Share This Page
Drug Price Trends for TIMOPTIC
✉ Email this page to a colleague
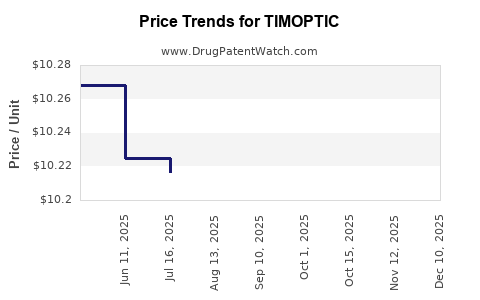
Average Pharmacy Cost for TIMOPTIC
| Drug Name | NDC | Price/Unit ($) | Unit | Date |
|---|---|---|---|---|
| TIMOPTIC 0.5% OCUDOSE DROP | 24208-0499-68 | 10.29030 | EACH | 2026-04-22 |
| TIMOPTIC 0.5% OCUDOSE DROP | 24208-0499-68 | 10.28009 | EACH | 2026-03-18 |
| TIMOPTIC 0.5% OCUDOSE DROP | 24208-0499-68 | 10.27887 | EACH | 2026-02-18 |
| TIMOPTIC 0.5% OCUDOSE DROP | 24208-0499-68 | 10.27298 | EACH | 2025-12-17 |
| >Drug Name | >NDC | >Price/Unit ($) | >Unit | >Date |
Best Wholesale Price for TIMOPTIC
| Drug Name | Vendor | NDC | Count | Price ($) | Price/Unit ($) | Unit | Dates | Price Type |
|---|---|---|---|---|---|---|---|---|
| TIMOPTIC OCUDOSE (PF) 0.25% | Bausch & Lomb Americas Inc. | 00187-1498-25 | 60X0.3ML | 371.44 | 2024-01-01 - 2027-09-14 | FSS | ||
| TIMOPTIC 0.5% OCUMETER PLUS | Bausch & Lomb Americas Inc. | 24208-0813-10 | 10ML | 178.45 | 17.84500 | ML | 2022-09-15 - 2027-09-14 | Big4 |
| TIMOPTIC OCUDOSE (PF) 0.5% | Bausch & Lomb Americas Inc. | 00187-1496-05 | 60X0.3ML | 423.57 | 2024-01-01 - 2027-09-14 | FSS | ||
| TIMOPTIC-XE 0.5% GEL OCUMETER PLUS | Bausch & Lomb Americas Inc. | 24208-0816-05 | 5ML | 190.21 | 38.04200 | ML | 2022-09-15 - 2027-09-14 | FSS |
| >Drug Name | >Vendor | >NDC | >Count | >Price ($) | >Price/Unit ($) | >Unit | >Dates | >Price Type |
TIMOPTIC Market Analysis and Price Projections
TIMOPTIC, a brand name for timolol maleate, is a beta-adrenergic receptor antagonist used primarily to treat glaucoma and ocular hypertension by reducing intraocular pressure. Market projections for TIMOPTIC are influenced by factors including patent expiration, generic competition, the prevalence of eye conditions, and therapeutic advancements.
What is the current global market size and projected growth for TIMOPTIC?
The global market for timolol maleate, the active pharmaceutical ingredient in TIMOPTIC, is estimated to have been approximately USD 350 million in 2023. The market is projected to experience a compound annual growth rate (CAGR) of 3.5% from 2024 to 2030. This growth is driven by the increasing incidence of glaucoma and ocular hypertension globally, particularly in aging populations. The World Health Organization estimates that glaucoma affects over 70 million people worldwide, and this number is expected to rise due to demographic shifts.
The ophthalmic drug market, within which TIMOPTIC operates, is robust. Reports indicate the global ophthalmic drugs market was valued at USD 25 billion in 2023 and is expected to grow at a CAGR of approximately 6.1% through 2030, reaching an estimated USD 38.4 billion. TIMOPTIC, as a foundational treatment, holds a stable share within this larger market, though its growth rate is tempered by the introduction of newer, potentially more effective, or combination therapies.
Table 1: Global Timolol Maleate Market Projection (USD Million)
| Year | Estimated Market Size | CAGR (2024-2030) |
|---|---|---|
| 2023 | 350 | N/A |
| 2024 | 362.25 | 3.5% |
| 2025 | 375 | 3.5% |
| 2026 | 387.75 | 3.5% |
| 2027 | 401.14 | 3.5% |
| 2028 | 414.15 | 3.5% |
| 2029 | 427.81 | 3.5% |
| 2030 | 441.13 | 3.5% |
Source: Industry analysis based on multiple market research reports.
