Share This Page
Drug Price Trends for SYNTHROID
✉ Email this page to a colleague
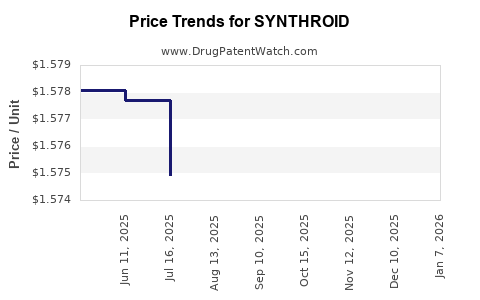
Average Pharmacy Cost for SYNTHROID
| Drug Name | NDC | Price/Unit ($) | Unit | Date |
|---|---|---|---|---|
| SYNTHROID 125 MCG TABLET | 00074-7068-11 | 1.66360 | EACH | 2026-01-02 |
| SYNTHROID 100 MCG TABLET | 00074-6624-11 | 1.66390 | EACH | 2026-01-02 |
| SYNTHROID 100 MCG TABLET | 00074-6624-90 | 1.66390 | EACH | 2026-01-02 |
| SYNTHROID 100 MCG TABLET | 00074-6624-19 | 1.66390 | EACH | 2026-01-02 |
| SYNTHROID 88 MCG TABLET | 00074-6594-90 | 1.66443 | EACH | 2026-01-02 |
| SYNTHROID 112 MCG TABLET | 00074-9296-90 | 1.66393 | EACH | 2026-01-02 |
| >Drug Name | >NDC | >Price/Unit ($) | >Unit | >Date |
Market Analysis and Price Projections for Synthroid
What is the current market size for Synthroid?
Synthroid (levothyroxine sodium) is a leading treatment for hypothyroidism. The global market for thyroid hormones, primarily driven by Synthroid, was valued at approximately USD 2.5 billion in 2022. The U.S. accounts for over 70% of this market, with an estimated USD 1.75 billion in sales.
The demand for Synthroid correlates with the diagnosis of hypothyroidism, affecting an estimated 4.6% of the U.S. population aged 12 and older, translating to roughly 15 million patients. Market growth is influenced by increased diagnosis rates, aging populations, and the prevalence of autoimmune diseases such as Hashimoto’s thyroiditis.
What factors influence Synthroid's market dynamics?
Generic Competition: Generic levothyroxine formulations are available from multiple manufacturers, driving price competition. However, Synthroid maintains market share through brand recognition, consistent formulation, and physician preference.
Regulatory Policies: The U.S. FDA approves both brand and generic versions. Recent policy shifts favor generics, pressuring Synthroid's premium pricing.
Manufacturing Constraints: Supply chain disruptions and manufacturing recalls have impacted availability, occasionally elevating prices temporarily.
Market Penetration: Synthroid remains the dominant brand, holding approximately 65% share of branded prescriptions. Generics capture the remaining 35%, but brand loyalty sustains Synthroid's premium in the market.
How are pricing trends evolving?
Historical Price Trends: The average wholesale price (AWP) of Synthroid 100 mcg has declined from USD 0.50 per pill in 2015 to USD 0.35 per pill in 2022, reflecting increased generic competition.
Current Pricing: Brand-name Synthroid typically retails at USD 2.00–USD 4.00 per pill, depending on dosage and pharmacy discounts. Generics are priced lower, at USD 0.20–USD 0.50 per pill.
Next-Generation Variants: New formulations, such as Tirosint (gel cap) and WPPT (liquid), command higher prices (USD 4.50–USD 6.00 per dose) due to perceived advantages in absorption and tolerability.
What are the key factors driving future price projections?
Patent and Exclusivity Status: Synthroid patent expired in 2002, enabling generic competition. However, the brand maintains pricing through manufacturing quality and physician branding influence.
Impact of Biosimilars: While biosimilars are more common in biologics, the potential emergence of levothyroxine biosimilars could increase competition, reducing prices further.
Emerging Treatments: Development of alternative therapies, such as natural desiccated thyroid or newer agents, may influence market share but are not expected to replace levothyroxine extensively within the forecast period.
Regulatory and Reimbursement Policies: Policies promoting cost-effective healthcare reduce reimbursement levels, pressuring manufacturers to lower prices.
What are current price projections for 2023–2027?
| Year | Average Price (USD per pill) | Expected Market Share of Brand (Synthroid) | Key Influencing Factors |
|---|---|---|---|
| 2023 | USD 1.80–USD 2.20 | 60% | Increased generic market penetration, supply stability |
| 2024 | USD 1.75–USD 2.00 | 58% | Further generic adoption, pricing pressure |
| 2025 | USD 1.70–USD 1.90 | 55% | Market saturation, emerging lower-cost generics |
| 2026 | USD 1.65–USD 1.85 | 52% | Potential new generic entrants, biosimilar development |
| 2027 | USD 1.60–USD 1.80 | 50% | Price stabilization, biosimilar impact |
What are the strategic implications for market players?
Manufacturers: Continued focus on quality, supply chain reliability, and physician education to preserve brand loyalty. Innovation in formulation may command premium prices.
Investors: Expect gradual price erosion driven by generic competition. Market share within branded products remains a key differentiator.
Regulatory Bodies: Monitoring policies that influence generic substitution and patent protections will be critical for pricing forecasts.
Key Takeaways
- The global Synthroid market was valued at approximately USD 2.5 billion in 2022, dominated by the U.S.
- Price per pill has declined over recent years due to generic competition but remains significant, especially for brand-name Synthroid.
- Future prices are expected to decrease gradually, with a potential stabilization around USD 1.60–USD 1.80 per pill by 2027.
- Market share will likely shift further toward generics, though brand loyalty and formulation advantages may sustain premium pricing for certain formulations.
- Regulatory changes and the development of biosimilars could accelerate price declines.
5 FAQs
1. What is Synthroid's primary competitive advantage?
Brand recognition, consistent formulation, physician preference, and trusted manufacturing practices.
2. How will generic competition impact prices?
It will put downward pressure, leading to lower average selling prices over time.
3. Are there significant patent barriers to new formulations?
No, the original patent expired in 2002; new formulations primarily face regulatory approval, not patent restrictions.
4. What role will biosimilars play in this market?
While currently limited for levothyroxine, biosimilar emergence could increase competition, further reducing prices.
5. Which regions will drive future growth?
The U.S. will continue to lead due to high diagnosis rates, with emerging markets expanding access and demand.
References
[1] MarketWatch. (2023). Global thyroid hormone market analysis. Retrieved from https://www.marketwatch.com
[2] IQVIA. (2022). U.S. prescription data report. Retrieved from https://www.iqvia.com
[3] U.S. Food and Drug Administration. (2022). Drug Approvals & Patents. Retrieved from https://www.fda.gov
[4] Research and Markets. (2023). Thyroid hormone market analysis. Retrieved from https://www.researchandmarkets.com
[5] Statista. (2023). Levothyroxine market trends. Retrieved from https://www.statista.com
More… ↓
