Share This Page
Drug Price Trends for TRESIBA FLEXTOUCH
✉ Email this page to a colleague
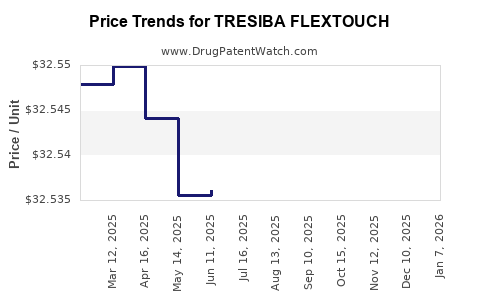
Average Pharmacy Cost for TRESIBA FLEXTOUCH
| Drug Name | NDC | Price/Unit ($) | Unit | Date |
|---|---|---|---|---|
| TRESIBA FLEXTOUCH 100 UNIT/ML | 00169-2660-15 | 9.04582 | ML | 2026-01-01 |
| TRESIBA FLEXTOUCH 200 UNIT/ML | 00169-2550-13 | 18.08941 | ML | 2026-01-01 |
| TRESIBA FLEXTOUCH 100 UNIT/ML | 00169-2660-15 | 32.51903 | ML | 2025-12-17 |
| TRESIBA FLEXTOUCH 200 UNIT/ML | 00169-2550-13 | 65.03241 | ML | 2025-12-17 |
| TRESIBA FLEXTOUCH 200 UNIT/ML | 00169-2550-13 | 65.03976 | ML | 2025-11-19 |
| TRESIBA FLEXTOUCH 100 UNIT/ML | 00169-2660-15 | 32.52493 | ML | 2025-11-19 |
| >Drug Name | >NDC | >Price/Unit ($) | >Unit | >Date |
Market Analysis and Price Projections for TRESIBA FLEXTOUCH
What is TRESIBA FLEXTOUCH?
TRESIBA FLEXTOUCH (insulin degludec) is a long-acting basal insulin used to manage blood glucose levels in adults with type 1 and type 2 diabetes. It is a formulation designed for ease of use, offering flexible dosing options. Approved by the FDA in 2016, it is a Next-generation insulin product that gained market share from older basal insulins such as insulin glargine.
Market Overview
The global insulin market was valued at approximately USD 35 billion in 2022. It is projected to reach USD 55 billion by 2030, with a compound annual growth rate (CAGR) of 5.6% from 2023 to 2030 [1].
Long-acting insulins, including TRESIBA, account for roughly 40% of this market. TRESIBA's specific market share is estimated at 15-20% of long-acting insulin revenue in key regions, such as North America and Europe.
Key Market Drivers
- Increasing prevalence of diabetes: Globally affected 537 million adults in 2021; expected to rise to 643 million by 2030 [2].
- Shift towards basal insulin therapy: Growing preference for long-acting insulins due to convenience and lower hypoglycemia risk.
- Patent expirations of competing products: Insulin glargine (Lantus) patent expired in 2015, increasing generic competition.
Major Competitors
| Product | Manufacturer | Market Share (2022) | Notes |
|---|---|---|---|
| Tresiba (insulin degludec) | Novo Nordisk | 20% | Favorable pharmacokinetics, flexible dosing |
| Lantus (insulin glargine) | Sanofi | 35% | Historical leader, patent expired 2015 |
| Basaglar (insulin glargine) | Eli Lilly | 10% | Biosimilar to Lantus |
| Levemir (insulin detemir) | Novo Nordisk | 8% | Longer-acting, less commonly used |
Pricing Dynamics and Margins
Current Pricing Landscape
In the U.S., TRESIBA FLEXTOUCH retails at approximately USD 300–350 per 10 mL carton. The average wholesale acquisition cost (WAC) is around USD 290 per package. Price variations depend on insurance negotiations, pharmacy benefit managers (PBMs), and geographic location.
