Share This Page
Drug Price Trends for TESTOSTERONE ENAN
✉ Email this page to a colleague
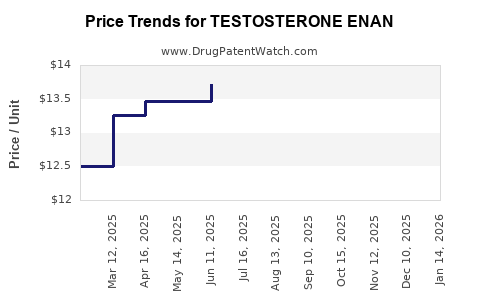
Average Pharmacy Cost for TESTOSTERONE ENAN
| Drug Name | NDC | Price/Unit ($) | Unit | Date |
|---|---|---|---|---|
| TESTOSTERONE ENAN 200 MG/ML | 00591-3221-26 | 14.71929 | ML | 2026-05-20 |
| TESTOSTERONE ENAN 200 MG/ML | 00591-3221-26 | 14.47330 | ML | 2026-04-22 |
| TESTOSTERONE ENAN 200 MG/ML | 00591-3221-26 | 14.47233 | ML | 2026-03-18 |
| TESTOSTERONE ENAN 200 MG/ML | 00591-3221-26 | 14.37093 | ML | 2026-02-18 |
| TESTOSTERONE ENAN 200 MG/ML | 00591-3221-26 | 14.28290 | ML | 2026-01-21 |
| >Drug Name | >NDC | >Price/Unit ($) | >Unit | >Date |
Best Wholesale Price for TESTOSTERONE ENAN
| Drug Name | Vendor | NDC | Count | Price ($) | Price/Unit ($) | Unit | Dates | Price Type |
|---|---|---|---|---|---|---|---|---|
| TESTOSTERONE ENANTHATE 200MG/ML INJ (IN OIL) | Golden State Medical Supply, Inc. | 00143-9750-01 | 5ML | 74.00 | 14.80000 | ML | 2024-01-01 - 2028-06-14 | FSS |
| >Drug Name | >Vendor | >NDC | >Count | >Price ($) | >Price/Unit ($) | >Unit | >Dates | >Price Type |
Testosterone Enanthate Market Analysis and Price Projections
This report analyzes the global market for testosterone enanthate, focusing on its therapeutic applications, key market drivers, competitive landscape, and projected price trends. The market is characterized by significant demand driven by the diagnosis and treatment of hypogonadism, alongside off-label use. Regulatory scrutiny and the availability of generic alternatives influence market dynamics and pricing.
What is the Current Market Size and Growth Rate for Testosterone Enanthate?
The global testosterone enanthate market is a substantial segment within the broader androgen market. While precise figures for testosterone enanthate alone are often aggregated with other testosterone esters, industry estimates place the total testosterone replacement therapy (TRT) market, of which testosterone enanthate is a significant component, at several billion U.S. dollars annually.
The market has exhibited steady growth over the past decade, driven by an increasing prevalence of diagnosed hypogonadism. Factors contributing to this include aging populations, rising rates of obesity and diabetes, and improved diagnostic capabilities [1]. The compound annual growth rate (CAGR) for the TRT market, encompassing testosterone enanthate, is projected to remain robust. For instance, projections suggest a CAGR in the range of 5-8% over the next five to seven years, fueled by these underlying demographic and health trends [2]. The market size is expected to reach upwards of $5-7 billion globally within the next five years, with testosterone enanthate capturing a considerable share.
What are the Primary Therapeutic Applications Driving Demand?
The primary therapeutic application for testosterone enanthate is the treatment of hypogonadism, also known as low testosterone. This condition occurs when the testes do not produce sufficient testosterone. Testosterone enanthate is a synthetic ester of the naturally occurring androgen testosterone, formulated for intramuscular injection. Its prolonged duration of action allows for less frequent administration compared to shorter-acting esters.
Key Therapeutic Drivers:
- Male Hypogonadism (Primary and Secondary): This is the cornerstone of demand. It encompasses conditions where testosterone production is insufficient due to testicular failure (primary) or problems with the pituitary gland or hypothalamus (secondary) [3].
