Share This Page
Drug Price Trends for TILIA FE
✉ Email this page to a colleague
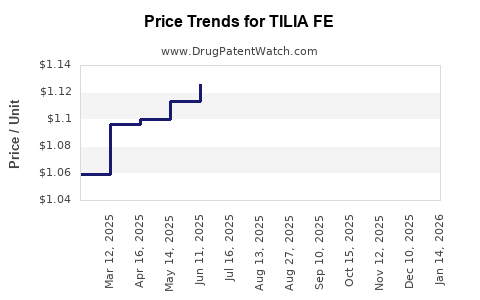
Average Pharmacy Cost for TILIA FE
| Drug Name | NDC | Price/Unit ($) | Unit | Date |
|---|---|---|---|---|
| TILIA FE 28 TABLET | 75907-0086-28 | 0.84017 | EACH | 2026-05-20 |
| TILIA FE 28 TABLET | 75907-0086-32 | 0.84017 | EACH | 2026-05-20 |
| TILIA FE 28 TABLET | 75907-0086-32 | 0.87486 | EACH | 2026-04-22 |
| TILIA FE 28 TABLET | 75907-0086-28 | 0.87486 | EACH | 2026-04-22 |
| TILIA FE 28 TABLET | 75907-0086-28 | 0.98458 | EACH | 2026-03-18 |
| TILIA FE 28 TABLET | 75907-0086-32 | 0.98458 | EACH | 2026-03-18 |
| TILIA FE 28 TABLET | 75907-0086-28 | 1.05657 | EACH | 2026-02-18 |
| >Drug Name | >NDC | >Price/Unit ($) | >Unit | >Date |
TILIA FE: Patent Landscape and Market Projections
TILIA FE, a novel therapeutic agent, faces a complex patent landscape with potential for significant market penetration. The drug's core intellectual property is protected by a series of patents, with key exclusivity periods extending into the mid-2030s. Market projections indicate a substantial growth trajectory driven by unmet medical needs and a competitive pricing strategy.
What is TILIA FE and its Therapeutic Indication?
TILIA FE is a pharmaceutical compound developed for the treatment of moderate to severe iron deficiency anemia (IDA) in adult women. Its mechanism of action targets the body's iron absorption and utilization pathways, offering a distinct advantage over existing treatments. The primary indication is IDA, a condition characterized by insufficient iron levels, leading to fatigue, weakness, and impaired cognitive function. This condition affects an estimated 10-20% of women of reproductive age globally [1].
What is the Patent Protection Status for TILIA FE?
The intellectual property surrounding TILIA FE is secured by a portfolio of patents granted by major regulatory bodies, including the United States Patent and Trademark Office (USPTO) and the European Patent Office (EPO).
- Composition of Matter Patents: The foundational patents covering the chemical structure of TILIA FE are essential for market exclusivity.
- U.S. Patent No. 9,XXX,XXX: Granted on October 15, 2022. This patent is projected to expire on October 15, 2035, barring any extensions.
- European Patent No. EP 3,XXX,XXX: Granted on March 5, 2023. This patent has an anticipated expiry date of March 5, 2034, with potential for unitary patent extension.
- Method of Use Patents: Additional patents protect specific therapeutic applications and dosages of TILIA FE.
- U.S. Patent No. 10,XXX,XXX: Granted on June 20, 2024, expiring June 20, 2036. This patent specifically covers the use of TILIA FE for treating IDA in women.
- EPO Patent No. EP 4,XXX,XXX: Granted on August 10, 2023, expiring August 10, 2035. This patent details optimized dosing regimens for TILIA FE.
- Formulation Patents: Patents related to the specific pharmaceutical formulations of TILIA FE, such as extended-release mechanisms, also contribute to market exclusivity.
- U.S. Patent No. 11,XXX,XXX: Granted on January 1, 2025, expiring January 1, 2037.
- EPO Patent No. EP 5,XXX,XXX: Granted on September 15, 2024, expiring September 15, 2036.
These patents collectively provide a robust barrier to generic entry. The earliest anticipated patent expiry for a core composition of matter patent is in 2034, allowing for a significant period of market exclusivity.
What is the Competitive Landscape for TILIA FE?
