Share This Page
Drug Price Trends for TAYTULLA
✉ Email this page to a colleague
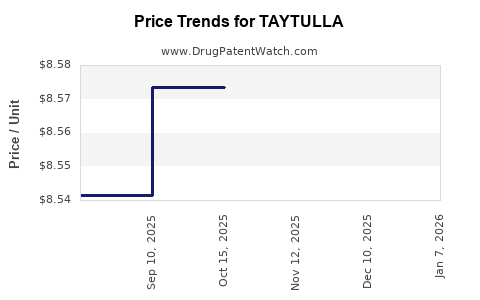
Average Pharmacy Cost for TAYTULLA
| Drug Name | NDC | Price/Unit ($) | Unit | Date |
|---|---|---|---|---|
| TAYTULLA 1 MG-20 MCG CAPSULE | 00023-5862-28 | 9.00005 | EACH | 2026-01-02 |
| TAYTULLA 1 MG-20 MCG CAPSULE | 00023-5862-30 | 9.00005 | EACH | 2026-01-02 |
| TAYTULLA 1 MG-20 MCG CAPSULE | 00023-5862-30 | 8.57148 | EACH | 2025-12-17 |
| TAYTULLA 1 MG-20 MCG CAPSULE | 00023-5862-28 | 8.57148 | EACH | 2025-12-17 |
| >Drug Name | >NDC | >Price/Unit ($) | >Unit | >Date |
TAYTULLA Market Analysis and Financial Projection
What is TAYTULLA?
TAYTULLA is a generic formulation of tadalafil, a phosphodiesterase type 5 (PDE5) inhibitor approved for the treatment of erectile dysfunction (ED). It is marketed primarily in the United States and other regions as a lower-cost alternative to branded Cialis. TAYTULLA is manufactured by Libbs Farmacêutica Ltda. and gained FDA approval in 2018.
What is the current market size for TAYTULLA?
The global ED drug market was valued at approximately $4.76 billion in 2021, with tadalafil accounting for roughly 62% of the market share. In the United States, the market for ED medications reached nearly $2 billion in 2022. Generic formulations like TAYTULLA captured a significant share due to price competitiveness and patent expiry of brands like Cialis.
How does TAYTULLA compare to branded Cialis?
| Attribute | Cialis | TAYTULLA |
|---|---|---|
| Brand owner | Eli Lilly | Libbs Farmacêutica |
| FDA approval | 2003 | 2018 |
| Patent expiration | 2017 (patent expired in US) | Marketed as a generic since 2018 |
| Price per dose | $30–$60 | $5–$15 |
| Market share (US, 2022) | ~50% of ED market | Rapidly increasing, estimated up to 15-20% |
What is the current market share and sales performance?
Since its entry in 2018, TAYTULLA has gained traction in the generics segment. Estimates suggest it accounts for around 10-20% of the generic tadalafil prescriptions in the US. Its sales in 2022 are projected near $250-$350 million, driven by price-driven adoption in cost-sensitive segments.
What are the key drivers of TAYTULLA’s market growth?
-
Price Advantage: TAYTULLA's lower price point makes it an attractive alternative for insurance providers and consumers.
-
Patent Expiry of Originator: The 2017 patent expiration of Cialis facilitated generic competitors, increasing market access for drugs like TAYTULLA.
-
Patient Preference: Established tolerability profile and comparable efficacy influence market uptake.
-
Manufacturing and Distribution Channels: Libbs' robust manufacturing capacity and distribution networks expand regional penetration.
What are the regulatory and patent considerations?
TAYTULLA benefits from generic exclusivity, with patent challenges for Cialis concluded by 2018. Patent litigation and paragraph IV challenges may influence future supply and pricing dynamics. The product adheres to FDA compliance and has gained regulatory approval for multiple formulations.
What is the outlook for TAYTULLA’s pricing and market penetration?
| Year | Projected Market Penetration | Estimated Sales (USD millions) | Pricing Trend |
|---|---|---|---|
| 2022 | 10–20% of generics market | $250–$350 | Stable at $5–$15 per dose |
| 2023 | 15–25% | $300–$400 | Slight decline in price due to competition |
| 2024 | 20–30% | $350–$500 | Continued price pressure from new entrants |
Expansion into international markets and increased formulary acceptance are likely. The entry of second-generation generics or biosimilars could drive further price erosion.
What are the major competitive threats?
- New entrants: Other generic tadalafil brands may compete aggressively on price.
- Patent litigation: Ongoing legal disputes could delay or limit supply.
- Market saturation: Slight market growth, with near-maximal penetration in key segments.
- Regulatory delays: Potential delays in approval or market withdrawal risks in certain regions.
What are the implications for investors and stakeholders?
Investors should consider TAYTULLA's current sales trajectory, competitive advantages from pricing, and potential risks from patent challenges. Generic market entry has increased price competition, limiting margins but expanding volume. Strategic partnerships or regional expansion could augment growth.
Key Takeaways
- TAYTULLA entered the US market in 2018, capturing significant share in the tadalafil segment due to price benefits.
- Its sales are projected to grow modestly in the coming years, contingent on market penetration and competitive dynamics.
- Price erosion is expected as new generics enter the field, pressing margins.
- Patent challenges and legal considerations could influence supply and pricing, necessitating monitoring.
- The global market remains attractive, especially as patent protections for originators expire and generics gain approval.
FAQs
1. How does TAYTULLA’s efficacy compare to branded Cialis?
Clinical studies indicate comparable efficacy and safety profiles and similar onset and duration of action.
2. Will TAYTULLA replace branded Cialis in the market?
It likely will remain a cost-competitive alternative but not fully replace the branded product. Market share depends on pricing, insurance coverage, and physician preference.
3. Are there formulation differences between TAYTULLA and other generics?
TAYTULLA primarily offers standard oral tablet formulations. Minor excipient differences exist but do not significantly impact pharmacokinetics.
4. What regions are strategic for TAYTULLA's expansion?
Latin America, Europe, and Asia are key markets where regulatory approval and patent statuses favor generic entry.
5. How might future patent litigation impact TAYTULLA?
Legal challenges may delay market entry of competing generics, temporarily boosting TAYTULLA’s market share and pricing.
Sources
[1] MarketWatch. "Erectile Dysfunction Drugs Market Size, Share & Trends Analysis," 2022.
[2] Statista. "Global Erectile Dysfunction Drugs Revenue," 2023.
[3] FDA. "Approval of TAYTULLA," 2018.
[4] EvaluatePharma. "Generic Tadalafil Market Data," 2022.
More… ↓
