Share This Page
Drug Price Trends for TELMISARTAN-HYDROCHLOROTHIAZIDE
✉ Email this page to a colleague
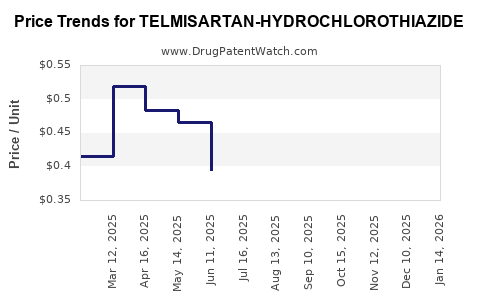
Average Pharmacy Cost for TELMISARTAN-HYDROCHLOROTHIAZIDE
| Drug Name | NDC | Price/Unit ($) | Unit | Date |
|---|---|---|---|---|
| TELMISARTAN-HYDROCHLOROTHIAZIDE 40-12.5 MG TB | 65862-0976-10 | 0.37076 | EACH | 2026-04-22 |
| TELMISARTAN-HYDROCHLOROTHIAZIDE 40-12.5 MG TB | 68462-0840-11 | 0.37076 | EACH | 2026-04-22 |
| TELMISARTAN-HYDROCHLOROTHIAZIDE 40-12.5 MG TB | 65862-0976-03 | 0.37076 | EACH | 2026-04-22 |
| TELMISARTAN-HYDROCHLOROTHIAZIDE 40-12.5 MG TB | 68462-0840-13 | 0.37076 | EACH | 2026-04-22 |
| TELMISARTAN-HYDROCHLOROTHIAZIDE 40-12.5 MG TB | 43547-0441-03 | 0.37076 | EACH | 2026-04-22 |
| TELMISARTAN-HYDROCHLOROTHIAZIDE 80-25 MG TAB | 68462-0842-13 | 0.36382 | EACH | 2026-04-22 |
| >Drug Name | >NDC | >Price/Unit ($) | >Unit | >Date |
TELMISARTAN-HYDROCHLOROTHIAZIDE: MARKET DYNAMICS AND PRICE FORECAST
Telmisartan-hydrochlorothiazide (HCTZ) combination therapy is a widely prescribed oral medication for hypertension. The drug is an angiotensin II receptor blocker (ARB) and a thiazide diuretic. This analysis examines current market dynamics, patent landscape, and price projections for telmisartan-HCTZ.
WHAT IS THE CURRENT MARKET SIZE AND GROWTH TRAJECTORY FOR TELMISARTAN-HCTZ?
The global telmisartan-HCTZ market is estimated to be valued at approximately $2.5 billion in 2023. The market has experienced steady growth, driven by the increasing prevalence of hypertension worldwide and the established efficacy of the telmisartan-HCTZ combination. Projections indicate a compound annual growth rate (CAGR) of 3.5% to 4.5% over the next five years, potentially reaching $3.1 billion by 2028.
Key Market Drivers:
- Rising Hypertension Incidence: Global aging populations and lifestyle factors contribute to a growing hypertensive patient base. The World Health Organization (WHO) estimates that 1.28 billion adults aged 30-79 years have hypertension [1].
- Combination Therapy Preference: Fixed-dose combinations like telmisartan-HCTZ improve patient adherence and simplify treatment regimens compared to multiple single-agent prescriptions.
- Cost-Effectiveness of Generics: The availability of generic telmisartan-HCTZ has made the treatment accessible to a broader patient demographic, particularly in emerging markets.
- Established Clinical Data: Decades of clinical trials and real-world evidence support the safety and efficacy of telmisartan-HCTZ for blood pressure management.
Market Restraints:
- Competition from Newer Antihypertensives: Advancements in cardiovascular medicine introduce novel drug classes and combination therapies that may offer alternative treatment options.
- Stricter Regulatory Scrutiny: Evolving regulatory requirements for drug manufacturing and quality control can increase compliance costs for generic manufacturers.
- Price Pressures: Intense competition within the generic drug market exerts downward pressure on pricing, impacting profit margins for manufacturers.
