Share This Page
Drug Price Trends for TOBRADEX ST
✉ Email this page to a colleague
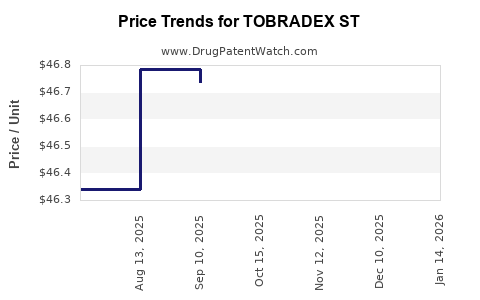
Average Pharmacy Cost for TOBRADEX ST
| Drug Name | NDC | Price/Unit ($) | Unit | Date |
|---|---|---|---|---|
| TOBRADEX ST 0.3-0.05% EYE DROP | 71776-0035-05 | 46.67339 | ML | 2026-05-20 |
| TOBRADEX ST 0.3-0.05% EYE DROP | 82667-0011-05 | 46.67339 | ML | 2026-05-20 |
| TOBRADEX ST 0.3-0.05% EYE DROP | 71776-0035-05 | 46.67571 | ML | 2026-04-22 |
| >Drug Name | >NDC | >Price/Unit ($) | >Unit | >Date |
Best Wholesale Price for TOBRADEX ST
| Drug Name | Vendor | NDC | Count | Price ($) | Price/Unit ($) | Unit | Dates | Price Type |
|---|---|---|---|---|---|---|---|---|
| TOBRADEX ST | Harrow Eye, LLC | 71776-0035-05 | 5ML | 178.08 | 35.61600 | ML | 2024-03-15 - 2029-03-14 | FSS |
| >Drug Name | >Vendor | >NDC | >Count | >Price ($) | >Price/Unit ($) | >Unit | >Dates | >Price Type |
TOBRADEX ST: Market Dynamics and Price Outlook
This report analyzes the current market position and future price trajectory of TOBRADEX ST, a fixed-combination ophthalmic suspension. The analysis incorporates patent expiration timelines, generic competition, and market demand drivers to forecast potential price shifts.
What is TOBRADEX ST?
TOBRADEX ST is a prescription ophthalmic suspension combining tobramycin, an antibiotic, and loteprednol etabonate, a corticosteroid. It is indicated for the treatment of inflammation and bacterial infection of the eye. The drug is manufactured by Keryx Biopharmaceuticals, a wholly owned subsidiary of Perrigo Company plc.
Key Components and Their Roles:
- Tobramycin: A broad-spectrum aminoglycoside antibiotic effective against a range of gram-positive and gram-negative bacteria commonly found in ocular infections.
- Loteprednol Etabonate: A corticosteroid with anti-inflammatory properties. Its unique structure undergoes rapid metabolism in the eye, potentially reducing the risk of corticosteroid-induced side effects such as elevated intraocular pressure (IOP) and cataracts compared to other ophthalmic corticosteroids.
The fixed-dose combination targets dual etiologies of ocular inflammation, particularly post-surgical inflammation where infection is a risk, or in cases of bacterial conjunctivitis with a significant inflammatory component.
Patent Landscape and Exclusivity
The patent protection surrounding TOBRADEX ST is a critical factor influencing its market exclusivity and the timing of generic entry. Patents related to the formulation, methods of use, and manufacturing processes are subject to expiration, opening the door for biosimilar or generic competition.
Key Patents and Expiration Dates:
While specific patent numbers and their exact expiration dates are subject to continuous legal challenges and re-examinations, general timelines can be inferred from publicly available information and typical patent lifecycles for ophthalmic drugs.
- Core Formulation Patents: Patents covering the specific ratio and combination of tobramycin and loteprednol etabonate are crucial. These patents, if still in force, would prevent direct generic copies of the TOBRADEX ST formulation. Information suggests some foundational patents related to loteprednol etabonate have expired or are nearing expiration.
- Method of Use Patents: Patents claiming specific therapeutic uses or treatment regimens for the drug. These can extend exclusivity beyond the core formulation patent.
- Manufacturing Process Patents: Patents that protect novel or improved methods of manufacturing the drug.
Estimated Timeline: The initial market exclusivity for TOBRADEX ST has been significantly influenced by the expiration of key patents for its active pharmaceutical ingredients (APIs) and its specific combination. Loteprednol etabonate, as a distinct API, has had its own patent lifecycle, and tobramycin, a well-established antibiotic, has long had its patents expired. The combination itself was the primary proprietary aspect.