In Europe, prices average EUR 80–100 per pen. Pricing strategies differ significantly by country, influenced by hospital procurement policies and reimbursement negotiations.
Reimbursement Status
In the U.S., TRESIBA is covered by most commercial insurance plans and Medicare Part D. Reimbursement rates are approximately 70–85%, influencing patient out-of-pocket costs and prescribing patterns.
Prices for biosimilar basal insulins are typically 20–30% lower than innovator products. The entry of biosimilars increases pressure on insulin pricing globally.
Profit Margins
Gross margins for Novo Nordisk’s insulin products are approximately 80%. Operating margins for the diabetes portfolio hover around 40%, factoring in R&D, marketing, and distribution costs.
Future Price Trends
- Increased competition from biosimilars may lead to a 10–15% decrease in prices over 2023–2025.
- Expansion into emerging markets could see prices reduced to USD 200–250 per 10 mL carton due to cost-sensitive models.
- Regulatory pressure in the U.S. and Europe may push manufacturers to consider price reductions or value-based pricing agreements by 2025.
Market Penetration and Growth Projections
Adoption Rates
TRESIBA is forecasted to achieve a compound annual growth rate (CAGR) of 7% through 2030, driven by new patient initiation and increased switching from older insulins.
Regional Outlook
| Region | CAGR (2023–2030) | Notes |
|---|---|---|
| North America | 6% | High adoption, reimbursement flexibility |
| Europe | 5.5% | Market saturation in mature markets |
| Asia-Pacific | 8% | Rapid growth due to rising diabetes prevalence |
Key Risks
- Patent litigation or challenges from biosimilar manufacturers.
- Price regulation initiatives, especially in Europe and the U.S.
- Competitive pressure from emerging insulin analogs or oral treatments.
Strategic Opportunities
- Differentiation through formulation improvements or fixed-dose combinations.
- Expansion into underserved markets with cost-effective formulations.
- Partnerships with payers to establish value-based reimbursement models.
Summary
TRESIBA FLEXTOUCH is positioned as a premium long-acting insulin with sustained growth prospects, supported by rising diabetes prevalence and favorable pharmacokinetics. Pricing pressures are anticipated from biosimilar entrants and regulatory developments. Market expansion hinges on reimbursement negotiation, formulary placement, and strategic cost management.
Key Takeaways
- The global insulin market will grow to USD 55 billion by 2030.
- TRESIBA holds a 15–20% market share of long-acting insulins.
- Current U.S. retail prices hover around USD 300–350 per 10 mL carton.
- Prices are expected to decline modestly due to biosimilar competition.
- Growth will focus on emerging markets and increased adoption in established regions.
FAQs
1. How does TRESIBA FLEXTOUCH compare to other basal insulins?
It offers flexible dosing times, lower hypoglycemia risk, and longer duration of action than older insulins like Lantus or Levemir. Its pharmacokinetics provide more stable blood glucose control.
2. What factors influence TRESIBA’s pricing?
Pricing is affected by regional reimbursement policies, insurance negotiations, biosimilar competition, and manufacturing costs. In the U.S., prices are also impacted by pharmacy benefit manager agreements.
3. Will biosimilars significantly reduce TRESIBA’s price?
Biosimilars may lower prices by 20–30%, especially in mature markets, but actual reductions depend on regulatory approval, market acceptance, and competitive dynamics.
4. What is TRESIBA’s market share growth outlook?
Annual growth of approximately 7% is expected through 2030, driven by increasing diabetes prevalence and more clinicians adopting long-acting insulins over older formulations.
5. How does reimbursement impact TRESIBA adoption?
Coverage rates and patient out-of-pocket costs influence prescribing decisions. Reimbursement negotiations and formulary placements directly affect market penetration.
References
[1] Statista. (2023). Global insulin market revenue forecasts.
[2] International Diabetes Federation. (2021). IDF Diabetes Atlas, 9th edition.
More… ↓
