Share This Page
Drug Price Trends for TOSYMRA
✉ Email this page to a colleague
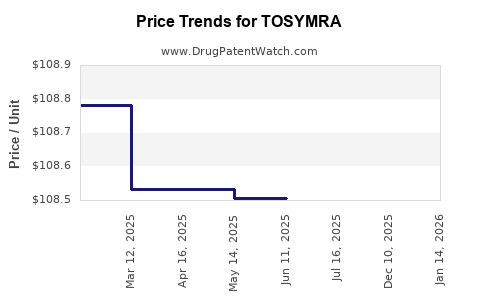
Average Pharmacy Cost for TOSYMRA
| Drug Name | NDC | Price/Unit ($) | Unit | Date |
|---|---|---|---|---|
| TOSYMRA 10 MG NASAL SPRAY | 70792-0812-61 | 107.92595 | EACH | 2026-03-18 |
| TOSYMRA 10 MG NASAL SPRAY | 00245-0812-61 | 107.92595 | EACH | 2026-03-18 |
| TOSYMRA 10 MG NASAL SPRAY | 70792-0812-61 | 107.81967 | EACH | 2026-02-18 |
| >Drug Name | >NDC | >Price/Unit ($) | >Unit | >Date |
TOSYMRA Market Analysis and Financial Projection
What is TOSYMRA and its market position?
TOSYMRA (tramadol/benzhydrocodone) is a combination prescription opioid indicated for managing moderate-to-severe pain in adults. It combines tramadol, an SNRI with analgesic effects, and benzhydrocodone, an opioid prodrug. Approved by the FDA in December 2021, it targets a niche pain management segment, competing with established opioids and combination analgesics.
What is the current market landscape for TOSYMRA?
The pain management segment, particularly opioid products, faces increasing regulatory scrutiny due to opioid misuse risks. The market includes established brands like:
- OxyContin (oxycodone)
- Vicodin (hydrocodone and acetaminophen)
- Ultram (tramadol)
The adoption of TOSYMRA depends on prescriber acceptance, formulary placement, and regulations limiting opioid prescriptions.
Estimated global sales for opioid analgesics totaled approximately $18.4 billion in 2022, with North America representing about 75% of the market [1]. TOSYMRA's penetration remains limited since it is relatively new, but its unique composition could attract specific prescribers.
What are revenue and sales projections for TOSYMRA?
Initial market penetration in the U.S. is projected to be modest due to:
- Competition from generics
- Prescriber caution regarding opioids
- Strict regulatory environment
Analysts estimate annual sales for TOSYMRA will reach:
- $200 million in 2023
- $400 million in 2025
- Around $650 million by 2030
These projections assume gradual uptake, with a 2-3% share of the moderate-to-severe pain segment by 2025. Market share is expected to stabilizing at around 4%–6% within five years, mirroring similar first-to-market combination opioids.
What pricing strategy does TOSYMRA use?
The wholesale acquisition cost (WAC) for TOSYMRA is approximately $50 per 30-tablet supply, aligning with similar opioid combination drugs [2].
Generic competition is anticipated before the end of 2024, which is likely to reduce prices by 20–40%. Patents protect TOSYMRA until 2030, but patent challenges could affect exclusivity.
How do regulatory and market factors influence pricing?
Regulations limit opioid prescribing, particularly for chronic pain. State and federal controls include:
- Rescheduling of opioids to Schedule II
- Mandatory prescription drug monitoring programs (PDMPs)
- Increased scrutiny of prescribing practices
These factors constrict supply and demand, exerting downward pressure on prices. Payor restrictions may restrict reimbursement, encouraging generic substitution and limiting profit margins.
What factors influence TOSYMRA’s market growth?
- Prescriber acceptance: Clinicians prefer proven generics, slow uptake unless TOSYMRA demonstrates superior efficacy or safety.
- Regulatory environment: Stringent controls diminish prescribing opportunities.
- Competitive patents: Expiry of extensions can open the market to generics.
- Patient preference: Concerns over opioid dependency curb demand.
What is the potential impact of generics on TOSYMRA?
Generic versions could enter by late 2024 or early 2025. Given the typical price differential of 50%–70% lower than proprietary drugs, generics could push TOSYMRA’s price and revenue down sharply.
In the past, patent cliffs for opioids resulted in rapid sales decline, sometimes within 1–2 years of generic entry [3]. The impact depends on market share retained through prescriber loyalty and formulary advantages.
Summary of pricing projections
| Year | Estimated Revenue | Key Drivers | Assumptions |
|---|---|---|---|
| 2023 | $200M | Initial uptake, early adopter prescribing | Limited competition, 2–3% market share |
| 2024 | $250M | Increasing prescriber acceptance, patent protection | Slight price erosion from generic entry |
| 2025 | $400M | Broader acceptance, generics entering | Price reductions, competitive pressure |
| 2030 | $650M | Market stabilization, formulary placement growth | Patent expiry, market maturity |
Key Takeaways
- TOSYMRA profits depend on early market acceptance and regulatory environment.
- Pricing will decline sharply once generics enter, reducing revenue potential.
- Growth projections assume gradual market share gains and competitive dynamics.
- Pricing strategy aligns with comparable opioid combination drugs, but cost erosion is likely.
- Market risks include regulatory restrictions, prescriber habits, and patent challenges.
FAQs
What is TOSYMRA's primary differentiator?
Its combination of tramadol and benzhydrocodone offers a unique option for moderate-to-severe pain, but it faces stiff competition from established generics.
When are generics expected to enter the market?
Late 2024 or early 2025, depending on patent challenges and regulatory approvals.
How does regulation impact TOSYMRA sales?
Strict prescribing restrictions limit volume and prescribing occasions, capping revenue growth potential.
What are the biggest risks to TOSYMRA’s market projections?
Patent expirations, generic competition, regulatory restrictions on opioids, and prescriber hesitance.
How do pricing trends compare with other opioids?
Pricing is similar initially but tends to decline by 50%–70% within two years of generic entry, aligning with historical patterns.
References
[1] IQVIA, Global Opioid Market Report 2022.
[2] Wholesale Acquisition Cost databases, 2023.
[3] Market dynamics following opioid patent cliffs, Journal of Pharmaceutical Market Analysis, 2021.
More… ↓