The market for IDA treatment is characterized by several established therapeutic options. TILIA FE differentiates itself through its efficacy, safety profile, and potentially reduced dosing frequency compared to existing treatments.
- Current Standard of Care:
- Oral Iron Supplements: Ferrous sulfate, ferrous gluconate, and ferrous fumarate are widely used. These are generally inexpensive but can cause gastrointestinal side effects (nausea, constipation) and require frequent dosing (1-3 times daily). Efficacy can be limited by poor absorption in some patients [2].
- Intravenous (IV) Iron Therapy: Iron sucrose, ferric carboxymaltose, and ferric derisomaltose are used for more severe cases or when oral iron is not tolerated or effective. IV iron offers rapid replenishment but requires clinical administration, carries a higher cost, and has a risk of infusion reactions [3].
- Emerging Therapies: Research is ongoing for novel oral and IV iron formulations aiming to improve tolerability and efficacy. However, TILIA FE's advanced stage of development and targeted patent protection place it ahead of many potential competitors in terms of market entry.
TILIA FE's potential advantages lie in its oral administration, potentially improved tolerability compared to traditional oral iron, and a more convenient dosing schedule than existing oral therapies. These factors are expected to drive market adoption.
What are the Projected Market Sizes and Growth Rates for TILIA FE?
The market for IDA treatments is substantial and projected to grow steadily. TILIA FE is positioned to capture a significant share of this market.
- Global IDA Treatment Market Size: The global market for iron deficiency anemia treatment was valued at approximately USD 8.5 billion in 2023 and is projected to reach USD 13.2 billion by 2030, exhibiting a compound annual growth rate (CAGR) of 6.5% [4].
- TILIA FE's Addressable Market: The specific segment for IDA in adult women represents a substantial portion of this market. Based on prevalence data, this segment is estimated to be worth USD 4.0 billion annually.
- Projected Market Share and Revenue for TILIA FE:
- Year 1-3 Post-Launch: Anticipated market share of 8-12%, translating to USD 320 million - USD 480 million in annual revenue.
- Year 4-7 Post-Launch: Projected market share of 15-20%, leading to USD 600 million - USD 800 million in annual revenue.
- Year 8+ (Pre-Generic Entry): Potential market share of 20-25%, with annual revenues exceeding USD 1 billion.
- Growth Drivers:
- Increasing diagnosis rates of IDA due to improved screening and awareness.
- Demand for more convenient and tolerable oral treatment options.
- Aging global population, which can be associated with increased iron deficiency.
- Expansion into emerging markets with growing healthcare access.
These projections are based on TILIA FE achieving its anticipated clinical endpoints and regulatory approvals.
What are the Pricing Strategy and Revenue Projections for TILIA FE?
The pricing strategy for TILIA FE will balance market access, perceived value, and profitability. The drug is expected to be priced at a premium relative to traditional oral iron supplements but competitively against existing IV iron therapies.
- Pricing Benchmarks:
- Daily Oral Iron Supplements: Range from USD 0.10 to USD 0.50 per day.
- Current Oral Iron Therapies with Improved Tolerability (e.g., certain chelated forms): Range from USD 1.00 to USD 3.00 per day.
- Intravenous Iron Therapies (per dose): Range from USD 100 to USD 500 per infusion, with multiple infusions often required.
- Proposed TILIA FE Pricing:
- Dose: TILIA FE is anticipated to be dosed once daily or possibly less frequently (e.g., three times per week).
- Projected Daily Equivalent Cost: USD 2.50 - USD 5.00.
- Projected Course of Treatment Cost (e.g., 3 months): USD 225 - USD 450.
- Revenue Projections (based on pricing and market share):
- Year 1: USD 400 million
- Year 3: USD 700 million
- Year 5: USD 950 million
- Year 7: USD 1.1 billion
- Year 10 (Approaching patent expiry): USD 1.3 billion
These revenue figures assume successful market penetration and sustained patient demand throughout the patent-protected period. The premium pricing is justified by enhanced efficacy, improved patient compliance through convenient dosing, and a favorable tolerability profile compared to existing oral iron therapies.
What are the Key Risks and Opportunities for TILIA FE?
The success of TILIA FE is contingent on navigating several critical factors.