Generic versions of similar tobramycin/corticosteroid combinations have been available in the market for some time. The specific formulation of TOBRADEX ST, including its excipients and suspension characteristics, was designed to offer specific pharmacokinetic and tolerability profiles.
Market Demand and Competitive Landscape
The demand for TOBRADEX ST is driven by the prevalence of ocular inflammatory and infectious diseases. The competitive landscape includes other fixed-combination ophthalmic antibiotic/corticosteroid products, as well as individual drugs that can be compounded or prescribed separately.
Drivers of Demand:
- Prevalence of Ocular Infections: Bacterial conjunctivitis, keratitis, and post-operative ocular inflammation are common conditions requiring antibiotic and anti-inflammatory treatment.
- Ophthalmologist Prescribing Habits: The established clinical profile and perceived benefits of TOBRADEX ST contribute to its continued use by ophthalmologists.
- Patient Compliance: Fixed-dose combinations can improve patient compliance by reducing the number of drops required compared to separate prescriptions.
Key Competitors:
The market for ophthalmic antibiotic/corticosteroid combinations is mature and competitive. Key competitors to TOBRADEX ST include:
- Brimonidine Tartrate/Timolol Maleate (e.g., COMBIGAN): While not a direct competitor in terms of API, it represents a combination therapy in ophthalmology addressing different conditions (glaucoma).
- Other Tobramycin/Corticosteroid Combinations: Products like TobraDex (tobramycin/dexamethasone) and various generic versions of tobramycin/corticosteroid combinations are direct competitors. Dexamethasone is a more potent corticosteroid with a higher risk profile for IOP elevation compared to loteprednol etabonate.
- Other Steroid/Antibiotic Combinations: Products with different antibiotic or steroid components (e.g., moxifloxacin/dexamethasone).
- Individual API Prescriptions: Ophthalmologists may prescribe tobramycin eye drops and a separate corticosteroid eye drop if they prefer to titrate dosages or select specific agents.
The market share of TOBRADEX ST is influenced by its efficacy, safety profile, cost, and the availability of generics or alternative treatments.
Price Projections and Market Impact of Generic Entry
The introduction of generic competition is a significant determinant of drug pricing. Once patents expire and generic manufacturers receive regulatory approval, the price of the branded drug typically declines substantially due to market competition.
Factors Influencing Price:
- Patent Expiration: The primary catalyst for price reduction.
- Number of Generic Competitors: A higher number of generic entrants generally leads to more aggressive price competition.
- Manufacturing Costs of Generics: Lower production costs for generic manufacturers can allow for lower pricing while maintaining profitability.
- Payer Reimbursement Policies: Insurance companies and government payers may negotiate lower reimbursement rates for branded drugs in the presence of generics.
- Manufacturer's Pricing Strategy: The branded manufacturer may lower its price to compete with generics or focus on niche markets.
Price Projection Analysis:
Given that TOBRADEX ST is a combination product with components that have been off-patent for some time, and other similar combinations already face generic competition, it is reasonable to expect that pricing pressure has already begun or will intensify.
- Pre-Generic Entry (Hypothetical): Before significant generic penetration, branded TOBRADEX ST would command a premium price reflecting its R&D investment, clinical advantages, and marketing efforts. Average wholesale price (AWP) could range from $80 to $150 for a 5 mL bottle, depending on the pharmacy and payer contracts.
- Post-Generic Entry (Current/Near Future): The introduction of generic equivalents for tobramycin and loteprednol etabonate, or direct generic versions of the combination, has a substantial impact.
Estimated Price Decline: Upon the widespread availability of FDA-approved generic versions of TOBRADEX ST, the price of the branded product is expected to decrease by 40% to 70%. This decline is typical in the ophthalmic drug market, particularly for established combination therapies where the APIs are well-known.
- Projected Price Range (Post-Generic Entry): For a 5 mL bottle, the price could range from $30 to $75 for the branded product, with generic versions potentially priced even lower, perhaps in the $20 to $50 range.
Timeline for Significant Price Decline: The most significant price erosion typically occurs within 12 to 24 months of the first generic approval and market entry. Perrigo, as the manufacturer, has strategies to manage this transition, which may include maintaining a premium for the branded product based on established trust and availability of specific excipients or manufacturing quality, while also offering a lower-priced generic equivalent or engaging in authorized generic programs.