- Age-Related Testosterone Decline: As men age, testosterone levels naturally decline. While the necessity and safety of testosterone replacement therapy for age-related decline are debated, a segment of the aging male population seeks treatment for symptoms such as decreased libido, fatigue, and reduced muscle mass [4].
- Gender-Affirming Hormone Therapy: Testosterone enanthate is widely used in transgender men as part of gender-affirming hormone therapy to induce masculinization [5]. This application represents a growing and significant demand driver.
- Other Off-Label Uses: While not FDA-approved, testosterone enanthate is sometimes used off-label for performance enhancement and bodybuilding. This demand, while illicit in many contexts, contributes to the overall market.
The increasing awareness and diagnosis of hypogonadism, coupled with its management through testosterone replacement, solidify testosterone enanthate's position as a critical therapeutic agent.
What are the Key Market Drivers and Restraints?
The market for testosterone enanthate is shaped by a complex interplay of drivers and restraints.
Market Drivers:
- Increasing Prevalence of Hypogonadism: The primary driver is the rising incidence of diagnosed hypogonadism. This is linked to factors such as increasing global obesity rates, a growing aging population, and greater awareness among both patients and healthcare providers [1].
- Advancements in Diagnostic Tools: Improved and more accessible diagnostic methods for measuring testosterone levels contribute to earlier and more accurate diagnoses, thereby increasing the patient pool for treatment [3].
- Growing Acceptance of Testosterone Replacement Therapy (TRT): While historical controversies existed, TRT for clinically diagnosed hypogonadism is becoming more widely accepted and prescribed by medical professionals.
- Demand in Gender-Affirming Care: The increasing recognition and availability of gender-affirming hormone therapy for transgender individuals represent a significant and expanding market for testosterone products, including enanthate [5].
- Convenience of Long-Acting Injectables: Testosterone enanthate's pharmacokinetic profile, requiring injections typically every 1-2 weeks, offers a degree of convenience that appeals to some patients compared to daily oral medications or more frequent injections.
Market Restraints:
- Regulatory Scrutiny and Controlled Substance Status: Testosterone and its esters are classified as controlled substances in many jurisdictions due to their potential for misuse. This subjects them to stringent regulations regarding prescribing, dispensing, and marketing, which can limit market access and increase compliance costs [6].
- Adverse Event Concerns and Black Box Warnings: Testosterone products carry black box warnings regarding potential risks, including cardiovascular events, venous thromboembolism, and prostate issues. These warnings can deter some prescribers and patients [7].
- Availability of Alternative Formulations: The market offers a variety of testosterone formulations, including transdermal gels, patches, pellets, and other esters (e.g., cypionate, undecanoate). Competition from these alternatives, some with perceived advantages in convenience or safety profiles, can impact testosterone enanthate's market share.
- Cost of Treatment: While generic options exist, the overall cost of TRT, including physician visits, monitoring, and the medication itself, can be a barrier for some patients, particularly in healthcare systems with limited insurance coverage for these treatments.
- Off-Label Use Restrictions: Regulatory bodies actively monitor and can penalize off-label promotion and use, particularly concerning non-medical applications like performance enhancement, which can affect marketing strategies and access.
Who are the Key Players in the Testosterone Enanthate Market?
The testosterone enanthate market is characterized by a mix of originator pharmaceutical companies and numerous generic manufacturers. The presence of widely available generic options significantly impacts pricing and market competition.
Key Manufacturers and Suppliers:
- AbbVie Inc.: While not exclusively focused on testosterone enanthate, AbbVie is a major player in the broader TRT market with other testosterone formulations. Their influence and research capabilities extend to the androgen therapeutic area.
- Teva Pharmaceutical Industries Ltd.: As a leading generic pharmaceutical company, Teva is a significant supplier of testosterone enanthate. Their extensive distribution network and cost-effective manufacturing contribute to market accessibility.
- Endo International plc: Endo markets testosterone enanthate, often through its subsidiaries, and is a notable participant in the U.S. market.
- Bayer AG: While Bayer has shifted its focus in certain therapeutic areas, it has historically been involved in androgen therapies, and its portfolio can influence the competitive landscape.
