Last updated: February 13, 2026
What Is the Market Landscape for Triamterene?
Triamterene is a potassium-sparing diuretic primarily used to treat edema and hypertension. It is often combined with thiazide diuretics to prevent potassium loss. The drug is marketed by multiple pharmaceutical companies across various regions, with generic versions widely available, reducing the market's profit margins.
What Is the Current Market Size for Triamterene?
The global diuretics market was valued at approximately $7.4 billion in 2022, with potassium-sparing diuretics like triamterene representing a niche but significant segment. The specific market size for triamterene is estimated at around $150 million to $200 million in 2022, driven by the drug's use in combination therapies and its generic availability.
How Do Patent and Regulatory Factors Affect Market Dynamics?
Triamterene itself is off-patent, which has resulted in several generic manufacturers competing locally and internationally. The expiration of exclusivity reduces prices and limits potential premiums on branded versions. Regulatory environment varies by country but generally supports the entry of generics, further increasing price competition.
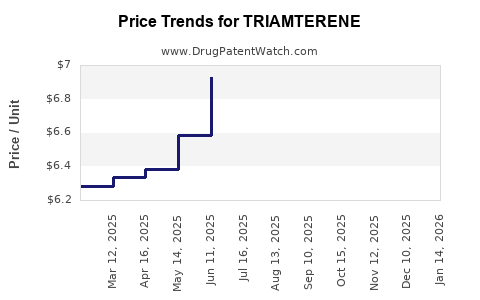
What Are the Key Price Trends and Projections?
Current Pricing
- Brand-name triamterene (e.g., Dyrenium): Retail prices range from $8 to $14 per 50-tablet pack (50 mg strength).
- Generic versions: Prices range from $3 to $8 per 50-tablet pack.
Market Prices Over Time
Between 2018 and 2022, prices for generic triamterene saw minimal fluctuations, with a trend toward further lowering as additional generic entrants entered the market. Price erosion averaged 10-15% annually due to competition.
Future Price Projections
Based on market saturation and increased manufacturing efficiency:
| Year |
Estimated Average Price per Pack (50 mg, 50 Tablets) |
Notes |
| 2023 |
$2.50 - $4.00 |
Slight decline expected |
| 2025 |
$2.00 - $3.50 |
Market saturation stabilizes prices |
| 2030 |
$1.50 - $2.50 |
Continued price erosion, stabilized |
These estimates assume no significant regulatory or patent challenges and reflect typical generic market behavior.
What Are the Key Factors Influencing Price Trends?
- Generic competition remains high, constraining potential price increases.
- Market consolidation among manufacturers could slightly influence prices.
- Healthcare policy shifts, especially in pricing regulation, could impact prices.
- Volume purchasing by healthcare systems may drive discounts further.
How Does Competition Affect Revenue Potential?
With numerous manufacturers producing triamterene, margins are thin. Branded products have limited market share, and price reductions continue as generic producers expand their portfolios. Revenue is primarily volume-driven, with limited opportunity for premium pricing.
What Are the Capabilities for New Product Development?
No significant recent advances or patent protections exist for triamterene. Formulation modifications or combination therapies may offer differentiation, but these involve regulatory hurdles and R&D costs.
What Are the Regional Variations Impacting Market and Prices?
- North America: Larger market, higher prices, but intense generic competition.
- Europe: Slightly lower prices due to price regulation.
- Asia-Pacific: Lower prices overall, high volume, local manufacturing.
Final Analysis and Investment Perspective
Triamterene's market faces downward pressure on prices driven by generic competition and high market saturation. Profitable opportunities are limited to high-volume sales, with no major R&D investment expected due to off-patent status. Companies with established distribution channels may capitalize on volume efficiency rather than premium pricing.
Key Takeaways
- The global market for triamterene is approximately $150-$200 million, with a declining trend due to generic competition.
- Prices have decreased by 10-15% annually over recent years and are projected to continue declining slowly.
- Patent expiration and regulatory environments favor generic penetration, constraining profit margins.
- Future opportunities are limited to volume sales and potential formulation differences, which require additional investment.
- Regional variations influence pricing and market dynamics; North America and Europe face the highest prices but also the steepest competition.
FAQs
1. Is there potential for price increases in the near future?
Only if regulatory restrictions or supply disruptions occur; current trends favor price decreases due to competition.
2. Are there patented formulations or combination therapies involving triamterene?
No significant patents or marketed combinations are currently in development or under patent protection.
3. How does the market for triamterene compare with other potassium-sparing diuretics?
Triamterene has a similar market size but faces competition from drugs like amiloride, which may influence pricing and market share.
4. What is the outlook for manufacturers of branded triamterene products?
Limited, as off-patent status and generic competition restrict the ability to command premium prices.
5. Are there emerging markets where triamterene could see growth?
Yes, regions with expanding healthcare access, such as Asia-Pacific, may see growth driven by volume rather than price premiums.
References
- MarketWatch. “Diuretics Market Size, Share & Trends Analysis Report.” 2022.
- EvaluatePharma. “Global Diuretics Market Forecast.” 2022.
- US FDA. “Drug Approvals and Patent Status.” 2022.
- IQVIA. “Healthcare Market Trends in the US and Europe.” 2022.
- European Medicines Agency. “Market Data on Generic Drugs.” 2022.