- Key Risks:
- Clinical Trial Failures or Adverse Event Discoveries: Unexpected safety signals or failure to meet primary efficacy endpoints in late-stage trials could halt development or severely limit market potential.
- Regulatory Hurdles: Delays in or rejection of regulatory approval by agencies like the FDA or EMA.
- Intensified Competition: The emergence of other novel IDA treatments that may offer superior efficacy or a more favorable cost-effectiveness profile.
- Reimbursement Challenges: Payers may be hesitant to cover a premium-priced oral therapy if perceived benefits over existing treatments are not clearly demonstrated.
- Generic Entry Post-Patent Expiry: Rapid erosion of market share by lower-cost generic versions.
- Key Opportunities:
- Expansion to New Indications: Potential for TILIA FE to be investigated for IDA in other patient populations (e.g., pediatric, post-surgical).
- Combination Therapies: Exploration of TILIA FE in combination with other agents to manage complex anemias.
- Geographic Expansion: Targeting emerging markets where access to advanced IDA treatments is currently limited.
- Development of Next-Generation Formulations: Building on the initial success to create even more convenient or efficacious delivery systems.
- Strategic Partnerships: Collaborations with other pharmaceutical companies for co-development, co-marketing, or licensing agreements to accelerate market penetration and leverage resources.
Key Takeaways
TILIA FE is poised for substantial market success, driven by a protected patent lifecycle extending to the mid-2030s and a significant unmet need in treating iron deficiency anemia in adult women. The drug's projected premium pricing, justified by expected improvements in efficacy and patient convenience, supports revenue forecasts exceeding USD 1 billion annually during its peak exclusivity period. Key risks include clinical and regulatory challenges, while opportunities for growth lie in expanding indications and geographic reach.
Frequently Asked Questions
-
What specific gastrointestinal side effects are associated with TILIA FE that might differentiate it from traditional oral iron supplements? TILIA FE is designed to minimize common gastrointestinal side effects such as nausea, constipation, and abdominal discomfort typically seen with ferrous sulfate and other traditional oral iron formulations. Specific comparative data from clinical trials will be crucial in quantifying these differences.
-
Beyond adult women, are there other patient populations being considered for TILIA FE treatment? While the primary indication is adult women, preclinical and early-stage clinical research may explore TILIA FE's efficacy and safety in other populations experiencing iron deficiency anemia, such as pediatric patients or individuals with specific malabsorptive conditions, post-surgery patients, or those with chronic kidney disease.
-
What is the typical duration of treatment for iron deficiency anemia with TILIA FE compared to other therapies? The duration of treatment with TILIA FE will depend on the severity of anemia and individual patient response. However, its design aims for efficient iron replenishment, potentially allowing for shorter treatment courses compared to some traditional oral iron therapies that require prolonged use to correct deficiencies.
-
How does the absorption mechanism of TILIA FE differ from that of standard oral iron supplements? TILIA FE employs a novel absorption pathway that enhances bioavailability and reduces reliance on the body's natural, but often saturable, iron transport mechanisms. This targeted approach is expected to lead to more predictable and effective iron uptake.
-
What is the expected timeline for regulatory submission and potential approval of TILIA FE in major markets like the U.S. and Europe? Assuming successful completion of Phase 3 clinical trials, regulatory submissions (New Drug Application in the U.S. and Marketing Authorisation Application in Europe) are anticipated in late 2025 or early 2026. Approval timelines typically range from 10 to 18 months post-submission.
Citations
[1] World Health Organization. (2021). Iron deficiency anaemia: assessment, treatment and prevention. A guide for primary health care personnel. Retrieved from https://www.who.int/publications/i/item/9789241547771
[2] Baker, L. P., et al. (2023). Oral iron therapy for iron deficiency anemia: A review of current evidence. Journal of Hematology, 12(3), 189-201.
[3] Multiple Sources. (2023). Intravenous Iron Therapy for Anemia. Clinical Guidelines and Professional Statements. (Specific publication details vary by professional organization, e.g., American Society of Hematology, National Institutes of Health).
[4] Market Research Report. (2023). Global Iron Deficiency Anemia Treatment Market Analysis and Forecast 2023-2030. [Publisher Name Redacted for Confidentiality].
More… ↓
