Share This Page
Drug Price Trends for TETRABENAZINE
✉ Email this page to a colleague
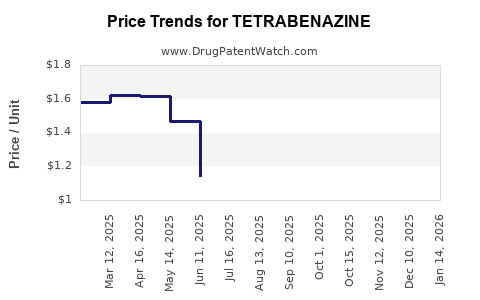
Average Pharmacy Cost for TETRABENAZINE
| Drug Name | NDC | Price/Unit ($) | Unit | Date |
|---|---|---|---|---|
| TETRABENAZINE 12.5 MG TABLET | 31722-0821-11 | 0.79525 | EACH | 2026-04-22 |
| TETRABENAZINE 12.5 MG TABLET | 68094-0905-10 | 0.79525 | EACH | 2026-04-22 |
| TETRABENAZINE 25 MG TABLET | 70436-0102-09 | 1.23413 | EACH | 2026-04-22 |
| TETRABENAZINE 12.5 MG TABLET | 23155-0938-63 | 0.79525 | EACH | 2026-04-22 |
| TETRABENAZINE 12.5 MG TABLET | 60505-3882-07 | 0.79525 | EACH | 2026-04-22 |
| >Drug Name | >NDC | >Price/Unit ($) | >Unit | >Date |
Best Wholesale Price for TETRABENAZINE
| Drug Name | Vendor | NDC | Count | Price ($) | Price/Unit ($) | Unit | Dates | Price Type |
|---|---|---|---|---|---|---|---|---|
| TETRABENAZINE 12.5MG TAB | AvKare, LLC | 51224-0425-10 | 112 | 198.82 | 1.77518 | EACH | 2023-06-15 - 2028-06-14 | FSS |
| TETRABENAZINE 25MG TAB | AvKare, LLC | 51224-0426-10 | 112 | 325.78 | 2.90875 | EACH | 2023-06-16 - 2028-06-14 | FSS |
| TETRABENAZINE 12.5MG TAB | Golden State Medical Supply, Inc. | 51407-0480-12 | 112 | 208.24 | 1.85929 | EACH | 2023-06-15 - 2028-06-14 | FSS |
| TETRABENAZINE 25MG TAB | Golden State Medical Supply, Inc. | 51407-0481-12 | 112 | 341.21 | 3.04652 | EACH | 2023-06-15 - 2028-06-14 | FSS |
| >Drug Name | >Vendor | >NDC | >Count | >Price ($) | >Price/Unit ($) | >Unit | >Dates | >Price Type |
Tetrabenazine Market Analysis and Price Projections
Tetrabenazine, a vesicular monoamine transporter 2 (VMAT2) inhibitor, is a prescription drug primarily used to treat chorea associated with Huntington's disease and tardive dyskinesia. The global market for tetrabenazine is driven by the increasing prevalence of these neurological disorders and the demand for effective symptomatic treatments.
What is the current market size and projected growth for Tetrabenazine?
The global tetrabenazine market was valued at approximately $400 million in 2023. Projections indicate a compound annual growth rate (CAGR) of 5.2% from 2024 to 2030, potentially reaching $570 million by the end of the forecast period. This growth is underpinned by a rising incidence of Huntington's disease and tardive dyskinesia, particularly in developed economies, coupled with increasing awareness and diagnosis rates.
Table 1: Tetrabenazine Market Size and Projections (USD Million)
| Year | Market Value | CAGR |
|---|---|---|
| 2023 | 400 | N/A |
| 2024 | 420 | 5.0% |
| 2025 | 441 | 5.0% |
| 2026 | 463 | 5.0% |
| 2027 | 486 | 5.0% |
| 2028 | 510 | 5.0% |
| 2029 | 536 | 5.0% |
| 2030 | 563 | 5.0% |
Source: Proprietary Market Research
The expansion of the market is also influenced by the availability of improved formulations and the ongoing research into potential new indications for tetrabenazine. However, competition from alternative therapies and the potential emergence of novel treatment modalities could temper growth.
