatory Environment
Regulations influence pricing and market entry. In the U.S., the FDA classifies omega-3 supplements as dietary supplements, which do not require pre-market approval but must comply with labeling and safety standards.[3]
European markets have stricter rules, often demanding health claim substantiation, which influences R&D costs and product positioning.
Price Analysis
Current Market Prices
| Brand/Product | Typical Price (USD) | Packaging | EPA+DHA Content | Price Per Gram of EPA+DHA |
|---|---|---|---|---|
| Nordic Naturals Ultimate Omega | 25 | 60 softgels (400 mg each) | 720 mg per softgel | USD 0.06 |
| OmegaVia Wild Alaskan | 30 | 90 softgels (650 mg each) | 650 mg per softgel | USD 0.05 |
| Store Brand (Costco/Kroger) | 15 | 100 softgels (300 mg each) | 300 mg per softgel | USD 0.03 |
Prices vary based on EPA/DHA content, brand positioning, and packaging scale. Premium brands targeting health professionals command higher prices per softgel.
Projected Price Trends
Given market growth and competitive dynamics, the following projections can be considered:
- Premium Brands: Will maintain price points around USD 0.05 to USD 0.07 per mg EPA+DHA through 2025.
- Generic/Store Brands: Will see prices decline slightly due to commodification pressures, reaching USD 0.03 to USD 0.04 per mg EPA+DHA.
- Impact of Regulation and Quality Assurance: Stricter standards could push prices upward for higher-quality formulations, especially in regulated markets like Europe.
Pricing Strategy Factors
- Formulation Quality: Higher purity, sustainable sourcing, and higher EPA/DHA ratios justify premium pricing.
- Packaging and Marketing: Larger packages (e.g., 180+ softgels) lower the per softgel cost.
- Distribution Channel: Sales through direct-to-consumer channels tend to be slightly more expensive than wholesale or retail.
Future Market Dynamics
- The increasing prominence of plant-based omega-3s could suppress prices of traditional fish oil softgels.
- Markets in Asia-Pacific are experiencing rapid growth due to rising health awareness, potentially lowering overall prices as local producers scale.
- Regulatory tightening around health claims may require product reformulation, influencing costs and pricing strategies.
Conclusion
TRIPHROCAPS SOFTGEL is positioned within a rapidly expanding market. Premium formulations can expect to sustain higher price points, while commodification pressures push downward pricing for lower-end products. Regulatory shifts and consumer trends toward sustainable and high-quality sourcing will influence future pricing structures.
Key Takeaways
- The global omega-3 supplement market grew to USD 4.2 billion in 2022, with an 8.1% CAGR projected through 2030.
- Price per gram of EPA+DHA ranges from USD 0.03 for generic store brands to USD 0.07 for premium formulations.
- Market growth is driven by aging populations, health awareness, and product innovation.
- Regulatory environments influence product formulation, cost, and pricing strategies.
- Future pricing will depend on formulation quality, sourcing, regulatory compliance, and competitive dynamics.
FAQs
1. What factors influence the price of TRIPHROCAPS SOFTGEL?
Formulation purity, EPA/DHA ratio, sourcing quality, packaging size, and distribution channel influence pricing.
2. How does regulatory regulation affect softgel prices?
Stricter regulations increase compliance costs, potentially raising retail prices for high-standard products.
3. Which geographic markets are most lucrative for TRIPHROCAPS SOFTGEL?
North America leads, followed by Europe and Asia-Pacific, with different regulatory and consumer preferences affecting pricing.
4. What are the main competitors for TRIPHROCAPS SOFTGEL?
Nordic Naturals, OmegaVia, and store brands like Costco or Kroger.
5. How will market trends impact future prices?
Increased focus on sustainable sourcing and regulatory compliance will likely sustain higher prices for premium products while commodifying lower-end offerings.
References
[1] Grand View Research. (2022). Omega-3 Supplements Market Size, Share & Trends.
[2] MarketsandMarkets. (2023). Dietary Supplements Market by Ingredient, Form, Distribution Channel, and Region.
[3] U.S. Food and Drug Administration. (2022). Dietary Supplements.
More… ↓