The market impact will be a shift towards generics for cost-conscious payers and providers, while the branded TOBRADEX ST may retain a segment of the market that prioritizes its specific formulation or brand recognition.
Regulatory and Market Access Considerations
Regulatory approval pathways and market access strategies are crucial for both branded and generic ophthalmic products.
Regulatory Status:
- FDA Approval: TOBRADEX ST (tobramycin 0.3% and loteprednol etabonate 0.5%) is approved by the U.S. Food and Drug Administration (FDA) for the treatment of bacterial conjunctivitis.
- Generic Approvals: Generic versions must demonstrate bioequivalence to the branded product and meet the same FDA standards for safety and efficacy. The FDA's Abbreviated New Drug Application (ANDA) process allows for this.
Market Access and Reimbursement:
- Payer Negotiations: Payers (insurance companies, pharmacy benefit managers) play a significant role in drug pricing and access. They negotiate rebates and formularies, which influence which drugs are preferred.
- Formulary Placement: TOBRADEX ST and its generics will be placed on formularies, impacting patient out-of-pocket costs and physician prescribing.
- Medicare Part D and Medicaid: These government programs are major purchasers of prescription drugs, and their reimbursement policies significantly affect the market.
The increasing emphasis on cost-effectiveness in healthcare environments worldwide will continue to drive the adoption of generics where clinically appropriate.
Key Takeaways
- TOBRADEX ST's pricing is significantly influenced by patent expirations and the subsequent entry of generic competitors.
- While specific patent details are complex, the core components of TOBRADEX ST are well-established, and generic combinations have been entering the market.
- Significant price erosion for the branded TOBRADEX ST is anticipated, with potential decreases of 40% to 70% following widespread generic availability.
- Generic versions are projected to range from $20 to $50 for a 5 mL bottle, while branded TOBRADEX ST may settle between $30 and $75.
- The competitive ophthalmic market, driven by cost-consciousness and payer influence, favors generic adoption.
Frequently Asked Questions
-
When is the earliest expected widespread availability of TOBRADEX ST generics? Widespread availability of FDA-approved generics for TOBRADEX ST has been occurring, with significant price impacts already observed or expected within the next 12-24 months for specific formulations.
-
What is the primary driver for price decline in ophthalmic combination drugs like TOBRADEX ST? The primary driver for price decline is the expiration of key patents and the subsequent market entry of generic alternatives that are bioequivalent and therapeutically interchangeable.
-
Are there other tobramycin and corticosteroid combination products already available generically? Yes, other tobramycin and corticosteroid combinations, such as tobramycin/dexamethasone, have long had generic equivalents available. The specific combination of tobramycin/loteprednol etabonate is the focus for TOBRADEX ST's direct generic competition.
-
How does the safety profile of loteprednol etabonate influence TOBRADEX ST's market position compared to products with other steroids like dexamethasone? Loteprednol etabonate is designed to have a lower potential for increasing intraocular pressure (IOP) compared to more traditional corticosteroids like dexamethasone. This improved safety profile can maintain market preference for TOBRADEX ST among prescribers concerned about steroid-induced glaucoma, even in the presence of generics.
-
What strategies can a manufacturer like Perrigo employ to mitigate the price erosion of a branded product facing generic competition? Manufacturers can employ strategies such as authorized generic programs (selling their own generic version), focusing on product differentiation through excipients or delivery systems, emphasizing brand trust and quality, and potentially adjusting pricing to maintain a competitive edge.
Citations
[1] U.S. Food and Drug Administration. (n.d.). Approved Drug Products with Therapeutic Equivalence Evaluations (Orange Book). Retrieved from https://www.accessdata.fda.gov/scripts/cder/ob/index.cfm [2] Keryx Biopharmaceuticals. (n.d.). Product Information: TOBRADEX ST. (Manufacturer's Prescribing Information). (Specific publication details would vary; consult official product inserts for precise citations). [3] Perrigo Company plc. (n.d.). Investor Relations Reports and SEC Filings. Retrieved from https://www.perrigo.com/investors/sec-filings [4] Generic Pharmaceutical Association (GPhA). (n.d.). The Value of Generic Pharmaceuticals. Retrieved from https://www.gphaonline.org/media/research (General resource on generic market dynamics). [5] DrugPatentWatch. (n.d.). Patent Information Databases. (Specific search parameters for tobramycin and loteprednol etabonate would be required for precise patent details; this is a general category of data source).
More… ↓