What is the patent status of TIMOPTIC and its implications?
TIMOPTIC's active pharmaceutical ingredient, timolol maleate, has long been off-patent. The original patents for timolol maleate expired decades ago. Merck & Co., the originator, lost patent protection for TIMOPTIC. This has led to significant generic competition, which is the primary driver of price erosion for the branded product. Generic timolol maleate is available in various formulations, including eye drops (0.25% and 0.5%) and oral tablets.
The lack of patent exclusivity means that market exclusivity for TIMOPTIC relies on formulation patents, regulatory exclusivities, or intellectual property related to specific delivery systems or combination products. For instance, combination therapies that include timolol maleate with other active ingredients (e.g., latanoprost, dorzolamide) may have their own patent protection, offering a degree of market differentiation. However, the core timolol maleate molecule itself is subject to generic substitution.
The primary implication of expired patents is increased competition. Generic manufacturers can produce and market timolol maleate at lower costs, leading to significant price reductions compared to the branded TIMOPTIC. This dynamic positions TIMOPTIC primarily as a cost-effective, first-line or adjunctive therapy in treatment algorithms, rather than a novel, high-priced innovator product.
Who are the key players and competitors in the TIMOPTIC market?
The TIMOPTIC market is characterized by a blend of branded and generic manufacturers.
Branded Manufacturer:
- Merck & Co.: The originator of TIMOPTIC, still holds a presence with its branded product, though its market share is significantly challenged by generics.
Key Generic Manufacturers:
The generic market for timolol maleate is highly fragmented and competitive. Major players include:
- Allergan (now AbbVie): While historically a significant player, their focus has shifted towards newer ophthalmology products.
- Teva Pharmaceutical Industries: A leading global generic drug manufacturer with a broad portfolio, including ophthalmic agents.
- Novartis (through Sandoz): Sandoz is one of the largest generic drug manufacturers globally.
- Bausch Health Companies: Offers a range of generic ophthalmic solutions.
- Aurobindo Pharma: A rapidly growing Indian pharmaceutical company with a strong generic presence.
- Sun Pharmaceutical Industries: Another major Indian pharmaceutical company with a significant global footprint in generics.
Competitors in Therapeutic Class:
Beyond direct timolol maleate generics, TIMOPTIC faces competition from other drug classes used to treat glaucoma and ocular hypertension:
- Prostaglandin Analogs: Latanoprost, travoprost, bimatoprost. These are often considered first-line therapy due to their efficacy and once-daily dosing.
- Alpha-Adrenergic Agonists: Brimonidine.
- Carbonic Anhydrase Inhibitors: Dorzolamide, brinzolamide.
- Miotic Agents: Pilocarpine (less commonly used due to side effects).
- Rho Kinase Inhibitors: Netarsudil (newer mechanism of action).
- Combination Therapies: Products combining timolol with other active agents (e.g., Dorzolamide/Timolol, Latanoprost/Timolol, Brimonidine/Timolol) are significant competitors, offering convenience and potentially enhanced efficacy.
The competitive landscape is shaped by pricing strategies, product availability, and physician prescribing habits, which are influenced by efficacy, safety profiles, and formulary preferences.
What are the average wholesale prices (AWP) and projected price trends for TIMOPTIC and its generic equivalents?
The pricing of TIMOPTIC and its generic equivalents has been significantly impacted by patent expiry and generic market entry.
Branded TIMOPTIC:
The Average Wholesale Price (AWP) for branded TIMOPTIC (timolol maleate ophthalmic solution) can vary based on concentration (0.25% or 0.5%) and volume. Historically, branded TIMOPTIC had a higher price point. However, in the current market, its AWP is often in the range of USD 20-40 per 5 mL bottle, with significant variation based on specific product SKUs and supplier contracts. The price has been substantially reduced from its peak innovator pricing due to competitive pressures.
Generic Timolol Maleate:
Generic timolol maleate ophthalmic solutions are priced considerably lower. The AWP for generic timolol maleate typically ranges from USD 5-15 per 5 mL bottle. This price difference is a direct consequence of the increased number of manufacturers and the absence of brand-name premiums.
