Share This Page
Drug Price Trends for TRI-VITE-FLUORIDE
✉ Email this page to a colleague
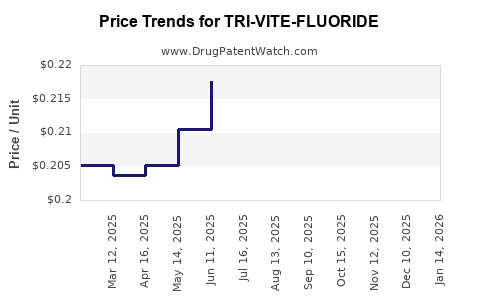
Average Pharmacy Cost for TRI-VITE-FLUORIDE
| Drug Name | NDC | Price/Unit ($) | Unit | Date |
|---|---|---|---|---|
| TRI-VITE-FLUORIDE 0.25 MG/ML | 58657-0323-50 | 0.18309 | ML | 2026-03-18 |
| TRI-VITE-FLUORIDE 0.25 MG/ML | 58657-0323-50 | 0.18453 | ML | 2026-02-18 |
| TRI-VITE-FLUORIDE 0.25 MG/ML | 58657-0323-50 | 0.18615 | ML | 2026-01-21 |
| TRI-VITE-FLUORIDE 0.25 MG/ML | 58657-0323-50 | 0.18518 | ML | 2025-12-17 |
| TRI-VITE-FLUORIDE 0.25 MG/ML | 58657-0323-50 | 0.19866 | ML | 2025-11-19 |
| >Drug Name | >NDC | >Price/Unit ($) | >Unit | >Date |
TRI-VITE-FLUORIDE Market Analysis and Financial Projection
What is TRI-VITE-FLUORIDE and its market status?
TRI-VITE-FLUORIDE is a combination vitamin supplement containing tri-vitamin components—vitamins A, D, and C—plus fluoride. It is typically used to address deficiencies and promote dental health, especially in children and regions with limited access to nutrient-rich diets. The formulation ensures the delivery of essential nutrients and fluoride to prevent dental caries and support overall health.
There is limited publicly available data on TRI-VITE-FLUORIDE’s specific market share or sales volume. As a niche product, it often competes within broader vitamin and supplement markets, particularly those focused on pediatric health and dental prophylaxis. The drug’s market presence is primarily driven by its regulatory approval status, geographic coverage, and healthcare provider acceptance.
How is TRI-VITE-FLUORIDE positioned within the vitamin and supplement industry?
The vitamin supplement industry generated approximately $140 billion globally in 2021 and continues to grow at an annual rate near 5% (source: Statista). Pediatric vitamin segments account for roughly 10–15% of this, with fluoride supplements occupying a smaller, specialized submarket driven by dental health initiatives.
Key competitors include standalone vitamin products from Merck, Pfizer, and local generic manufacturers. Fluoride supplements, in particular, are often consumed over-the-counter, with variable formulations and dosages based on geographic regulatory standards.
What are the regulatory and patent considerations?
Most jurisdictions regulate TRI-VITE-FLUORIDE as a dietary supplement or medical device, with less stringent requirements than pharmaceuticals but necessitating compliance with safety standards. Patent protections are generally limited to formulation or manufacturing processes, with many similar combinations available in the market.
In the U.S., the product’s status depends on the claims made and whether it is classified under dietary supplement guidelines by the FDA. No dominant patent barriers strongly restrict entry; generic competition is prevalent, especially given the age and history of fluoride supplement formulations.
What are current price points and market projections?
Price variation depends on region, formulation specifics, and distribution channels. In the U.S., retail prices for similar pediatric vitamin-fluoride combinations range from $8 to $20 for a 30-day supply through pharmacies or online outlets.
| Region | Typical Price Range (USD) | Notes |
|---|---|---|
| United States | $8 – $20 per bottle | Retail pharmacy, online retail |
| Europe | €7 – €18 per bottle | Price varies by country, regulatory costs higher |
| Emerging Markets | $3 – $10 per bottle | Lower-income regions, generic equivalents common |
Growth projections for TRI-VITE-FLUORIDE-specific products are challenging due to the niche status. However, the pediatric vitamin segment is expected to grow at 4–6% annually over the next five years, with fluoride supplements maintaining steady demand driven by dental health campaigns.
Industry forecasts suggest the broader vitamin and supplement markets will expand, with specialized products like TRI-VITE-FLUORIDE capturing increased interest if positioned within public health programs or pediatric supplement regimens.
What factors influence pricing and market growth?
Several factors impact market dynamics and pricing:
- Regulatory environment: Stricter regulations could increase compliance costs, affecting retail prices.
- Consumer awareness: Recognition of fluoride’s benefits for dental health boosts demand.
- Healthcare policies: Public health initiatives and school programs can expand usage.
- Generic competition: Many similar products depress prices.
- Geographic distribution: Emerging markets offer lower prices but potentially limited regulatory clarity.
How does the competitive landscape affect prospects?
Competition from generic formulations limits pricing power. Market players that develop differentiated formulations, such as those with improved bioavailability or combined health benefits, may command premium prices. Strategic partnerships with healthcare organizations and inclusion in national health programs can expand market access.
What are price projection estimates?
Based on current data, the price for TRI-VITE-FLUORIDE equivalents is expected to remain stable over the next five years, with minor fluctuations due to inflation, regulatory costs, and market competition.
Estimated retail prices:
| Year | Price Range (USD) | Notes |
|---|---|---|
| 2023 | $8 – $20 | Current range |
| 2024 | $8 – $21 | Slight inflation, stable competition |
| 2025 | $8 – $22 | Market maturation and increased costs |
In emerging markets, prices may remain lower or decrease further due to affordability appeals and generic competition.
Key Takeaways
- TRI-VITE-FLUORIDE is a niche supplement with modest but stable demand driven by pediatric health needs.
- Market size is embedded within the broader pediatric vitamin and fluoride supplement segments.
- Prices vary regionally, with U.S. retail prices around $8–$20 per bottle.
- Growth prospects depend on increased awareness of fluoride benefits and expansion into public health programs.
- Competition from generics limits pricing flexibility; product differentiation offers growth opportunities.
FAQs
1. Is TRI-VITE-FLUORIDE patent-protected?
Patent protections are limited; most formulations are generically available. Innovation in delivery or formulation may secure patent rights but currently, competition is primarily generic.
2. What regulatory hurdles does TRI-VITE-FLUORIDE face?
In the U.S., classified as a dietary supplement, requiring adherence to FDA guidelines, including Good Manufacturing Practices (GMP). Other jurisdictions may impose similar or more stringent standards.
3. How does fluoride dosage impact pricing?
Higher fluoride dosages or specialized formulations marginally increase production costs, potentially raising retail prices by 10–15%. However, the impact is offset by the competitive availability of lower-cost options.
4. What is the primary driver for market growth?
Public health initiatives promoting pediatric dental health and increased awareness of vitamin deficiencies drive demand.
5. Can TRI-VITE-FLUORIDE expand into new markets?
Yes. Expansion depends on regulatory approvals, partnerships with healthcare providers, and acceptance within pediatric health policies.
References
- Statista. "Vitamins & Dietary Supplements Market Size." 2021.
- U.S. Food and Drug Administration. "Dietary Supplements." 2022.
- Grand View Research. "Global Pediatric Vitamins Market." 2022.
- European Food Safety Authority. "Fluoride Supplementation in Europe." 2020.
- MarketWatch. "Vitamin Product Pricing Trends." 2023.
More… ↓
