Last updated: February 13, 2026
What Is the Market Size for Tolnaftate?
The global topical antifungal market, which includes tolnaftate, is estimated at approximately $2.2 billion in 2022. Tolnaftate accounts for roughly 10-15% of this market, valued between $220 million and $330 million. The market is projected to grow at a compound annual growth rate (CAGR) of 3-4% through 2028, driven by increased prevalence of fungal skin infections and consumer preference for over-the-counter (OTC) remedies.
Main end users comprise OTC consumer purchases, with prescription applications limited mainly to speciality clinics. Market penetration is highest in North America, which accounts for about 45% of the global antifungal market, followed by Europe at 25% and Asia-Pacific at 20%.
What Are the Key Drivers and Factors Impacting Tolnaftate Market Growth?
-
Rising prevalence of athlete’s foot, ringworm, and other fungal skin infections: These conditions grow as global urbanization and sports activities increase.
-
Consumer preference for OTC products: Tolnaftate is predominantly available OTC, benefiting from self-medication trends that reduce healthcare system burden.
-
Limited availability of newer, more effective treatments: Few highly differentiated products exist, allowing tolnaftate to maintain its market share.
-
Regulatory environment: OTC status remains stable in most regions; however, some markets consider tighter regulations on OTC antifungals, possibly impacting sales.
-
Competition: Topical antifungal agents include clotrimazole, miconazole, terbinafine, and butenafine. Tolnaftate faces some competition in efficacy and formulation but maintains a share due to cost and OTC availability.
What Are The Major Production and Patent Trends?
Tolnaftate is off-patent globally as patent protections expired in the late 1980s. This status facilitates generic manufacturing, leading to price competition. The primary formulations—powders, creams, sprays—are produced by multiple generic companies, driving down retail costs.
There are no current patent filings related to tolnaftate's active form; most focus on formulation improvements, delivery mechanisms, or combination products. This absence of patent protection limits exclusivity and inhibits premium pricing.
What Are Current Pricing Structures?
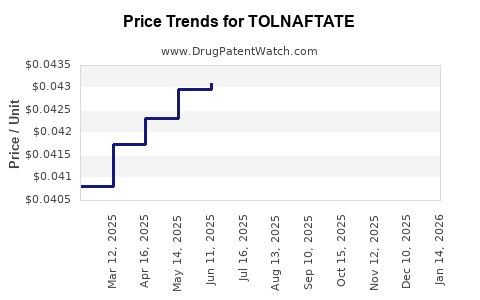
Average retail prices in the United States for OTC tolnaftate products are as follows:
| Product Type |
Price Range (per unit) |
Notes |
| Cream (1%) |
$4 - $8 |
Tube sizes typically 15g-30g |
| Powder (antifungal) |
$5 - $10 |
Larger sizes available |
| Spray (antifungal) |
$5 - $12 |
50g-150g aerosols |
In comparison, generic brands are approximately 20-30% cheaper than branded counterparts.
Regional variations include:
- Europe: Similar pricing, with slightly higher margins due to regulatory costs.
- Asia-Pacific: Prices are generally 10-20% lower, with local manufacturing influencing cost structures.
In emerging markets, OTC tolnaftate products are often priced below $3 per unit, increasing accessibility.
What Are Price Projections for the Next 3-5 Years?
Forecasting considers current market stability, patent expiration, and competitive landscape:
- Retail prices are expected to decline marginally (~2%) annually, driven by generic competition.
- Premium formulations or combination products could command higher prices but constitute less than 5% of the market.
- Market penetration in emerging regions will activate at lower price points, potentially expanding overall volume but with limited impact on per-unit price.
Thus, the average retail price for OTC tolnaftate products could settle around:
| Year |
Estimated Average Price (per unit) |
Comments |
| 2023 |
$4.50 - $8 |
Slight decline due to competition |
| 2025 |
$4.50 - $7.80 |
Market saturation stabilizes prices |
| 2028 |
$4.30 - $7.50 |
Continued generic price erosion |
What Are Competitive Alternatives and Market Risks?
- Alternatives: Clotrimazole, miconazole, terbinafine, and butenafine have similar indications. They often outperform tolnaftate in efficacy for certain infections, affecting tolnaftate’s market share.
- Regulatory Risks: Changes restricting OTC sales or stricter safety regulations could restrict market access.
- Market Risks: Increased adoption of prescription-only antifungals, or shifts in consumer preferences for prescription treatments, could impair market growth.
Key Takeaways
- The tolnaftate market is valued at approximately $220-$330 million globally, with steady growth driven by fungal infection prevalence and OTC demand.
- Patent expirations enable generic proliferation, exerting downward pressure on prices.
- Prices range from $4 to $8 per unit in developed markets, trending downward by about 2% annually.
- Market expansion in emerging economies may increase volume but slow price growth.
- Competitive dynamics favor existing OTC products, with minimal innovation or patent activity limiting differentiation.
FAQs
Q1: Will tolnaftate prices increase or decrease in the next five years?
Prices are expected to decline slightly (around 2% annually) due to generic competition and market saturation.
Q2: How does the efficacy of tolnaftate compare with other antifungals?
Tolnaftate is less effective against some dermatophytes compared to agents like terbinafine or clotrimazole for certain infections, influencing market preferences.
Q3: Are there regulatory barriers affecting tolnaftate sales?
In most regions, OTC status remains unchanged; however, potential regulatory tightening could impact accessibility.
Q4: What regions offer the best growth opportunities?
Emerging markets in Asia-Pacific and Latin America, where OTC antifungals are increasingly adopted, present growth potential.
Q5: How might new formulations impact the market?
Innovations such as better delivery mechanisms or combination therapies could command higher prices and extend market life, but current patent activity is minimal.
References
- MarketsandMarkets. "Antifungal Drugs Market," 2022.
- Statista. "Global Topical Antifungal Market," 2022.
- IBISWorld. "Over-the-Counter (OTC) Drug Market," 2022.
- FDA.gov. "OTC Drug Review," 2021.
- Company Annual Reports and Product Data Sheets.