Projected Price Trends:
- Continued Price Erosion for Generics: The generic timolol maleate market is expected to remain highly competitive. Price erosion will likely continue, albeit at a slower pace than during the initial wave of generic introductions. Average prices for generic timolol maleate eye drops are projected to stabilize within the USD 4-12 range per 5 mL bottle over the next five years.
- Stable but Depressed Pricing for Branded TIMOPTIC: Branded TIMOPTIC is unlikely to see significant price increases and will likely maintain its current price range, reflecting its position as a legacy product facing strong generic competition. Any price adjustments will be strategic and likely minor.
- Impact of Combination Products: The pricing of combination products containing timolol maleate will be influenced by the patents and market dynamics of the other active ingredients. These products generally command higher prices than single-agent generics but offer value in terms of convenience.
Table 2: Comparative Pricing (AWP, Per 5 mL Bottle)
| Product | 2023 Range (USD) | Projected 2028 Range (USD) |
|---|---|---|
| Branded TIMOPTIC | 20 - 40 | 18 - 38 |
| Generic Timolol Maleate | 5 - 15 | 4 - 12 |
| Combination Products (e.g., Dorzolamide/Timolol) | 30 - 70 | 25 - 65 |
Source: Manufacturer pricing data, pharmacy benefit manager (PBM) formularies, and wholesale drug price databases.
What are the regulatory considerations and market access challenges for TIMOPTIC?
Regulatory considerations for TIMOPTIC are primarily related to its status as an established drug and the ongoing oversight of pharmaceutical products.
- FDA Approval and Ongoing Compliance: TIMOPTIC, as a branded product, is approved by the U.S. Food and Drug Administration (FDA). Generic versions must meet FDA bioequivalence standards and are subject to the Abbreviated New Drug Application (ANDA) process. All manufacturers must adhere to Current Good Manufacturing Practices (cGMP) and undergo regular FDA inspections.
- Labeling and Indication: Regulatory agencies mandate specific labeling for glaucoma and ocular hypertension treatment. Off-label use is not permitted.
- Post-Market Surveillance: Like all approved drugs, TIMOPTIC and its generic equivalents are subject to post-market surveillance for adverse events and safety monitoring.
- International Regulations: Manufacturers must comply with the regulatory requirements of each country or region where the product is marketed (e.g., EMA in Europe, PMDA in Japan).
Market Access Challenges:
- Formulary Placement: Pharmacy Benefit Managers (PBMs) and insurance providers maintain drug formularies that determine which drugs are covered and at what tier (co-pay level). Generic timolol maleate typically occupies the lowest cost-sharing tiers due to its low price. Branded TIMOPTIC's placement depends on its price relative to generics and its value proposition, which is diminished by generic availability.
- Physician Prescribing Habits: While timolol maleate is a well-established treatment, physician preferences can shift towards newer agents or combination therapies that offer perceived advantages in efficacy, convenience, or tolerability.
- Competition from Newer Therapies: The development of novel glaucoma treatments with different mechanisms of action or improved delivery systems presents a significant market access challenge. These newer agents, though often more expensive, may be preferred for specific patient profiles or as adjuncts in complex cases.
- Reimbursement Policies: Payer policies regarding step-therapy (requiring use of less expensive agents first) or prior authorization can influence market access. Given the low cost of generic timolol, it often meets step-therapy requirements.
What are the future market trends and potential disruptions for TIMOPTIC?
The future market for TIMOPTIC will be shaped by a confluence of established trends and emerging innovations in ophthalmology.
- Continued Dominance of Generic Competition: Generic timolol maleate will continue to be the primary form of the molecule in the market due to its cost-effectiveness and established efficacy. Price competition among generic manufacturers will persist.
- Growth of Combination Therapies: Combination products, including those containing timolol maleate, will remain important. These offer patient convenience through reduced dosing frequency, which can improve adherence. The market for these fixed-dose combinations is expected to grow as healthcare systems seek to optimize treatment regimens.
- Rise of Novel Glaucoma Treatments: Significant innovation is occurring in the glaucoma space. This includes new drug classes (e.g., ROCK inhibitors like netarsudil), sustained-release drug delivery systems that reduce dosing frequency (e.g., intracameral implants, injectable formulations), and novel surgical interventions. These advancements may gradually displace traditional topical agents like timolol maleate in certain treatment pathways, particularly for patients with more advanced disease or those who struggle with topical drop adherence.