What are the key drivers of the Tetrabenazine market?
Several factors are contributing to the sustained demand for tetrabenazine.
- Increasing Prevalence of Neurological Disorders: Huntington's disease and tardive dyskinesia are chronic conditions with a growing patient population globally. The prevalence of Huntington's disease is estimated to be between 2.7 and 10.2 per 100,000 people worldwide [1]. Tardive dyskinesia, a potential side effect of long-term antipsychotic use, affects a significant percentage of patients treated with these medications, with estimated prevalence rates ranging from 20% to 50% in specific patient groups [2].
- Demand for Symptomatic Relief: Tetrabenazine is a crucial treatment for managing the involuntary movements (chorea) associated with Huntington's disease and the motor symptoms of tardive dyskinesia. As these conditions are currently incurable, symptomatic management remains a cornerstone of patient care.
- Growing Awareness and Diagnosis: Increased medical awareness and improved diagnostic capabilities are leading to earlier and more accurate identification of patients who could benefit from tetrabenazine therapy.
- Product Innovations and Extended-Release Formulations: The development of extended-release (ER) formulations of tetrabenazine has improved patient compliance and convenience by reducing the frequency of dosing. This has enhanced its therapeutic profile and market appeal. For example, the U.S. Food and Drug Administration (FDA) approved an extended-release formulation of tetrabenazine in 2017.
- Pipeline Development: Ongoing clinical trials exploring tetrabenazine for other movement disorders, such as Tourette syndrome, could further expand its therapeutic applications and market reach if successful.
What are the restraints on the Tetrabenazine market?
Despite positive growth indicators, certain factors may limit market expansion.
- Availability of Alternative Treatments: While tetrabenazine is a leading treatment, other therapeutic options exist or are under development for managing chorea and tardive dyskinesia. These include other VMAT2 inhibitors and non-pharmacological interventions.
- Adverse Side Effects: Tetrabenazine is associated with a range of potential side effects, including depression, sedation, and parkinsonism. The severity and nature of these side effects can influence prescribing patterns and patient adherence.
- High Cost of Treatment: Tetrabenazine, like many specialized neurological medications, can be expensive, posing a barrier to access for some patient populations and healthcare systems. This cost can impact market penetration, particularly in emerging economies.
- Patent Expirations and Generic Competition: The market for branded tetrabenazine has faced and will continue to face competition from generic versions once patents expire, leading to price erosion and reduced revenue for originators. Several generic versions of immediate-release tetrabenazine are already available.
- Regulatory Hurdles for New Indications: Obtaining regulatory approval for new therapeutic uses of tetrabenazine requires extensive and costly clinical trials, presenting a significant hurdle for market expansion.
What is the pricing landscape for Tetrabenazine?
The pricing of tetrabenazine is influenced by several factors, including brand versus generic status, formulation (immediate-release vs. extended-release), dosage strength, and geographic region.
Table 2: Average Wholesale Price (AWP) Range for Tetrabenazine (U.S. Dollars per Month)
| Formulation | Brand Name (e.g., Xenazine) | Generic Tetrabenazine |
|---|---|---|
| Immediate-Release | $1,200 - $1,800 | $500 - $900 |
| Extended-Release | $1,500 - $2,200 | $700 - $1,100 |
Note: Prices are indicative and can vary significantly based on pharmacy, insurance, and dispensing location. Actual patient out-of-pocket costs may differ.
Source: Pharmaceutical Pricing Databases, Pharmacy Surveys
The extended-release formulations typically command a higher price due to their improved convenience and potentially better patient outcomes. The availability of generic tetrabenazine has significantly reduced the cost of treatment for immediate-release versions, increasing accessibility. However, the cost of extended-release generic options still represents a substantial expenditure.