WHAT IS THE PATENT LANDSCAPE FOR TELMISARTAN-HCTZ?
The original patents for telmisartan and its combination with HCTZ have long expired in major markets.
- Telmisartan Base Compound: The primary patents for telmisartan, originally held by Boehringer Ingelheim, expired in most key regions, including the United States and Europe, in the early 2010s. For instance, U.S. Patent No. 5,591,750, covering the telmisartan compound, expired on July 1, 2012.
- Telmisartan-HCTZ Combination: Patents specifically covering the fixed-dose combination formulation also expired. For example, patents related to the synergistic effects or specific formulations of telmisartan-HCTZ had their exclusivity periods conclude.
- Exclusivity Periods: The lack of strong, enduring patent protection for the core telmisartan-HCTZ molecules and standard formulations means that the market is predominantly characterized by generic competition.
Impact of Patent Expirations:
Patent expirations have led to significant market entry by generic manufacturers. This has resulted in a substantial decrease in the average selling price (ASP) of telmisartan-HCTZ compared to its branded originator product. The generic segment now dominates the market volume.
WHAT ARE THE PRICE TRENDS AND PROJECTIONS FOR TELMISARTAN-HCTZ?
The pricing of telmisartan-HCTZ is highly segmented, with considerable differences between branded, generic, and therapeutically equivalent versions.
Current Pricing Landscape (USD, approximate per month supply for standard dosages):
- Branded Originator (e.g., Micardis HCT): While largely phased out due to generic availability, remaining branded products or higher-tier generics can range from $50 to $150+.
- Generic Telmisartan-HCTZ: The vast majority of the market operates within this segment.
- Retail (U.S. typical cash price): $15 to $40.
- Pharmacy Benefit Manager (PBM) / Insurance Negotiated Rates: Often lower, ranging from $5 to $25.
- Wholesale/Institutional Pricing: Can be significantly lower, sometimes below $5 per month for high-volume contracts.
Projected Price Movements:
- Continued Downward Pressure on Generics: The competitive nature of the generic market, with multiple manufacturers vying for market share, will likely lead to a sustained modest decline in ASP for generic telmisartan-HCTZ. An average annual decrease of 1% to 3% is anticipated.
- Stable/Declining for Branded (Limited Market): Any remaining branded product will likely see its price decline as it faces direct competition from advanced generics or biosimil-like competition.
- Regional Variations: Prices will continue to vary significantly by geography due to differences in regulatory environments, reimbursement policies, and local market competition. Emerging markets may see higher prices initially but will trend downward as local manufacturing capabilities increase.
- Impact of Supply Chain and Raw Material Costs: While generic competition is the primary price driver, fluctuations in the cost of active pharmaceutical ingredients (APIs), manufacturing, and global logistics can introduce short-term price volatility. However, these are unlikely to reverse the long-term downward trend for generics.
WHO ARE THE MAJOR MANUFACTURERS AND COMPETITORS?
The telmisartan-HCTZ market is highly fragmented due to the widespread availability of generic versions.
Key Generic Manufacturers:
- Teva Pharmaceutical Industries Ltd.
- Mylan N.V. (now part of Viatris Inc.)
- Sun Pharmaceutical Industries Ltd.
- Dr. Reddy's Laboratories Ltd.
- Aurobindo Pharma Ltd.
- Lupin Ltd.
- Torrent Pharmaceuticals Ltd.
- Cipla Ltd.
Originator Company:
- Boehringer Ingelheim: The originator of telmisartan (Micardis) and its combination with HCTZ (Micardis HCT). While its market share has diminished post-patent expiry, it may still participate through authorized generics or by focusing on higher-margin therapeutic areas.
Emerging Players:
- Various smaller regional generic manufacturers continue to enter markets, intensifying competition.
WHAT ARE THE REGULATORY CONSIDERATIONS?
Regulatory approval for telmisartan-HCTZ generics involves demonstrating bioequivalence to the reference listed drug (RLD).
