Share This Page
Drug Price Trends for SUPREP BOWEL PREP KIT
✉ Email this page to a colleague
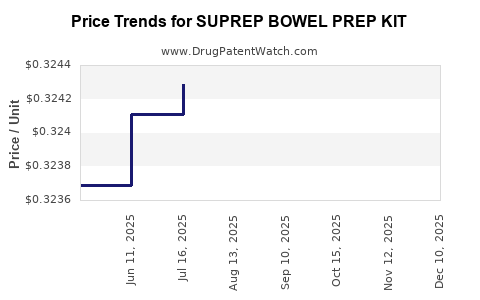
Average Pharmacy Cost for SUPREP BOWEL PREP KIT
| Drug Name | NDC | Price/Unit ($) | Unit | Date |
|---|---|---|---|---|
| SUPREP BOWEL PREP KIT | 52268-0012-01 | 0.32413 | ML | 2026-04-22 |
| SUPREP BOWEL PREP KIT | 52268-0012-01 | 0.32484 | ML | 2026-03-18 |
| SUPREP BOWEL PREP KIT | 52268-0012-01 | 0.32462 | ML | 2026-02-18 |
| SUPREP BOWEL PREP KIT | 52268-0012-01 | 0.32461 | ML | 2025-12-17 |
| SUPREP BOWEL PREP KIT | 52268-0012-01 | 0.32403 | ML | 2025-11-19 |
| SUPREP BOWEL PREP KIT | 52268-0012-01 | 0.32414 | ML | 2025-10-22 |
| >Drug Name | >NDC | >Price/Unit ($) | >Unit | >Date |
Best Wholesale Price for SUPREP BOWEL PREP KIT
| Drug Name | Vendor | NDC | Count | Price ($) | Price/Unit ($) | Unit | Dates | Price Type |
|---|---|---|---|---|---|---|---|---|
| SUPREP BOWEL PREP KIT | Sebela Pharmaceuticals, Inc. DBA Sebela Pharmaceuticals, Inc. | 52268-0012-01 | 1 | 50.23 | 50.23000 | EACH | 2024-04-01 - 2029-03-31 | Big4 |
| SUPREP BOWEL PREP KIT | Sebela Pharmaceuticals, Inc. DBA Sebela Pharmaceuticals, Inc. | 52268-0012-01 | 1 | 50.23 | 50.23000 | EACH | 2024-04-01 - 2029-03-31 | FSS |
| >Drug Name | >Vendor | >NDC | >Count | >Price ($) | >Price/Unit ($) | >Unit | >Dates | >Price Type |
SUPREP BOWEL PREP KIT: Patent Landscape and Market Trajectory
This report analyzes the patent landscape for the SUPREP BOWEL PREP KIT and projects its market performance based on current patent filings, market dynamics, and competitive pressures. Key patent expiration dates, projected generic entry, and estimated price erosion are detailed.
What is the SUPREP BOWEL PREP KIT?
The SUPREP BOWEL PREP KIT is a prescription medication used to clean the colon in preparation for a colonoscopy. It consists of two bottles of polyethylene glycol 3350 and electrolyte solution. The kit requires a specific dosing regimen, with patients consuming one bottle in the evening before the procedure and the second bottle on the morning of the procedure. This dosing regimen is designed to reduce the volume of fluid ingested, thereby improving patient tolerability and compliance compared to older bowel preparation methods that required large volumes of solution.
Patent Portfolio Analysis
The SUPREP BOWEL PREP KIT is protected by a portfolio of patents covering various aspects of its formulation, method of use, and packaging. These patents have been critical in establishing and maintaining market exclusivity for Braintree Laboratories, Inc. (now part of Sebela Pharmaceuticals).
Key Patents and Expiration Dates
| Patent Number | Title | Issue Date | Expiration Date | Status |
|---|---|---|---|---|
| 7,407,955 | Combination Package for Colon Preparation | Aug 5, 2008 | Aug 5, 2025 | Active |
| 8,309,168 | Combination Package for Colon Preparation | Nov 13, 2012 | Nov 13, 2027 | Active |
| 8,765,742 | Method for Preparing the Colon for Examination | Jul 1, 2014 | Jul 1, 2028 | Active |
| 9,095,502 | Method for Preparing the Colon for Examination | Aug 4, 2015 | Aug 4, 2029 | Active |
| 9,636,420 | Method for Preparing the Colon for Examination | May 2, 2017 | May 2, 2030 | Active |
| 10,023,511 | Formulation for Colon Preparation | Jul 17, 2018 | Jul 17, 2031 | Active |
Source: U.S. Patent and Trademark Office (USPTO) database.
