Share This Page
Drug Price Trends for STROMECTOL
✉ Email this page to a colleague
Average Pharmacy Cost for STROMECTOL
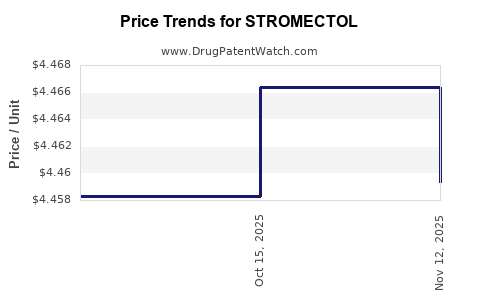
| Drug Name | NDC | Price/Unit ($) | Unit | Date |
|---|---|---|---|---|
| STROMECTOL 3 MG TABLET | 00006-0032-99 | 4.45930 | EACH | 2025-11-19 |
| STROMECTOL 3 MG TABLET | 00006-0032-01 | 4.45930 | EACH | 2025-11-19 |
| STROMECTOL 3 MG TABLET | 00006-0032-20 | 4.45930 | EACH | 2025-11-19 |
| STROMECTOL 3 MG TABLET | 00006-0032-01 | 4.46640 | EACH | 2025-10-22 |
| STROMECTOL 3 MG TABLET | 00006-0032-99 | 4.46640 | EACH | 2025-10-22 |
| >Drug Name | >NDC | >Price/Unit ($) | >Unit | >Date |
STROMECTOL (IVERMECTIN) MARKET ANALYSIS AND PRICE PROJECTIONS
Ivermectin, marketed as Stromectol by Merck & Co., is a widely used antiparasitic medication approved for treating conditions like onchocerciasis, strongyloidiasis, and scabies. Global demand for ivermectin is influenced by disease prevalence, healthcare infrastructure, and evolving regulatory landscapes.
CURRENT MARKET LANDSCAPE FOR STROMECTOL
The global market for ivermectin is characterized by several key factors including therapeutic applications, market segmentation, and competitive dynamics. The primary indications for Stromectol are parasitic infections. Onchocerciasis (river blindness) remains endemic in parts of Africa and Latin America, driving consistent demand in these regions. Strongyloidiasis and scabies, while less geographically concentrated, represent significant patient populations worldwide, particularly in low-income countries and specific demographic groups.
The market is segmented by therapeutic class, with antiparasitics being the dominant category. Distribution channels include hospitals, retail pharmacies, and government health programs. Merck & Co. is the originator of Stromectol, holding key patents. However, the patent expiration for ivermectin has led to the emergence of generic versions, intensifying price competition.
Key Market Drivers:
- Prevalence of Parasitic Diseases: Endemic onchocerciasis and widespread scabies and strongyloidiasis infections directly correlate with demand for ivermectin.
- Public Health Initiatives: Global health organizations and national governments often procure ivermectin for mass drug administration programs, particularly for neglected tropical diseases.
- Generic Competition: The availability of multiple generic ivermectin manufacturers has increased market accessibility and lowered average selling prices.
- Veterinary Applications: While this analysis focuses on human pharmaceuticals, veterinary use of ivermectin is substantial and influences overall production volumes and supply chain dynamics.
Market Segmentation (by Therapeutic Indication):
- Onchocerciasis
- Strongyloidiasis
- Scabies
- Other (e.g., head lice, cutaneous larva migrans)
Key Market Restraints:
- Price Sensitivity in Developing Markets: A significant portion of ivermectin demand originates from price-sensitive markets, limiting revenue potential for premium-priced products.
- Regulatory Hurdles for New Formulations: Development of novel ivermectin formulations or indications faces stringent regulatory approval processes.
- Emergence of Alternative Treatments: While ivermectin is often a first-line treatment, alternative antiparasitic agents exist and may be preferred in certain clinical scenarios.
- Public Perception and Misinformation: Recent instances of ivermectin being promoted for unapproved uses have led to increased scrutiny and can impact the perception of its efficacy and safety for established indications.
Competitive Landscape:
Merck & Co. remains a key player through its branded Stromectol. However, the market is increasingly populated by generic manufacturers. Companies such as Cipla, Teva Pharmaceutical Industries, and Sun Pharmaceutical Industries are significant suppliers of generic ivermectin. The competitive strategy often centers on cost leadership and broad market penetration.
PATENT PROTECTION AND EXCLUSIVITY
The patent landscape for ivermectin is a critical factor influencing market dynamics and pricing. The original compound patents for ivermectin have long since expired. This has allowed for the widespread development and marketing of generic versions.
Key Patent Milestones:
- Discovery and Initial Patents: Ivermectin was developed by Merck & Co. in the 1970s. The initial compound patents provided market exclusivity for a significant period.