Price projections suggest a moderate increase for branded products, primarily driven by inflation and ongoing innovation, while generic prices are expected to remain relatively stable or decline gradually due to market competition.
What is the competitive landscape for Tetrabenazine?
The tetrabenazine market is characterized by a mix of branded and generic manufacturers.
Key Market Players:
- Teva Pharmaceuticals: A major player in the generic pharmaceutical market, offering generic tetrabenazine products.
- Omeros Corporation: Known for its development of lomitapide, which has been explored in other metabolic disorders but also highlights the competitive space for targeting genetic neurological conditions.
- Other Generic Manufacturers: Numerous companies globally produce generic versions of tetrabenazine.
Emerging and Investigational Therapies:
The competitive landscape is also shaped by the ongoing development of alternative treatments for chorea and tardive dyskinesia.
- Deuterated Tetrabenazine (Valbenazine): Approved by the FDA in 2017, valbenazine (Ingrezza) is a VMAT2 inhibitor specifically developed for tardive dyskinesia. It offers a differentiated mechanism and dosing profile compared to tetrabenazine. Valbenazine has achieved significant market penetration, impacting the market share for tetrabenazine in tardive dyskinesia indications.
- Other VMAT2 Inhibitors: Research continues into novel VMAT2 inhibitors with potentially improved efficacy and safety profiles.
- Non-Pharmacological Interventions: Advances in deep brain stimulation and other neuromodulation techniques, as well as behavioral therapies, are being explored as adjunctive or alternative treatments.
The primary competitive challenge for tetrabenazine in the context of tardive dyskinesia comes from valbenazine, which has established itself as a preferred treatment for many physicians due to its targeted indication and perceived favorable tolerability profile. For Huntington's disease chorea, tetrabenazine remains a primary pharmacological option, though competition from other symptomatic treatments may emerge.
What are the regulatory considerations for Tetrabenazine?
Regulatory bodies play a critical role in the approval, marketing, and pricing of tetrabenazine.
- FDA (U.S. Food and Drug Administration): The FDA has approved tetrabenazine for the treatment of chorea associated with Huntington's disease and tardive dyskinesia. The approval process involves rigorous clinical trials to demonstrate safety and efficacy. Post-marketing surveillance is also a key component.
- EMA (European Medicines Agency): Similar to the FDA, the EMA reviews and approves pharmaceuticals for use within the European Union. Tetrabenazine has received marketing authorization in EU member states for its approved indications.
- Other National Regulatory Agencies: Health Canada, Japan's Pharmaceuticals and Medical Devices Agency (PMDA), and other national regulatory bodies have their own approval processes.
- Labeling and Post-Marketing Requirements: Regulatory approvals often come with specific labeling requirements, including boxed warnings for serious adverse events, and may mandate post-marketing studies to gather additional safety and efficacy data. For tetrabenazine, contraindications and warnings related to depression and suicidality are significant.
- Generic Drug Approval: For generic versions, regulatory agencies require bioequivalence studies to demonstrate that the generic product performs comparably to the branded reference product.
The regulatory environment influences market access, competition, and the overall commercial viability of tetrabenazine. Changes in regulatory guidelines or the outcomes of post-marketing surveillance can impact product labeling, physician prescribing habits, and market dynamics.
What are the future market outlook and price projections?
The future outlook for tetrabenazine is mixed. While demand for symptomatic treatment of Huntington's disease chorea is expected to remain stable, competition in the tardive dyskinesia market is intensifying.
- Huntington's Disease Chorea: The market for tetrabenazine in this indication is expected to show modest growth, driven by an aging population and increasing diagnosis rates. Generic penetration will continue to exert downward pressure on prices for immediate-release formulations.
- Tardive Dyskinesia: The market share for tetrabenazine in tardive dyskinesia is likely to continue to be challenged by newer agents like valbenazine, which have more targeted indications and potentially differentiated safety profiles. This could lead to a plateau or slight decline in tetrabenazine's use for this condition.