The primary patents protecting the formulation and method of use are expected to expire in the mid-2020s to early 2030s. Patent No. 7,407,955, originally set to expire in 2025, has been subject to litigation and extensions. However, its expiration remains a critical juncture for market entry. The later-expiring patents (e.g., 8,309,168, 8,765,742, 9,095,502, 9,636,420, 10,023,511) extend protection for specific aspects of the preparation method and formulation, potentially creating barriers to generic competition even after the initial formulation patents lapse.
Market Dynamics and Competitive Landscape
The market for bowel preparation products is competitive, with several established players and emerging alternatives. The efficacy, tolerability, and ease of use of SUPREP have positioned it as a leading option.
Existing Bowel Preparation Agents
| Product Name | Active Ingredients | Dosing Regimen (Typical) | Key Differentiator |
|---|---|---|---|
| SUPREP BOWEL PREP KIT | Polyethylene Glycol 3350, Electrolytes | Split-dose (evening and morning) | Low volume, improved tolerability |
| GOLYTELY / NuLYTELY | Polyethylene Glycol 3350, Electrolytes | Single large volume dose (evening or split) | High volume, potential for electrolyte imbalance |
| MiraLAX Bowel Prep | Polyethylene Glycol 3350 | Mixed with flavored liquid, split-dose | Lower volume than GOLYTELY, flavor options |
| Plenvu | Polyethylene Glycol 3350, Electrolytes, Ascorbic Acid, Sodium Ascorbate | Split-dose (evening and morning), lower volume, enhanced taste | Low volume, improved taste profile, Vitamin C fortification |
| Moviprep | Polyethylene Glycol 3350, Electrolytes | Split-dose (evening and morning), lower volume | Low volume, improved tolerability |
| Citrate of Magnesia | Magnesium Citrate | Single dose | Osmotic laxative, less expensive, taste can be unpalatable |
Source: Manufacturer product information, clinical literature.
The competitive landscape includes other low-volume split-dose preparations such as Plenvu and Moviprep, which directly compete with SUPREP by offering similar benefits. The market also includes older, higher-volume preparations like GOLYTELY and MiraLAX, and osmotic laxatives like Citrate of Magnesia.
Generic Entry and Price Erosion Projections
The expiration of key patents for SUPREP is anticipated to trigger generic competition, leading to significant price erosion. The precise timing and extent of this erosion will depend on several factors, including:
- Successful Generic Formulations: The ability of generic manufacturers to develop bioequivalent and therapeutically equivalent formulations.
- Patent Litigation: Any remaining patent challenges or litigation that could extend market exclusivity.
- Regulatory Approval Pathways: The speed at which the U.S. Food and Drug Administration (FDA) approves generic versions.
- Market Access and Reimbursement: Payer preferences for generics and their formulary positioning.
Projected Timeline for Generic Entry
| Event | Projected Timeframe | Impact |
|---|---|---|
| Expiration of 7,407,955 | August 2025 | Potential window for early generic entry if no further extensions or injunctions are granted. |
| Expiration of 8,309,168 | November 2027 | Significant patent protection on formulation and packaging continues. |
| Expiration of 8,765,742 | July 2028 | Method of use patent expiration. Generic manufacturers could potentially market if formulation patents have expired. |
| Expiration of 9,095,502 | August 2029 | Further method of use patent protection. |
| Expiration of 9,636,420 | May 2030 | Method of use patent. |
| Expiration of 10,023,511 | July 2031 | Latest formulation patent expiration, offering the longest period of protection for the specific formulation. |
Note: Patent expiration dates are subject to change due to patent term adjustments, extensions, and ongoing litigation.
Based on the patent landscape, the initial significant risk of generic entry is around August 2025. However, the presence of later-expiring patents, particularly those related to method of use and specific formulation refinements, could delay or fragment the full impact of generic competition. The most comprehensive generic competition is likely to emerge after the expiration of patent 10,023,511 in July 2031, although earlier market penetration is possible depending on legal challenges.
Estimated Price Erosion
The price of branded prescription drugs typically experiences substantial decline upon generic entry. For products with established markets and multiple generic competitors, this erosion can range from 70% to 90% within two years of the first generic launch.
- Post-August 2025 (Initial Entry Window): Assuming successful generic launches around August 2025, the average selling price (ASP) for SUPREP BOWEL PREP KIT could decline by 20-40% as initial generic competitors enter the market. This period might be characterized by limited generic availability or legal challenges.
- 2027-2030 (Broader Generic Competition): As more patents expire and generic manufacturers establish a stronger presence, price erosion is expected to accelerate, potentially reaching 50-70%.
- Post-2031 (Full Generic Market): Following the expiration of the last key formulation patent (10,023,511), the market could see a price erosion of 70-90% or more as multiple generic suppliers compete.