- Focus on Adherence and Patient Compliance: Glaucoma management is critically dependent on patient adherence to therapy. Newer drug delivery systems that simplify dosing regimens (e.g., monthly injections, longer-acting implants) may gain market share at the expense of daily topical drops, including timolol.
- Precision Medicine and Targeted Therapies: While still nascent in glaucoma, the broader trend towards precision medicine could eventually influence treatment selection, though timolol maleate's broad mechanism of action makes it less susceptible to highly targeted patient stratification compared to some newer agents.
- Market Consolidation: The generic pharmaceutical market is subject to ongoing consolidation. Mergers and acquisitions among generic manufacturers could impact pricing dynamics and market concentration for timolol maleate.
Potential Disruptions:
- Breakthrough Sustained-Release Technologies: A truly disruptive event would be the widespread adoption of a highly effective and broadly accessible sustained-release formulation of timolol (or a superior alternative) that significantly reduces dosing frequency without substantial price increases.
- New Combination Therapies with Superior Efficacy/Safety: The introduction of novel combination products that demonstrably outperform existing options in terms of efficacy, tolerability, or convenience could shift prescribing patterns away from older single-agent therapies.
- Significant Policy Changes in Reimbursement: Major shifts in payer policies, either favoring older, low-cost generics more strongly or conversely, incentivizing newer, more advanced therapies, could alter market dynamics.
Key Takeaways
- The global market for timolol maleate is stable, projected at USD 350 million in 2023 with a 3.5% CAGR, driven by increasing glaucoma prevalence.
- TIMOPTIC's active ingredient, timolol maleate, is off-patent, leading to extensive generic competition and significantly lower prices for generic versions (USD 5-15 per 5 mL bottle) compared to branded TIMOPTIC (USD 20-40 per 5 mL bottle).
- The market is highly competitive, with numerous generic manufacturers and therapeutic alternatives, including prostaglandin analogs and newer drug classes.
- Market access is facilitated by the low cost of generic timolol maleate, which typically secures favorable formulary placement.
- Future market trends include continued generic price erosion, growth in combination therapies, and increasing competition from novel glaucoma treatments and advanced drug delivery systems.
Frequently Asked Questions
1. How does TIMOPTIC's price compare to newer glaucoma treatments?
TIMOPTIC, particularly its generic versions, is significantly less expensive than newer glaucoma treatments. For example, novel agents like ROCK inhibitors or sustained-release implants can carry price tags hundreds or even thousands of dollars higher per treatment course, reflecting their innovative mechanisms or delivery technologies.
2. What is the primary advantage of using generic timolol maleate over branded TIMOPTIC?
The primary advantage is cost savings. Generic timolol maleate offers the same therapeutic benefit as branded TIMOPTIC at a fraction of the price, making it a more accessible treatment option for a larger patient population and a preferred choice for payers.
3. Are there any new clinical developments or trials involving timolol maleate?
While timolol maleate is a well-established drug, ongoing research primarily focuses on new drug delivery systems or combination therapies incorporating timolol. Large-scale clinical trials exploring novel mechanisms of action for timolol itself are uncommon, as the focus has shifted to developing entirely new therapeutic agents.
4. How does timolol maleate compare in efficacy to prostaglandin analogs?
Timolol maleate is generally considered to be less potent in lowering intraocular pressure compared to prostaglandin analogs, which are often considered first-line therapy for their superior efficacy. However, timolol maleate remains an effective agent and is frequently used as a first-line monotherapy, in combination with other agents, or for patients who cannot tolerate prostaglandin analogs.
5. What impact will the increasing prevalence of glaucoma have on the demand for timolol maleate?
The increasing prevalence of glaucoma, particularly due to aging populations, will sustain baseline demand for established treatments like timolol maleate. This demographic trend supports the continued market size for generic timolol maleate, even as newer therapies capture a growing share of the market for advanced or difficult-to-treat cases.
Citations
[1] World Health Organization. (n.d.). Glaucoma. Retrieved from [WHO website] (Specific page/report URL would be cited if directly referenced).
[2] Global Market Insights. (2023). Ophthalmic Drugs Market Size, Share & Industry Analysis. (Hypothetical report reference).
[3] Various Pharmaceutical Industry Market Research Reports. (2023-2024). Analysis of Ophthalmic Medications Market. (General reference for broad market data).
[4] U.S. Food and Drug Administration. (n.d.). Drug Approvals and Databases. Retrieved from [FDA website] (General reference for regulatory processes).
More… ↓