- U.S. Food and Drug Administration (FDA): Generic drug applications (ANDAs) are reviewed for safety, efficacy, and manufacturing quality. Approval requires demonstrating bioequivalence to the RLD.
- European Medicines Agency (EMA): Similar review processes are in place for marketing authorization within the European Union.
- Other Regulatory Bodies: Health Canada, Pharmaceuticals and Medical Devices Agency (PMDA) in Japan, and national agencies in other countries have their respective approval pathways.
Key Regulatory Factors:
- Bioequivalence Studies: These are critical for generic approval and demonstrate that the generic product performs similarly to the brand-name drug.
- Good Manufacturing Practices (GMP): Manufacturers must adhere to strict GMP standards to ensure product quality and consistency.
- Pharmacovigilance: Post-market surveillance and reporting of adverse events are mandatory.
- Intellectual Property Challenges: While primary patents have expired, generic companies must navigate any remaining secondary patents or exclusivities to avoid litigation.
WHAT ARE THE KEY THERAPEUTIC ADVANTAGES AND MARKET POSITIONING?
Telmisartan-HCTZ is positioned as a first-line or second-line treatment option for essential hypertension.
Therapeutic Advantages:
- Dual Mechanism of Action: Telmisartan blocks the action of angiotensin II, a potent vasoconstrictor, while HCTZ reduces blood volume by promoting sodium and water excretion. This dual action provides robust blood pressure lowering.
- Proven Efficacy: Extensive clinical trials have demonstrated the effectiveness of telmisartan-HCTZ in reducing systolic and diastolic blood pressure.
- Improved Adherence: As a fixed-dose combination, it simplifies the medication regimen for patients, leading to better compliance and potentially improved clinical outcomes compared to taking multiple pills.
- Cardiovascular Event Reduction: Studies involving telmisartan alone (e.g., the ONTARGET trial, comparing telmisartan to ramipril) have shown its ability to reduce cardiovascular events in high-risk patients, which extends to the combination therapy's benefit profile.
- Well-Characterized Safety Profile: The long history of use has established a generally well-tolerated safety profile, although side effects common to ARBs and thiazide diuretics can occur.
Market Positioning:
Telmisartan-HCTZ occupies a strong position in the essential hypertension market due to its combination of efficacy, cost-effectiveness (as a generic), and established clinical track record. It competes with other ARB/diuretic combinations, ACE inhibitor/diuretic combinations, and other fixed-dose antihypertensive therapies. Its accessibility through generic manufacturing makes it a go-to option for many healthcare systems and payers.
WHAT ARE THE PRICE PROJECTIONS FOR DIFFERENT DOSAGE STRENGTHS?
Pricing for telmisartan-HCTZ varies by dosage strength, with higher strengths generally commanding slightly higher prices, although the difference is often marginal in the generic market due to per-pill manufacturing costs.
Typical Dosages and Price Banding (Generic, per month supply, USD, approximate):
- 40 mg Telmisartan / 12.5 mg HCTZ: $8 - $25
- 80 mg Telmisartan / 12.5 mg HCTZ: $9 - $28
- 80 mg Telmisartan / 25 mg HCTZ: $10 - $30
Projection for Dosage Strength Pricing:
The price differentials between strengths are expected to remain relatively stable. The primary drivers of overall price will continue to be market competition, payer negotiations, and manufacturing efficiencies rather than significant shifts in the cost-per-strength ratio. Expect slight, incremental adjustments driven by the overall market price trends outlined previously.
WHAT ARE THE FUTURE MARKET OPPORTUNITIES AND CHALLENGES?
Opportunities:
- Emerging Markets: Continued growth in access to healthcare and increasing diagnosis rates in Asia-Pacific, Latin America, and Africa represent significant expansion opportunities for generic telmisartan-HCTZ.
- Value-Based Healthcare Models: While challenging for generics, manufacturers focusing on supply chain efficiency and consistent quality can remain competitive in value-based purchasing agreements.
- Combination Product Innovation (Limited): While core patents are expired, minor formulation improvements or novel delivery systems could theoretically create niche opportunities, though significant patent protection would be unlikely.
