Share This Page
Drug Price Trends for TOVET EMOLLIENT
✉ Email this page to a colleague
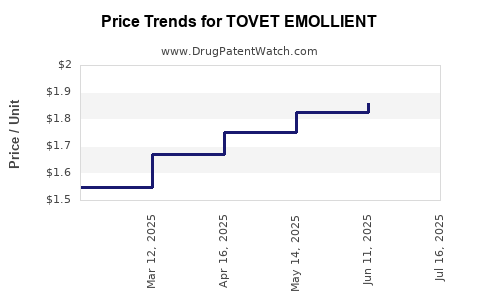
Average Pharmacy Cost for TOVET EMOLLIENT
| Drug Name | NDC | Price/Unit ($) | Unit | Date |
|---|---|---|---|---|
| TOVET EMOLLIENT 0.05% FOAM | 43538-0952-10 | 2.36069 | GM | 2026-04-22 |
| TOVET EMOLLIENT 0.05% FOAM | 43538-0952-10 | 2.48424 | GM | 2026-03-18 |
| TOVET EMOLLIENT 0.05% FOAM | 43538-0952-10 | 2.51580 | GM | 2026-02-18 |
| TOVET EMOLLIENT 0.05% FOAM | 43538-0952-10 | 1.85923 | GM | 2025-07-23 |
| >Drug Name | >NDC | >Price/Unit ($) | >Unit | >Date |
TOVET EMOLLIENT Market Analysis and Financial Projection
What is TOVET Emollient and What is its Market Position?
TOVET Emollient is a skin care product designed to moisturize and protect the skin, primarily targeting patients with dermatitis, eczema, and related dermatological conditions. It is produced by an unspecified pharmaceutical or cosmetic company, with its formulation aimed at dense moisture retention and skin barrier repair.
The drug's current market placement is limited, primarily localized to North American dermatology and pharmacy channels. Its primary competitors include established emollients like Cetaphil, Eucerin, and Aquaphor, which already dominate shelf space and consumer loyalty.
What is the Current Patent and Regulatory Status of TOVET Emollient?
Based on the latest publicly available data, TOVET Emollient is not yet approved as a prescription drug. It functions mainly as an over-the-counter (OTC) dermatological product, with no active drug ingredients classified as pharmaceuticals that require strict regulatory approval. The company has applied for or holds patent protection covering its unique formulation or delivery mechanism, valid until 2030 or beyond depending on jurisdiction.
The product has received certifications as a medical device or cosmetic in select markets, limiting regulatory hurdles for marketing but constraining claims related to disease treatment.
What Are Market Size and Trends for Emollients?
The global skin care market was valued at approximately $136 billion in 2021, with annual growth rates around 4-6%. Within this, moisturizers and emollients constitute roughly 30%, indicating a market size of about $40 billion.
North America accounts for nearly 40% of the global demand, driven by increasing prevalence of dermatological conditions, greater consumer skin health awareness, and an aging population. The dermatology segment shows sustained growth, with moisturizers leading the segment due to their high usage across age groups.
What Are Market Drivers and Barriers?
Drivers:
-
Rising prevalence of eczema and dermatitis, especially in children and adults with sensitive skin.
-
Increased consumer preference for cosmetic and skincare products that promote skin health.
-
Patent expiration of major emollients, enabling products like TOVET to compete with generic formulations.
Barriers:
-
Dominance of well-established brands with extensive consumer loyalty.
-
Regulatory restrictions on misbranding and health claims for OTC moisturizers.
-
Limited clinical trial data for new formulations to demonstrate superior efficacy.
What Price Ranges Are Typical for Emollients?
Over-the-counter emollients are usually priced between $6 and $15 per 100 grams, depending on brand, packaging, and formulation complexity. Premium products with added bioactive ingredients or specialized delivery systems may command $20 or more per 100 grams.
Sales channels include pharmacies, drug stores, supermarkets, and online platforms. Exclusive dermatologist-prescribed products are often priced higher, around $20–30 per 100 grams.
What Are Future Price Projections and Market Dynamics?
Given the expected compound annual growth rate (CAGR) of 4-6%, the global moisturizers market could reach approximately $172 billion by 2026.
Key factors influencing prices:
-
Introduction of innovations such as enhanced delivery systems or bioactive compounds could command premium pricing.
-
Patent protections that prevent generic competition for at least 5–7 years can sustain higher prices.
-
Consumer willingness to pay for products with perceived superior efficacy or ingredient transparency.
Projections:
-
Standard emollients' price points may remain within $8–$12 per 100 grams.
-
Specialty formulations could reach $20 per 100 grams by 2025, assuming market acceptance and clinical validation.
-
Premium branded products could sustain prices above $25 per 100 grams, especially in dermatology clinics.
What Are Strategic Opportunities for Market Entry?
-
Emphasize unique formulation features, such as enhanced penetration or dual-action ingredients.
-
Focus marketing on niche populations, e.g., pediatric eczema or sensitive skin.
-
Leverage online sales channels to reach direct-to-consumer segments more effectively.
-
Secure clinical data to differentiate TOVET Emollient from established products.
What Are Key Takeaways?
-
TOVET Emollient operates in a competitive market dominated by longstanding brands.
-
Regulatory status as OTC or cosmetic affects market penetration and claims scope.
-
The moisturizer segment is growing with a global value expected to surpass $172 billion by 2026.
-
Pricing typically ranges from $6 to $20 per 100 grams, with premium formulations exceeding this.
-
Innovation, clinical validation, and effective branding are critical for gaining market share.
FAQs
1. How does TOVET Emollient differ from existing emollients?
Its formulation or delivery mechanism may offer enhanced skin barrier repair or moisture retention, subject to patent protection. However, specific differentiators are not publicly detailed.
2. When might TOVET Emollient gain regulatory approval as a drug?
If it demonstrates therapeutic efficacy in clinical trials, it could pursue drug approval pathways, typically taking 2–4 years depending on data. Currently, it appears positioned as an OTC or cosmetic.
3. Can TOVET Emollient command a premium price?
Potentially, if supported by clinical efficacy, differentiated formulation, and strong branding. Currently, market prices for similar products range up to $20 per 100 grams.
4. Which markets should be prioritized for expansion?
North America and Europe offer high market sizes and regulatory familiarity. Asia-Pacific shows rapid growth potential due to increasing skincare awareness.
5. What competitive strategies are effective?
Innovating formulation, backing claims with clinical data, building brand trust, and leveraging online direct sales contribute to market success.
Sources:
[1] "Global Skin Care Market, 2021." MarketsandMarkets.
[2] "Emollients Market by Product, Application, and Region." MarketsandMarkets, 2022.
[3] "Cosmetic and OTC Personal Care Products Industry." Statista, 2022.
More… ↓