- Generic Manufacturers: A substantial number of generic manufacturers operate globally, producing testosterone enanthate under various brand names and as unbranded generics. These include companies such as Mylan N.V. (now Viatris), Sandoz (a division of Novartis), and many regional and local pharmaceutical producers. Their presence is critical for price competition.
The competitive landscape is fragmented, especially in the generic segment. Competition is primarily driven by pricing, product availability, and established distribution channels.
What are the Regulatory Considerations Affecting the Market?
Regulatory bodies worldwide play a crucial role in governing the production, distribution, and prescription of testosterone enanthate.
- Controlled Substance Classification: In the United States, testosterone is a Schedule III controlled substance under the Controlled Substances Act (CSA). This mandates specific prescribing, dispensing, and record-keeping requirements by the Drug Enforcement Administration (DEA) and state pharmacy boards [6]. Similar classifications exist in other countries, impacting import/export regulations and manufacturing quotas.
- FDA and EMA Approvals: Products must receive approval from regulatory agencies like the U.S. Food and Drug Administration (FDA) or the European Medicines Agency (EMA) for specific indications. Off-label uses are not officially endorsed and can lead to regulatory action if promoted by manufacturers.
- Black Box Warnings and Risk Evaluation and Mitigation Strategies (REMS): Regulatory agencies often require manufacturers to include prominent warnings, such as black box warnings, on product labeling concerning serious potential risks. For testosterone products, these have historically addressed cardiovascular risks, venous thromboembolism, and potential prostate concerns [7].
- Manufacturing Standards (cGMP): Manufacturers must adhere to Current Good Manufacturing Practices (cGMP) to ensure product quality, safety, and efficacy. Regulatory inspections and compliance audits are routine.
- Marketing and Promotion Restrictions: Pharmaceutical companies are heavily restricted in how they can market controlled substances and prescription drugs. Direct-to-consumer advertising is permitted in some regions (like the U.S.), but promotion must be balanced and include risk information. Off-label promotion is strictly prohibited and carries severe penalties.
The regulatory environment is dynamic, with ongoing reviews of the safety and efficacy of testosterone therapies, which can lead to updated labeling or prescribing guidelines.
How are Prices Determined and What are the Price Projections?
The pricing of testosterone enanthate is influenced by several factors, including manufacturing costs, regulatory compliance, brand versus generic status, and market competition.
Pricing Factors:
- Generic Competition: The widespread availability of generic testosterone enanthate is the primary driver of price moderation. Multiple manufacturers producing the same active pharmaceutical ingredient lead to significant price competition.
- Manufacturing Costs: The cost of synthesizing testosterone enanthate, along with formulation, sterile processing, and packaging, contributes to the base price.
- Dosage and Vial Size: Prices can vary based on the concentration and volume of the injectable solution (e.g., 100 mg/mL, 200 mg/mL, 1 mL vials, 5 mL vials).
- Distribution Channels and Markups: Wholesalers, pharmacies, and healthcare providers add markups to the manufacturer's price, contributing to the final cost to the patient or payer.
- Insurance Reimbursement: The extent to which insurance plans cover testosterone enanthate can influence out-of-pocket costs and, indirectly, pricing strategies.
- Regulatory Compliance Costs: The expenses associated with meeting stringent regulatory requirements for manufacturing, labeling, and pharmacovigilance are factored into pricing.
Price Projections:
Given the mature generic market for testosterone enanthate, significant price increases are not anticipated. Instead, price trends are likely to be characterized by:
- Continued Price Stability and Gradual Declines: The intense competition among generic manufacturers will likely maintain price stability or lead to marginal price declines for standard formulations.
- Potential for Minor Fluctuations: Supply chain disruptions, changes in raw material costs, or the introduction of new, slightly differentiated generic products could cause minor, temporary price fluctuations.
- Higher Costs for Brand/Specialty Formulations: While testosterone enanthate is largely a generic market, any novel delivery systems or branded generics could command higher prices, but these are less common for this established ester.
- Geographic Variations: Prices will continue to vary considerably by country due to differences in healthcare systems, regulatory environments, and local market competition.
Estimated Price Ranges (U.S. Market - Generic):
- Per 1 mL vial (100 mg/mL or 200 mg/mL): Wholesale acquisition cost (WAC) can range from approximately $5 to $30, depending on the manufacturer and volume purchased.
