Last updated: February 26, 2026
What Is the Current Market Size for Trazodone?
Trazodone is an antidepressant primarily used for major depressive disorder and off-label for insomnia. The global antidepressant market size was valued at approximately USD 15.5 billion in 2022, with trazodone representing a substantial share, estimated at USD 1.2 billion. The drug's off-label use for sleep disorders drives further volume.
In the United States, approximately 35 million prescriptions were dispensed in 2021, with trazodone ranking among the top prescribed antidepressants, especially for off-label sleep use. The market is driven by increased mental health awareness and the off-label prescribing trend.
What Factors Influence Trazodone’s Market Dynamics?
Patent Status
Trazodone's original patents expired in the late 1990s. Generic versions entered the market shortly after, resulting in low retail prices. Recent formulations or new indications have not secured significant new patents, limiting exclusivity.
Competition
The market faces competition from newer antidepressants such as sertraline and escitalopram, which have more favorable side effect profiles. The off-label use for sleep competes with melatonin, benzodiazepines, and Z-drugs.
Off-Label Use and Prescribing Trends
Off-label prescribing accounts for over 70% of trazodone prescriptions. This trend sustains demand despite limited approved indications for sleep.
Regulatory Environment
No recent regulatory barriers prevent generic proliferation. Some local regulatory guidelines for off-label medication use may influence strict prescribing patterns but do not impact drug availability.
What Are Current Pricing Trends?
Wholesale and Retail Pricing
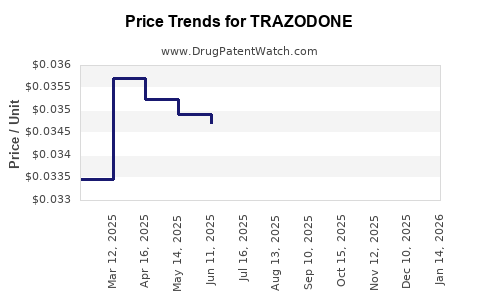
The average wholesale price (AWP) for generic trazodone (50 mg tablets) ranges from USD 0.02 to 0.04 per tablet. Retail prices vary by pharmacy and insurance coverage but remain low due to generic competition.
Price Variability by Formulation
- Trazodone tablets (50 mg): USD 0.02 - 0.04 per tablet.
- Extended-release formulations or compounded versions command higher prices but account for a negligible market share.
Medicaid and Insurance Coverage
Insurance negotiations and formulary placements significantly influence patient out-of-pocket costs. Trazodone's generic status results in low access barriers.
What Is the Future Price Projection?
Short-term (Next 1-2 Years)
Prices are expected to remain stable due to consistent generic supply and stable demand driven by off-label use. The price range for the standard 50 mg tablet should stay in the USD 0.02 - 0.04 range per tablet.
Medium-term (3-5 Years)
Market saturation of generic versions will likely keep prices flat unless new formulations or indications emerge. If a patent application is filed for an extended-release version or combination product, prices could increase temporarily.
Long-term (Beyond 5 Years)
Any significant changes depend on regulatory decisions, new therapeutic claims, or formulation innovations. Without such developments, prices are expected to stay near current levels, with inflation-adjusted minor fluctuations.
Impact of Biosimilars or Specialty Products
Current evidence suggests no biosimilar competition is on the horizon. The drug's status as a small-molecule compound limits the scope for biosimilar emergence.
Key Drivers and Risks
| Driver |
Impact |
Risk |
| Off-label use growth |
Maintains demand |
Regulatory scrutiny reducing prescribing |
| Patent expirations |
Lowers prices |
None expected |
| New formulations |
Could increase prices temporarily |
Limited pipeline development |
| Market competition |
Keeps prices low |
Shift to alternative therapies |
Conclusion
Trazodone remains a low-cost, widely prescribed antidepressant with stable pricing driven by extensive generic competition and off-label use. Price projections suggest minimal variation in the foreseeable future unless a new patent-protected formulation or indication emerges.
Key Takeaways
- The global market for trazodone is approximately USD 1.2 billion, mainly driven by off-label sleep use.
- The drug's price for generic 50 mg tablets remains at USD 0.02 - 0.04 per tablet.
- Price stability is expected over the short to medium term, with potential increases only if new formulations are developed.
- Competition from newer antidepressants is offset by the off-label use trend.
- No significant patent protections or biosimilar developments are anticipated to impact pricing dynamics currently.
FAQs
1. Will trazodone's price increase with patent protection renewal?
No. The original patents expired decades ago, and no current patent protections are in place. Price increases from patent protection are unlikely.
2. How does off-label prescribing influence trazodone's market?
Off-label use accounts for most prescriptions, sustaining demand despite limited approved indications, which stabilizes the overall market volume.
3. Are there risks of generic price increases?
Minimal. The market has extensive generic competition, which caps price increases unless regulatory or formulation changes occur.
4. Could a new formulation alter the market?
Yes. An FDA-approved extended-release or combination formulation could temporarily raise prices, but regulatory approval is uncertain.
5. What competitive threats exist from newer antidepressants?
They offer better side effect profiles, but trazodone retains demand for sleep off-label, which diminishes competition significance.
References
[1] IBISWorld. (2022). Global antidepressant market report. Retrieved from https://www.ibisworld.com
[2] IQVIA. (2022). Prescription Data for antidepressants in the U.S.
[3] U.S. Food and Drug Administration. (2021). Patent and exclusivity data for trazodone.
[4] National Library of Medicine. (2022). Off-label prescription practices for trazodone.
[5] Statista. (2023). Average prices of generic antidepressants in the U.S.