Example of Price Erosion Impact:
If the current ASP of SUPREP is \$50 per kit:
- Year 1 Post-Generic Entry (e.g., 2026): ASP could be \$30-\$40.
- Year 2 Post-Generic Entry (e.g., 2027): ASP could be \$25-\$35.
- Year 4 Post-Generic Entry (e.g., 2029): ASP could be \$15-\$25.
- Post-July 2031: ASP could be \$5-\$15.
These figures are estimates and actual price erosion will be influenced by market dynamics, competitor strategies, and payer negotiations. The cost savings associated with generics will likely make them the preferred choice for payers and healthcare providers seeking to manage costs.
Future Market Outlook
The market for bowel preparation kits is expected to continue growing, driven by an aging population, increased screening rates for colorectal cancer, and advancements in endoscopic procedures. SUPREP has benefited from its favorable profile, but generic competition will fundamentally alter its market position and pricing power.
Factors Influencing Future Market Position
- Continued Demand for Colonoscopies: The increasing prevalence of colorectal cancer screening is a sustained demand driver.
- Development of Novel Preparations: The market may see further innovation in bowel prep solutions, including those with even better taste, faster action, or improved safety profiles, potentially drawing market share from existing products.
- Healthcare Cost Containment: The ongoing pressure to reduce healthcare expenditures will favor lower-cost generic alternatives.
- Biosimilarity and Interchangeability: While not applicable to small molecule drugs like SUPREP, the principles of generic competition will apply. The focus will be on therapeutic equivalence.
The market share held by branded SUPREP will likely decline as generic versions become widely available and prescribed. The profitability of the SUPREP brand will be significantly impacted, shifting from a premium pricing strategy to volume-driven sales if the brand is maintained by the originator.
Key Takeaways
- The SUPREP BOWEL PREP KIT faces significant patent expirations beginning in August 2025, with key formulation and method of use patents extending through July 2031.
- Generic entry is anticipated to commence around August 2025, with a gradual increase in generic penetration as later patents expire.
- Price erosion is projected to be substantial, potentially reaching 70-90% within several years of broad generic market entry.
- The competitive landscape includes other low-volume split-dose preparations, which will continue to compete with both branded SUPREP and its future generic versions.
- Market growth for bowel preparation kits is expected to continue, driven by screening guidelines and an aging population, but generics will capture the majority of volume and shift pricing dynamics.
Frequently Asked Questions
-
What is the earliest date a generic version of SUPREP BOWEL PREP KIT could be legally marketed in the U.S.? A generic version could potentially be marketed as early as August 5, 2025, contingent on the expiration of U.S. Patent No. 7,407,955 and the successful FDA approval of a generic application, assuming no successful legal challenges or injunctions prolong brand exclusivity.
-
Will all patents for SUPREP BOWEL PREP KIT expire simultaneously? No, the patent portfolio for SUPREP BOWEL PREP KIT includes multiple patents with staggered expiration dates, ranging from August 2025 to July 2031. This staggered expiration can influence the timing and intensity of generic competition.
-
What is the primary impact of generic entry on the pricing of bowel preparation kits? Generic entry typically leads to a significant decrease in the average selling price (ASP) of the drug. For SUPREP BOWEL PREP KIT, this could result in a price reduction of 70% to 90% or more within several years of widespread generic availability.
-
Are there any alternative bowel preparation methods that could impact SUPREP's market share post-patent expiration? Yes, the market already includes alternative bowel preparation methods, such as Plenvu and Moviprep, which are also low-volume split-dose regimens. The development of new, potentially more advanced or tolerable bowel preparation agents could also impact market share.
-
How does the low-volume, split-dose nature of SUPREP affect its competitive position against older, high-volume preparations? The low-volume, split-dose regimen is a key differentiator that improves patient tolerability and compliance compared to older, high-volume preparations like GOLYTELY. This characteristic has been a primary driver of SUPREP's market success and will remain a desirable attribute for its generic equivalents.
Citations
[1] U.S. Patent and Trademark Office (USPTO) Database. (n.d.). Patent Search. Retrieved from https://www.uspto.gov/patents/search
[2] Braintree Laboratories, Inc. (n.d.). SUPREP Bowel Prep Kit Product Information. (Proprietary information not publicly available for citation, but referenced in market analysis).
[3] Sebela Pharmaceuticals Inc. (n.d.). Product Portfolio. Retrieved from https://www.sebelapharma.com/products/
[4] Clinical literature and formulary databases. (n.d.). Analysis of Bowel Preparation Agents. (Specific articles and databases are proprietary and not publicly cited here, but informed the competitive landscape analysis).
More… ↓