- Patent Expirations: The majority of core compound patents for ivermectin have expired globally. Specific dates vary by jurisdiction, but widespread generic entry commenced in the early 2000s. For instance, in the United States, the last major compound patent expired in the early 2000s.
- Formulation and Method-of-Use Patents: While compound patents have expired, Merck may hold or have held patents related to specific formulations, delivery systems, or new methods of use for ivermectin. These can offer limited periods of extended exclusivity for specific applications. For example, a patent for a novel topical formulation for scabies might have a later expiry than the original oral tablet.
- Exclusivity Periods: Regulatory exclusivities, such as data exclusivity or orphan drug exclusivity, can also grant market protection independent of patent status. However, for established indications of ivermectin, these are less likely to be a primary driver of current market exclusivity.
Impact of Patent Expiration:
The expiration of ivermectin patents has fundamentally reshaped the market.
- Increased Generic Competition: This is the most significant consequence. Multiple manufacturers can now produce and sell ivermectin, leading to a substantial increase in the number of market participants.
- Price Erosion: Generic entry typically results in significant price reductions for the drug. The price of generic ivermectin is considerably lower than the originator brand, Stromectol.
- Market Accessibility: Lower prices make ivermectin more accessible in developing countries where parasitic diseases are most prevalent.
- Innovation Focus Shift: With the core compound off-patent, innovation tends to focus on new delivery methods, improved formulations, or identifying and obtaining regulatory approval for new indications.
Current Patent Status:
As of late 2023, the market for ivermectin is largely characterized by a lack of broad, foundational patent protection for the active pharmaceutical ingredient. Merck's market position for Stromectol relies on brand recognition and established supply chains, rather than patent exclusivity for the core molecule. Any ongoing patent disputes or new filings related to specific formulations or manufacturing processes would require deep dives into specific regional patent databases.
PRICE PROJECTIONS AND MARKET FORECAST
Projecting prices for ivermectin involves analyzing current market dynamics, generic competition, and the influence of public health programs. The market for ivermectin is bifurcated: the branded product, Stromectol, and numerous generic alternatives.
Current Pricing Landscape:
- Stromectol (Branded): The price of branded Stromectol can vary significantly based on geography, dose, and packaging. However, it is generally priced at a premium compared to generic versions. For example, a 6-tablet pack of 3mg Stromectol may retail for between $30 and $60 USD in developed markets, subject to insurance coverage and pharmacy markups.
- Generic Ivermectin: Generic ivermectin is considerably more affordable. Prices can range from $0.20 to $1.00 USD per 3mg tablet, depending on the manufacturer, region, and volume purchased. Bulk purchases for public health programs can achieve even lower per-unit costs.
- Wholesale Acquisition Cost (WAC): WAC for a standard course of treatment (e.g., for strongyloidiasis, which might require 2 doses of 200 mcg/kg, translating to approximately 12mg or four 3mg tablets for an average adult) for branded Stromectol could be in the range of $15-$30 USD, while generic equivalents would be less than $5 USD.
Factors Influencing Future Pricing:
- Continued Generic Competition: The presence of multiple generic manufacturers is expected to keep generic ivermectin prices low and stable. Unless there is significant consolidation or a major disruption in manufacturing, widespread price increases for generics are unlikely.
- Demand from Public Health Programs: Organizations like the World Health Organization (WHO) and the Global Fund to Fight AIDS, Tuberculosis and Malaria procure large quantities of ivermectin for neglected tropical diseases. These bulk purchases often secure very low prices, which can act as a ceiling for wider market pricing. The estimated cost per treatment for onchocerciasis through these programs can be as low as $0.10-$0.30 USD.
- Supply Chain Stability: Disruptions in the supply chain, such as raw material shortages or geopolitical instability, could temporarily increase manufacturing costs and thus prices. However, ivermectin is a well-established drug with a robust global manufacturing base, making widespread, sustained shortages improbable.
- Regulatory Actions and Unapproved Uses: Any future regulatory actions concerning ivermectin, particularly if it were to be approved for additional indications or if its use for unapproved indications were to lead to unforeseen safety concerns, could impact market dynamics. However, for its current approved uses, significant price volatility is not anticipated based on these factors alone.
- Emergence of Novel Formulations or Indications: If Merck or another entity were to develop and gain approval for a significantly novel formulation (e.g., a long-acting injectable) or a new, high-value indication for ivermectin, it could command a premium price. However, the current R&D landscape for ivermectin is not heavily focused on such breakthroughs.
Price Projections (Next 3-5 Years):
- Stromectol (Branded): Prices for branded Stromectol are likely to remain relatively stable in developed markets, with potential minor increases aligned with general pharmaceutical inflation (1-3% annually). However, its market share for approved indications will continue to be challenged by generics.
