Share This Page
Drug Price Trends for TOBRAMYCIN-DEXAMETH OPHTH SUSP
✉ Email this page to a colleague
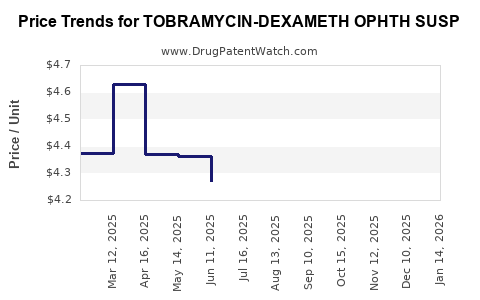
Average Pharmacy Cost for TOBRAMYCIN-DEXAMETH OPHTH SUSP
| Drug Name | NDC | Price/Unit ($) | Unit | Date |
|---|---|---|---|---|
| TOBRAMYCIN-DEXAMETH OPHTH SUSP | 61314-0647-25 | 5.31734 | ML | 2026-05-20 |
| TOBRAMYCIN-DEXAMETH OPHTH SUSP | 00574-4031-05 | 4.56434 | ML | 2026-05-20 |
| TOBRAMYCIN-DEXAMETH OPHTH SUSP | 00574-4031-10 | 4.96437 | ML | 2026-05-20 |
| TOBRAMYCIN-DEXAMETH OPHTH SUSP | 24208-0295-05 | 4.56434 | ML | 2026-05-20 |
| >Drug Name | >NDC | >Price/Unit ($) | >Unit | >Date |
Market analysis and price projections for TOBRAMYCIN-DEXAMETH OPHTH SUSP
Market Overview
Tobramycin-Dexameth Ophthalmic Suspension (TOBRAMYCIN-DEXAMETH) is a combination antibiotic and anti-inflammatory eye drop used primarily for bacterial conjunctivitis and keratitis. It is marketed under various generic and brand names globally.
The global ophthalmic antibiotics market reached approximately $5.2 billion in 2022, with a compounded annual growth rate (CAGR) of 4.5% expected through 2030 [1]. Within this, the segment for combination drugs—especially antibiotics with anti-inflammatory agents—is expanding due to increasing prevalence of ocular infections and rising demand for multi-mechanism treatments.
Market Drivers
- Rising incidence of bacterial ocular infections, driven by increased outdoor activities and urbanization.
- Growing aging population with higher susceptibility to ocular conditions.
- Drug resistance concerns shifting prescribing patterns towards combination therapies.
- Regulatory approvals for new formulations or improved delivery mechanisms.
Market Challenges
- Competition from monotherapies and alternative combination drugs.
- Cost sensitivity in emerging markets.
- Patent expiration of some formulations, increasing availability of generics.
Competitive Landscape
Several formulations of tobromycin-DEXAMETH are available globally, both as branded and generic products. Key players include:
- Alcon
- Sandoz (Novartis)
- Sun Pharma
- Teva Pharmaceuticals
- Several regional generic manufacturers
Branded formulations like "Tobradex" have dominated in certain markets, but generic versions have increased accessibility.
Patent Status
Most formulations have lost patent protection or are nearing expiry, leading to a surging availability of generics [2]. This enhances price competition but may impact revenues for originators.
Pricing Analysis
Current Price Range (US Market)
- Branded Tobradex: $20 - $40 per 10 mL bottle.
- Generic Tobramycin-Dexameth: $10 - $20 per 10 mL bottle.
Pricing Factors
- Market maturity
- Regulatory environment
- Brand recognition
- Packaging and formulation innovations
Cost Dynamics
Manufacturing costs are reduced for generics due to simplified regulatory pathways. Contract manufacturing organizations (CMOs) report production costs of approximately $2 - $4 per 10 mL unit, depending on scale and quality standards [3].
Price Projections (Next 5 Years)
- 2023-2025: Prices stabilize at $10 - $20 for generics in major markets, with minor fluctuations due to production costs and competition.
- 2026-2030: Assume increased competition leads to further price erosion. Expect prices to decline 10-15% annually, reaching ~$8 - $15 per 10 mL in the US and Europe.
Market Penetration and Pricing in Emerging Markets
- Lower prices: $2 - $10 per 10 mL.
- Growth facilitated by local manufacturing and regulatory approvals.
Impact of Formulation Improvements
- Sustained market value may depend more on brand loyalty, delivery improvements, or combination with sustained-release mechanisms rather than price alone.
Key Market Trends
- Increased adoption of generic formulations reduces prices.
- Emerging markets experience more dramatic price decreases due to lower healthcare budgets.
- Potential for biosimilar or novel delivery formats to influence future pricing strategies.
Summary
The market for Tobramycin-Dexameth ophthalmic suspension is characterized by declining prices driven by patent expirations and generic competition. Short-term price stability for branded versions exists, but over the next five years, prices are likely to decline steadily, especially with increasing regional competition. Revenue potential will hinge on geographic market dynamics, formulation innovations, and regulatory landscapes.
Key Takeaways
- The global ophthalmic antibiotics market is growing, with combination drugs like Tobramycin-Dexameth maintaining a substantial share.
- Patent expiries are catalyzing price reductions through increased generic competition.
- US marketed generics are priced between $10 and $20, with further declines projected.
- Emerging markets display more significant price erosion but offer growth opportunities.
- Innovation in delivery methods could sustain pricing power longer.
FAQs
1. What is the primary use of Tobramycin-Dexameth ophthalmic suspension?
It treats bacterial eye infections accompanied by inflammation, such as conjunctivitis and keratitis.
2. How does patent expiration impact the drug’s price?
Patent expiry introduces generic competitors, leading to a significant price decrease due to competition.
3. Are branded versions more expensive than generics?
Yes. Branded Tobradex can cost twice as much as generic Tobramycin-Dexameth formulations.
4. What regions are seeing the fastest price declines?
Emerging markets experience more rapid price erosion, often due to local manufacturing and regulatory approvals.
5. Is there room for price increases in the future?
Limited, unless new formulations, delivery innovations, or indications extend market exclusivity.
References
[1] Market Research Future. “Global Ophthalmic Drugs Market Report,” 2022.
[2] U.S. Patent and Trademark Office. “Patent status of Tobramycin formulations,” 2023.
[3] Contract Manufacturing Organization (CMO) Reports, 2022.
More… ↓