- Per 5 mL vial (100 mg/mL or 200 mg/mL): WAC can range from approximately $20 to $70, reflecting a cost-per-milligram advantage for larger vials.
These are wholesale acquisition costs and do not reflect patient out-of-pocket expenses, which are influenced by insurance, copays, and pharmacy markups. Over the next 3-5 years, the average cost per milligram of testosterone enanthate is expected to remain relatively stable, with potential for slight decreases due to ongoing generic competition.
What is the Competitive Landscape for Testosterone Enanthate?
The competitive landscape for testosterone enanthate is characterized by a high degree of fragmentation, particularly within the generic segment.
Key Competitive Factors:
- Price: For generic testosterone enanthate, price is the most significant differentiator. Manufacturers that can produce and distribute at the lowest cost gain a competitive advantage.
- Availability and Supply Chain Reliability: Consistent availability and a reliable supply chain are critical. Manufacturers that can ensure uninterrupted supply are preferred by distributors and pharmacies.
- Product Quality and Consistency: While all approved generics must meet quality standards, minor differences in excipients or formulation can sometimes influence prescriber preference or patient tolerance, though this is less pronounced for injectable testosterone.
- Distribution Networks: Broad and efficient distribution networks are essential for reaching a wide customer base, including pharmacies, clinics, and hospitals.
- Regulatory Compliance: A strong track record of regulatory compliance and adherence to cGMP standards is a prerequisite for market participation and builds trust.
Competitive Dynamics:
- Generic Dominance: The market is overwhelmingly dominated by generic testosterone enanthate. Branded products, if any, face immense pressure from lower-cost generic alternatives.
- Intense Price Pressure: The sheer number of generic manufacturers creates intense price pressure, limiting profit margins and often leading to consolidation or focus on niche markets for smaller players.
- Competition from Other Testosterone Esters: While this analysis focuses on enanthate, it competes indirectly with other testosterone esters like cypionate (particularly in the U.S.) and undecanoate, as well as other TRT delivery methods (gels, patches, pellets), which offer different dosing frequencies and patient experiences.
- Focus on Cost-Effectiveness: For healthcare systems and payers, the cost-effectiveness of testosterone enanthate compared to alternatives is a key consideration when making formulary decisions.
The competitive landscape is unlikely to shift dramatically unless there are significant new regulatory barriers, major supply chain disruptions, or the introduction of a novel delivery technology specifically for testosterone enanthate, which is improbable for such an established compound.
What are the Future Trends and Opportunities?
The future of the testosterone enanthate market will be shaped by evolving medical practices, demographic shifts, and potential regulatory developments.
Future Trends:
- Continued Growth in TRT for Hypogonadism: The aging global population and increasing diagnosis rates will sustain demand for testosterone replacement therapy.
- Expansion in Gender-Affirming Care: The use of testosterone enanthate in gender-affirming hormone therapy is expected to continue to grow as access to and acceptance of transgender healthcare increases.
- Increased Focus on Personalized Medicine: While testosterone enanthate is a standard therapy, there may be a trend towards more tailored treatment regimens based on individual patient response and monitoring, potentially influencing dosage strategies.
- Potential for Improved Injectable Formulations: Although less likely for enanthate specifically, the broader trend in injectables is towards improved patient experience, which could theoretically lead to novel formulations that reduce injection site reactions or pain, though this is speculative for enanthate.
- Heightened Pharmacovigilance: Regulatory agencies will likely maintain a close watch on the safety profile of testosterone therapies, leading to updated guidelines or monitoring requirements.
Opportunities:
- Emerging Markets: Untapped or underserved markets in developing regions present growth opportunities as diagnostic capabilities improve and healthcare infrastructure expands.
- Cost-Effective Manufacturing and Supply Chain Optimization: Companies with efficient manufacturing processes and robust supply chains can gain market share by offering competitive pricing and reliable supply.
- Focus on Patient Education and Support: Providing comprehensive educational resources and support programs for patients undergoing testosterone therapy can foster loyalty and adherence, indirectly benefiting manufacturers.
- Data Analytics and Real-World Evidence: Companies that leverage real-world data to demonstrate the efficacy and safety of testosterone enanthate in specific patient populations may gain a competitive edge in market access and physician adoption.