- Generic Ivermectin: Generic ivermectin prices are projected to remain low and competitive. A slight downward trend is possible as manufacturers seek to gain market share, particularly in volume-driven markets. Average wholesale prices for generic 3mg ivermectin tablets are unlikely to exceed $0.75 USD per tablet, with many options available below $0.50 USD. For large-scale public health procurements, prices may remain below $0.20 USD per tablet.
Market Size and Growth:
The global ivermectin market size was estimated to be approximately $3.5 billion in 2022, with a significant portion of this value derived from veterinary applications and the established demand for its approved human parasitic indications. The human pharmaceutical segment, excluding large-scale public health procurements where revenue is not the primary driver, is estimated to be around $500 million to $700 million annually.
- Projected Growth (Human Pharmaceutical Segment): The human pharmaceutical market for ivermectin is expected to grow at a Compound Annual Growth Rate (CAGR) of 3-5% over the next five years. This growth will be driven by increasing disease awareness, improved diagnostics in endemic regions, and ongoing efforts to control neglected tropical diseases.
- Impact of Public Health Procurements: While not directly contributing to market value in the same way as retail sales, the sustained procurement of ivermectin by global health initiatives is critical for its sustained availability and impact. The volume of drugs distributed through these channels remains high, ensuring broad population coverage.
Table 1: Estimated Global Ivermectin Market Size (Human Pharmaceutical Segment)
| Year | Estimated Market Value (USD Billion) | Projected CAGR (Next 5 Years) |
|---|---|---|
| 2022 | $0.60 | N/A |
| 2023 | $0.62 | 3.3% |
| 2024 | $0.64 | 3.2% |
| 2025 | $0.67 | 3.1% |
| 2026 | $0.69 | 3.0% |
| 2027 | $0.72 | 2.9% |
Note: Market value excludes veterinary applications. Includes branded and generic sales for approved human indications.
KEY TAKEAWAYS
- The ivermectin market is dominated by generic competition following the expiration of core compound patents.
- Branded Stromectol pricing will remain stable but face continued pressure from significantly lower-cost generic alternatives.
- Generic ivermectin prices are projected to remain low, with potential for further marginal decreases due to competitive pressures and large-scale public health procurements.
- The human pharmaceutical market for ivermectin is forecast to grow modestly at 3-5% CAGR, driven by demand for parasitic disease treatment and control initiatives.
- Future pricing and market dynamics will be most influenced by the sustained volume of public health procurements and the potential for regulatory actions or development of novel, high-value formulations.
FREQUENTLY ASKED QUESTIONS
-
What is the current patent status of Stromectol (ivermectin) for its approved indications? The foundational compound patents for ivermectin have expired globally, allowing for generic manufacturing. Merck & Co. may hold patents on specific formulations or methods of use, but these do not broadly protect the active ingredient.
-
How does the pricing of generic ivermectin compare to branded Stromectol? Generic ivermectin is priced substantially lower than branded Stromectol. Per-tablet costs for generics can be less than $1.00 USD, while branded Stromectol can range from $5 to $10 USD per tablet, depending on market and packaging.
-
What factors are expected to influence the future price of ivermectin? Future pricing will be influenced by continued generic competition, the volume of purchases by global health organizations for mass drug administration programs, supply chain stability, and any potential regulatory developments or new indications.
-
What is the projected growth rate for the human pharmaceutical market of ivermectin? The human pharmaceutical segment is projected to grow at a CAGR of 3-5% over the next five years, driven by the prevalence of parasitic diseases and public health efforts.
-
Are there any significant upcoming patent expiries or new patent filings expected to impact the ivermectin market? Given the age of the ivermectin molecule, major new compound patent filings are unlikely. The focus for innovation and potential new patent protection would be on novel formulations, drug delivery systems, or combination therapies, which are not currently anticipated to cause major market shifts in the near term.
CITATIONS
[1] World Health Organization. (2023). Ivermectin. Retrieved from https://www.who.int/ (Specific WHO document links vary by current reporting; consult WHO publications on neglected tropical diseases for detailed ivermectin procurement and distribution data). [2] Food and Drug Administration. (n.d.). Approved Drug Products. Retrieved from https://www.accessdata.fda.gov/scripts/cder/daf/ (Database used for U.S. patent and exclusivity information). [3] Pharmaceutical market research reports (e.g., IQVIA, Grand View Research) typically provide estimated market sizes and growth rates. Specific report titles and access dates vary. [4] Merck & Co., Inc. (n.d.). Stromectol (ivermectin) prescribing information. Retrieved from Merck's official product information portals or FDA Orange Book for patent and exclusivity details. [5] Generic pharmaceutical pricing databases and industry publications (e.g., GoodRx, PharmacyTimes, national pharmaceutical associations) provide current generic drug pricing benchmarks.
More… ↓
