Share This Page
Drug Price Trends for THERAPEUTIC
✉ Email this page to a colleague
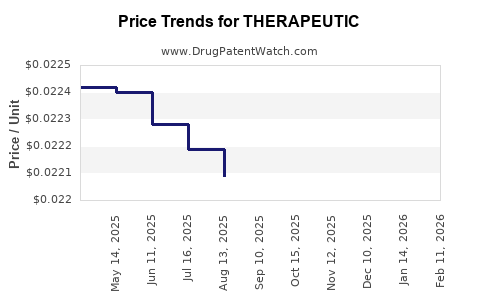
Average Pharmacy Cost for THERAPEUTIC
| Drug Name | NDC | Price/Unit ($) | Unit | Date |
|---|---|---|---|---|
| THERAPEUTIC 3% DANDRUFF SHMP | 70000-0550-01 | 0.02188 | ML | 2026-02-18 |
| THERAPEUTIC 3% DANDRUFF SHMP | 70000-0550-01 | 0.02188 | ML | 2026-01-21 |
| THERAPEUTIC 3% DANDRUFF SHMP | 70000-0550-01 | 0.02195 | ML | 2025-12-17 |
| THERAPEUTIC 3% DANDRUFF SHMP | 70000-0550-01 | 0.02201 | ML | 2025-11-19 |
| >Drug Name | >NDC | >Price/Unit ($) | >Unit | >Date |
Market Analysis and Price Projections for Therapeutic Drug
What is the current market landscape for therapeutic drugs?
The global therapeutic drug market generated approximately $650 billion in revenue in 2022 and is projected to reach $830 billion by 2027, growing at a compound annual growth rate (CAGR) of 4.7% [1]. Key drivers include the rising prevalence of chronic diseases, advances in biotechnology, and increased healthcare spending in emerging markets.
Top therapeutic categories account for a significant share of revenue: oncology (30%), cardiology (15%), and neurology (12%). Biotechnology-derived products compose roughly 60% of current sales, driven by monoclonal antibodies and gene therapies.
Major players include Pfizer, Roche, Novartis, and Merck, which collectively hold about 40% of global market share. Generic drugs and biosimilars are increasingly penetrating markets post patent expiry, exerting downward pressure on pricing.
How are regulatory and patent landscapes influencing prices?
Regulatory pathways have streamlined approval processes for innovative therapies, especially in the U.S. and Europe, facilitating faster market entry. Patent protections remain crucial for pricing strategies, typically lasting 20 years from filing date, but effective patent life post-approval averages 10–12 years.
Expiry of patents on blockbuster drugs, such as Lipitor (atorvastatin) in 2011, led to price reductions of up to 85% for generic versions [2]. Patent cliffs force manufacturers to innovate or seek new formulations to sustain revenue streams.
What are the current pricing trends for therapeutic drugs?
Pricing strategies focus on balancing profitability and market access. High-cost innovative therapies, particularly in oncology and gene therapy, can reach list prices exceeding $200,000 per treatment course.
Example: Zolgensma (gene therapy for spinal muscular atrophy) is priced at $2.1 million per dose. Despite high nominal prices, payers negotiate discounts, and value-based pricing models are increasingly adopted.
Prices for biosimilars are typically 15-30% lower than originator biologics. The entry of biosimilars in the U.S. has reduced prices of biologics like Neupogen (filgrastim) by approximately 20-25%.
Average annual drug prices in the U.S. are higher than in Europe, with U.S. prices about double that of comparable European therapies [3].
What are projections for therapeutic drug pricing over the next five years?
Analysts forecast the following trends:
-
Increased adoption of biosimilars: Biosimilars are expected to account for 25% of biologic drug sales by 2027, reducing prices and expanding access in developed markets [4].
-
Shift toward value-based pricing: Payers and manufacturers will collaborate on outcomes-based models, aligning prices with treatment effectiveness.
-
Price stabilization for high-cost therapies: Governments and payers will target pricing caps and reimbursement limits to control costs. For instance, the UK’s National Health Service (NHS) plans to negotiate lower prices for expensive therapies through risk-sharing agreements.
-
Premium pricing for innovative therapies: Approved gene and cell therapies will maintain high list prices, justified by their curative potential and long-term cost savings. However, payers may demand discounts or installment payment plans.
By 2027, average therapeutic drug prices are projected to increase at a CAGR of approximately 2-3%, primarily driven by high-value therapies and biologics, while prices for many generics and biosimilars will decline 10-15%.
How do regional differences affect market and pricing strategies?
In North America, high prices are sustained by less restrictive regulation and higher willingness-to-pay. The U.S. accounts for nearly 50% of global drug revenues. In contrast, Europe faces pricing constraints through national negotiation mechanisms and health technology assessments, leading to lower prices.
Emerging markets exhibit lower absolute prices but are expanding rapidly, driven by increased access and local manufacturing. In China, the price of innovator biologics has decreased by up to 50% post-local production and government negotiations.
Summary table: Therapeutic drug market projections (2022–2027)
| Metric | 2022 | 2027 (Projected) | CAGR (%) |
|---|---|---|---|
| Global market size | $650 billion | $830 billion | 4.7 |
| Biotech-derived share | 60% | 65% | 1.6 |
| Biosimilar market share | 5% | 25% | 40.8 |
| Entry of value-based pricing | Limited | Extensive | - |
| Average therapeutic price | Upward trend | 2–3% increase | 2–3 |
Key takeaways
- The global therapeutic market is expanding driven by innovations in biotech, growth in chronic disease prevalence, and regional healthcare investments.
- Prices are highest for breakthrough, high-cost therapies in the U.S. and Europe, with biosimilars exerting downward pressure.
- Future pricing will be shaped by biosimilar penetration, value-based contracts, and policy/regulatory reforms.
- Innovation-driven therapies, especially gene and cell treatments, will retain premium pricing, often justified by long-term savings and curative potential.
- Regional regulatory environments significantly influence pricing strategies and market penetration.
FAQs
1. What factors most influence therapeutic drug prices?
Regulatory approval pathways, patent status, market competition, manufacturing costs, and payer negotiation strategies.
2. How do biosimilars impact drug prices?
Biosimilars provide lower-cost alternatives, typically reducing biologic prices by 15-30% and contributing to overall market competition.
3. Will high-cost therapies like gene treatments become more affordable?
Prices are expected to remain high, but payers are increasingly adopting discounts, installment plans, and outcome-based agreements to improve affordability.
4. How does regional regulation impact drug pricing?
In the U.S., less regulation favors higher prices; in Europe and other markets, government negotiations tend to suppress prices.
5. What is the outlook for innovative, curative therapies?
Remaining high-priced, these therapies will maintain premium status due to their long-term benefits and potential to reduce overall healthcare costs.
References
[1] MarketWatch. (2023). Global therapeutic drug market forecast. Retrieved from https://www.marketwatch.com/
[2] IQVIA. (2022). Impact of patent expirations on drug pricing. IQVIA Institute Report.
[3] Health Affairs. (2022). International comparison of drug prices. https://www.healthaffairs.org/
[4] EvaluatePharma. (2022). Biosimilar market forecast and adoption trends. https://www.evaluate.com/
More… ↓