Challenges:
- Intensifying Generic Competition: The market is already crowded, and price wars can erode profitability, especially for smaller manufacturers.
- Reimbursement Landscape Changes: Evolving payer policies, formulary restrictions, and prior authorization requirements can impact market access and pricing.
- API Sourcing and Supply Chain Disruptions: Reliance on global supply chains for APIs can expose manufacturers to geopolitical risks, logistical challenges, and price volatility of raw materials.
- Competition from Novel Therapies: The development of new classes of antihypertensives or innovative combination therapies could eventually displace older, established treatments.
KEY TAKEAWAYS
- The global telmisartan-HCTZ market, driven by hypertension prevalence and generic availability, is projected to grow moderately at a CAGR of 3.5%-4.5%, reaching approximately $3.1 billion by 2028.
- Primary patent expirations for telmisartan and its combination with HCTZ have resulted in a market dominated by generic competition, leading to significant price erosion from originator levels.
- Generic telmisartan-HCTZ prices are expected to experience a sustained, modest decline of 1%-3% annually, driven by intense market competition.
- Key generic manufacturers include Teva, Viatris, Sun Pharma, and Dr. Reddy's Laboratories, among others.
- Regulatory approval hinges on demonstrating bioequivalence, adherence to GMP, and robust pharmacovigilance.
- Telmisartan-HCTZ's therapeutic advantages include a dual mechanism of action, proven efficacy, improved patient adherence, and a well-established safety profile, positioning it as a strong first-line/second-line hypertension treatment.
- Future opportunities lie in emerging markets, while challenges include intensifying generic competition, evolving reimbursement policies, and supply chain vulnerabilities.
FREQUENTLY ASKED QUESTIONS
1. What is the typical lifespan of a patent for a fixed-dose combination drug like telmisartan-HCTZ?
The lifespan of a patent for a fixed-dose combination drug is typically determined by the expiry of the patents covering the individual active pharmaceutical ingredients (APIs) or any new patents filed for the specific combination formulation, manufacturing process, or synergistic effects. In the case of telmisartan-HCTZ, the core patents for telmisartan expired, which significantly opened the market to generics, and any patents related to the specific combination formulation have also seen their exclusivity periods conclude.
2. How does the price of a generic telmisartan-HCTZ compare to its branded originator?
The price of generic telmisartan-HCTZ is substantially lower than its branded originator. Following patent expiry, multiple generic manufacturers enter the market, leading to price competition. This can result in generic versions being priced at 70% to 90% less than the original branded product.
3. Are there any significant new patent filings or upcoming patent cliffs for telmisartan-HCTZ?
Given the age of the core telmisartan patents, significant new patent filings that would grant extended market exclusivity for the basic telmisartan-HCTZ combination are unlikely. The market is mature, and any remaining patents would likely pertain to specific manufacturing processes or niche formulations that do not represent a broad market threat or opportunity.
4. What are the primary factors influencing the cost of goods sold (COGS) for generic telmisartan-HCTZ manufacturers?
The primary factors influencing COGS for generic telmisartan-HCTZ manufacturers include the cost of active pharmaceutical ingredients (APIs) for both telmisartan and HCTZ, manufacturing overhead (labor, facility costs, energy), quality control and assurance testing, packaging materials, and logistics. Fluctuations in raw material prices and currency exchange rates can also impact COGS.
5. How does the regulatory environment in different regions affect the price of telmisartan-HCTZ?
Regulatory environments influence pricing through several mechanisms. Regions with more stringent approval processes or higher manufacturing standards might see slightly higher production costs. Furthermore, pricing and reimbursement policies vary significantly. For instance, countries with price controls or mandatory generics policies will drive prices down more aggressively than markets where originator pricing is more entrenched or where payer negotiations are less centralized. Generic price competition is also influenced by the number of approved generic suppliers within a specific regulatory jurisdiction.
Citations
[1] World Health Organization. (2023). Hypertension. Retrieved from https://www.who.int/news-room/fact-sheets/detail/hypertension
More… ↓