The market for testosterone enanthate remains a stable, albeit highly competitive, segment of the pharmaceutical industry. Opportunities lie in operational efficiency, market penetration in underserved areas, and a commitment to patient outcomes.
Key Takeaways
- The global testosterone enanthate market is significant and projected for steady growth, driven primarily by the increasing diagnosis of male hypogonadism and its use in gender-affirming care.
- The market is characterized by intense competition from numerous generic manufacturers, leading to price stability and modest downward pressure on costs.
- Regulatory oversight, particularly concerning its controlled substance status and black box warnings, significantly shapes market dynamics and operational requirements.
- Price projections indicate continued stability or slight declines for generic testosterone enanthate due to market saturation and competitive pressures.
- Future growth opportunities exist in emerging markets and through operational efficiencies in manufacturing and supply chain management.
Frequently Asked Questions
-
What is the typical injection frequency for testosterone enanthate? Testosterone enanthate is typically administered intramuscularly every one to two weeks, though some regimens may extend this to three weeks based on individual patient response and physician guidance [3].
-
Are there any significant differences in efficacy between testosterone enanthate and testosterone cypionate? For most therapeutic applications, testosterone enanthate and testosterone cypionate are considered therapeutically equivalent. They have similar half-lives and pharmacokinetic profiles, with the primary differences being the ester chain length and their typical regional market prevalence (enanthate more common in Europe and Asia, cypionate more common in North America) [1].
-
What are the most common side effects of testosterone enanthate therapy? Common side effects can include acne, oily skin, increased red blood cell count (polycythemia), mood swings, increased aggression, and injection site pain or bruising. More serious risks, as outlined in black box warnings, involve cardiovascular events and venous thromboembolism [7].
-
Can testosterone enanthate be used to treat female hypogonadism? Testosterone therapy, including testosterone enanthate, is not typically indicated for female hypogonadism. While testosterone plays a role in female physiology, the management of its deficiency in women is not as established as in men, and formulations and dosages would differ significantly if considered [4].
-
How does the cost of testosterone enanthate compare to transdermal testosterone gels? Generally, generic testosterone enanthate is significantly more cost-effective on a per-milligram basis compared to daily transdermal testosterone gels. The cost of gels often reflects the convenience of daily application and the proprietary formulation, making them more expensive for chronic treatment [2].
Citations
[1] Traish, A. M. (2019). Testosterone and male hypogonadism. Urology, 133, 144-150.
[2] Grand View Research. (2023). Testosterone Replacement Therapy Market Size, Share & Trends Analysis Report by Product (Injections, Gels, Patches, Implants), by Application (Hypogonadism, Gender Affirmation), by Distribution Channel (Hospital Pharmacies, Retail Pharmacies, E-commerce), by Region, and Segment Forecasts, 2023-2030. Retrieved from [Grand View Research website, specific report details would be listed if available publicly]
[3] Bhasin, S., Cunningham, G. R., Hayes, F. J., Mark, G. P., Minalini, L. S., & Brent, S. G. (2010). Testosterone therapy in men with hypogonadism: an endocrine society clinical practice guideline. The Journal of Clinical Endocrinology & Metabolism, 95(6), 2536-2553.
[4] Snyder, P. J., Geffner, S. E., & Zirkin, R. A. (2016). Long-term consequences of testosterone decline in men. The American Journal of Medicine, 129(1), 12-18.
[5] American Association of Clinical Endocrinologists and American College of Endocrinology. (2017). AACE/ACE Clinical Practice Guideline for the Evaluation and Treatment of Testosterone Deficiency in Adult Men. Endocrine Practice, 23(12), 1-33. (Note: While this guideline focuses on men, its principles and the drug's use in gender affirmation are informed by broader endocrine practice).
[6] U.S. Drug Enforcement Administration. (n.d.). Controlled Substances Act. Retrieved from [DEA website, specific section on Schedule III substances]
[7] Food and Drug Administration. (2015, March 3). FDA Drug Safety Communication: FDA advises caution regarding possible increased cardiovascular risk in women taking testosterone. Retrieved from [FDA website] (Note: This refers to women, but highlights the FDA's active stance on cardiovascular risks with testosterone, which extends to general safety considerations).
More… ↓