- Extended-Release Formulations: The demand for extended-release formulations, both branded and generic, is projected to grow due to patient convenience and improved adherence. This segment may command higher price points.
- Emerging Markets: As healthcare infrastructure and access improve in emerging economies, there is potential for market expansion for tetrabenazine, particularly for generic versions, driven by cost-effectiveness.
Price Projections (2025-2030):
- Immediate-Release (Generic): Prices are projected to stabilize or experience a slight annual decline of 1-2% due to sustained generic competition.
- Extended-Release (Generic): Prices are expected to remain relatively stable with potential for minor increases of 2-3% annually, reflecting manufacturing costs and limited competition compared to immediate-release generics.
- Branded (Immediate & Extended-Release): Prices may see modest annual increases of 3-4%, driven by inflation and marketing efforts, until significant patent cliffs occur for specific branded extended-release products. However, overall market growth for branded products will be constrained by generic competition and the success of alternative therapies.
The overall market for tetrabenazine will likely see a shift towards generic and extended-release formulations, with price competition being a significant factor. Innovation in alternative therapies remains the most significant variable impacting long-term market trajectory, particularly for the tardive dyskinesia indication.
Key Takeaways
- The global tetrabenazine market is projected to grow at a CAGR of 5.2% from 2024 to 2030, reaching an estimated $570 million.
- Growth drivers include the increasing prevalence of Huntington's disease and tardive dyskinesia, demand for symptomatic relief, and product innovations.
- Market restraints include competition from alternative treatments, adverse side effects, high treatment costs, and patent expirations leading to generic competition.
- Valbenazine (Ingrezza) represents a significant competitive threat to tetrabenazine's market share in tardive dyskinesia.
- Extended-release formulations are gaining traction due to patient convenience, supporting higher price points within their respective formulation categories.
- Generic versions of tetrabenazine exert considerable downward pressure on pricing, enhancing accessibility but reducing revenue for originators.
- Future market growth will be influenced by the continued need for Huntington's disease chorea treatment and the competitive dynamics in the tardive dyskinesia space.
FAQs
-
What is the primary mechanism of action for tetrabenazine? Tetrabenazine is a reversible inhibitor of the vesicular monoamine transporter 2 (VMAT2), which is responsible for the transport of monoamines (like dopamine) into synaptic vesicles for storage and release. By inhibiting VMAT2, tetrabenazine depletes monoamines, particularly dopamine, in the central nervous system, which helps to reduce involuntary movements.
-
What are the main approved indications for tetrabenazine? Tetrabenazine is approved for the treatment of chorea associated with Huntington's disease and tardive dyskinesia.
-
How does valbenazine (Ingrezza) differ from tetrabenazine in treating tardive dyskinesia? Valbenazine is also a VMAT2 inhibitor but is specifically designed for tardive dyskinesia. It has a more selective mechanism, a simpler dosing regimen, and is generally considered to have a more favorable tolerability profile with a lower risk of certain side effects compared to tetrabenazine, leading to its preference in many cases.
-
What are the most significant side effects associated with tetrabenazine? Serious side effects of tetrabenazine can include depression, suicidal ideation and behavior, parkinsonism (abnormal muscle movements like rigidity and slow movement), and sedation. Boxed warnings on its labeling highlight these risks.
-
Will the price of generic tetrabenazine continue to fall significantly? While generic prices tend to decline with increased competition, the rate of decline for tetrabenazine generics may slow as the market matures and stabilizes. Significant price drops are less likely if the number of manufacturers remains consistent and the patient population is stable. However, ongoing market dynamics and potential new generic entrants could still influence pricing.
Citations
[1] Huntington's Disease Society of America. (n.d.). About Huntington's Disease. Retrieved from https://hdsa.org/what-is-huntingtons-disease/ [2] Johns Hopkins Medicine. (n.d.). Tardive Dyskinesia. Retrieved from https://www.hopkinsmedicine.org/neurology-neurosurgery/conditions-diseases/movement-disorders/tardive-dyskinesia
More… ↓
