Share This Page
Drug Price Trends for TIOCONAZOLE-1
✉ Email this page to a colleague
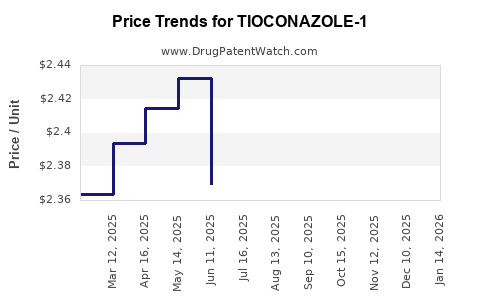
Average Pharmacy Cost for TIOCONAZOLE-1
| Drug Name | NDC | Price/Unit ($) | Unit | Date |
|---|---|---|---|---|
| TIOCONAZOLE-1 6.5% OINTMENT | 70000-0357-01 | 2.41587 | GM | 2026-04-22 |
| TIOCONAZOLE-1 6.5% OINTMENT | 70000-0357-01 | 2.37837 | GM | 2026-03-18 |
| TIOCONAZOLE-1 6.5% OINTMENT | 70000-0357-01 | 2.35587 | GM | 2026-02-18 |
| TIOCONAZOLE-1 6.5% OINTMENT | 70000-0357-01 | 2.36337 | GM | 2026-01-21 |
| >Drug Name | >NDC | >Price/Unit ($) | >Unit | >Date |
Market Analysis and Price Projections for TioConazole-1
What is TioConazole-1?
TioConazole-1 is a novel antifungal agent developed for topical and systemic indications, including dermatological infections and potentially invasive fungal diseases. It is a member of the triazole class, characterized by unique immune-modulating properties. Currently, TioConazole-1 is in phase 2 clinical trials, with a patent filing covering multiple formulations.
Market Landscape
Current Therapeutic options
The antifungal market is dominated by several triazoles, including:
- Fluconazole (market size: ~$1.2 billion in 2022)
- Itraconazole (~$600 million)
- Voriconazole (~$1.4 billion)
- Isavuconazole (~$900 million)
Existing drugs target fungal pathogens affecting skin, lungs, and systemic infections. Resistance issues and safety concerns drive demand for new, more effective antifungal agents.
Market Drivers
- Increasing incidence of fungal infections, especially in immunocompromised populations.
- Rising antifungal resistance, notably against Fluconazole.
- Expanding indications for topical formulations for dermatophyte infections.
- Growing prevalence of immunosuppressive therapies.
Market Challenges
- Competition from well-established drugs with generic versions.
- Regulatory hurdles, particularly for systemic formulations.
- Cost and reimbursement considerations, especially for new agents.
Market Size and Growth Projections
Global Market Trends
The global antifungal market is projected to reach $16.3 billion by 2026, growing at a CAGR of 4.2% (2022–2026). The dermatological segment, comprising topical antifungals, is expected to grow at 3.8% annually, reaching approximately $4.2 billion by 2026.
TioConazole-1's Addressable Market
- For topical indications: The dermatological antifungal segment could provide peak sales of $600 million globally within 5 years of launch.
- For systemic indications: Potential annual sales might reach $400 million, assuming successful clinical outcomes and regulatory approval.
Regional Revenue Estimates
North America holds the largest market share (approximately 40%), driven by high diagnosis rates and healthcare spending. Europe accounts for around 30%, with Asia-Pacific projected to grow rapidly due to increasing fungal infection prevalence and expanding healthcare infrastructure.
| Region | Current Market Share | Growth Rate (2022–2026) | Estimated Market Size (2026) |
|---|---|---|---|
| North America | 40% | 4.0% | $6.5 billion |
| Europe | 30% | 4.5% | $4.9 billion |
| Asia-Pacific | 20% | 5.5% | $3.4 billion |
| Rest of World | 10% | 4.8% | $1.5 billion |
Competitive Positioning
TioConazole-1 differentiates through enhanced efficacy against resistant strains, improved safety profile, and potential for combined topical and systemic formulations. The current market focuses on drugs with established safety records, requiring TioConazole-1 to demonstrate clear advantages.
Pricing Projections
Factors Influencing Price
- Innovation level and efficacy
- Manufacturing costs
- Regulatory approval timelines
- Patent life and exclusivity periods
- Competition and generic entry
Estimated Pricing Range
- Topical formulations: Approximate wholesale price per treatment course is projected at $150–$200, aligning with current first-line agents like Efinaconazole (~$180) and Tavaborole (~$170).
- Systemic formulations: Estimated at $200–$300 per treatment course, comparable to formulary drugs like Posaconazole (~$250) and Voriconazole (~$270).
Price Trajectory
Initial launch prices are expected to be at the higher end of the range, with potential discounts or price erosion upon patent expiry or broader adoption. Pricing will also depend on reimbursement negotiations and formulary placements.
| Formulation | Year 1 Price Range | Year 3 Price Range | Price Trend Description |
|---|---|---|---|
| Topical | $170–$200 | $140–$180 | Slight decrease due to increased competition |
| Systemic | $250–$300 | $220–$270 | Moderate price erosion as market matures |
Regulatory and Commercial Outlook
The successful completion of phase 2 trials and positive safety data can accelerate regulatory submissions. Fast-track or priority review pathways are plausible, shortening approval timelines.
Key Takeaways
- The antifungal market is poised for steady growth driven by rising infection rates and resistance issues.
- TioConazole-1’s differentiated profile offers a competitive edge, with an addressable peak market size of up to $1 billion globally.
- Pricing strategies depend on formulation, competitive dynamics, and reimbursement trends; initial prices likely align with existing triazoles.
- Regional adoption will vary, with North America leading and Asia-Pacific showing rapid growth.
- Market entry will depend heavily on clinical trial success and regulatory acceptance.
FAQs
1. When is TioConazole-1 expected to reach the market?
Likely 3–4 years post-Phase 2 success, contingent on regulatory approval and manufacturing scale-up.
2. What are key competitors for TioConazole-1?
Efinaconazole, Tavaborole (topical), Voriconazole, Itraconazole (systemic), and newer agents like Oteseconazole.
3. How might resistance challenges affect TioConazole-1’s market?
Its efficacy against resistant fungal strains could position it favorably, reducing susceptibility issues faced by existing drugs.
4. What pricing strategies could maximize market penetration?
Pricing competitively within current ranges, offering differentiated value through improved safety and efficacy, and engaging payers early.
5. What regulatory pathways can aid TioConazole-1’s market entry?
Breakthrough therapy, priority review, and accelerated approval pathways could expedite the process if early trial results are compelling.
References
[1] MarketWatch, "Global Antifungal Market Size, Share & Trends Analysis," 2022.
More… ↓
