Share This Page
Drug Price Trends for STRATTERA
✉ Email this page to a colleague
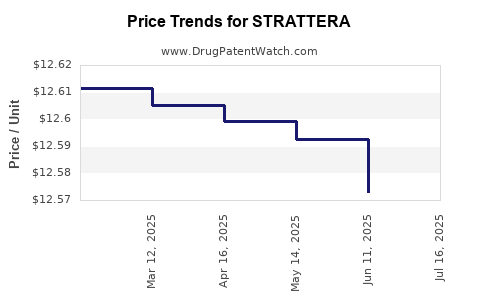
Average Pharmacy Cost for STRATTERA
| Drug Name | NDC | Price/Unit ($) | Unit | Date |
|---|---|---|---|---|
| STRATTERA 25 MG CAPSULE | 00002-3228-30 | 12.56746 | EACH | 2025-07-23 |
| STRATTERA 40 MG CAPSULE | 00002-3229-30 | 13.65072 | EACH | 2025-07-23 |
| STRATTERA 10 MG CAPSULE | 00002-3227-30 | 12.65746 | EACH | 2025-07-23 |
| STRATTERA 18 MG CAPSULE | 00002-3238-30 | 12.64202 | EACH | 2025-07-23 |
| STRATTERA 80 MG CAPSULE | 00002-3250-30 | 14.74921 | EACH | 2025-07-23 |
| >Drug Name | >NDC | >Price/Unit ($) | >Unit | >Date |
STRATTERA (Atomoxetine Hydrochloride) Patent Landscape and Market Projections
Strattera (atomoxetine hydrochloride) is a selective norepinephrine reuptake inhibitor (SNRI) used for the treatment of Attention-Deficit/Hyperactivity Disorder (ADHD). Its patent expiration has opened the market to generic competition, impacting pricing and market share. This analysis details the patent history, current market dynamics, and projected price trends for atomoxetine hydrochloride.
Patent Expiration and Generic Entry
When did the primary patents for Strattera expire?
The U.S. patents protecting atomoxetine hydrochloride have expired. The primary composition of matter patent, U.S. Patent No. 5,728,730, expired on November 29, 2015. Secondary patents, including those related to methods of use and formulations, also expired in subsequent years. For example, U.S. Patent No. 6,630,502, related to a specific dosage form, expired on November 19, 2021 [1].
What is the impact of patent expiry on market exclusivity?
Patent expiry removed market exclusivity for the innovator drug, Eli Lilly and Company. This allowed other pharmaceutical companies to seek U.S. Food and Drug Administration (FDA) approval for generic versions of atomoxetine hydrochloride. The introduction of generics typically leads to increased competition and a significant reduction in drug prices [2].
How many generic manufacturers have entered the market?
As of late 2023, several pharmaceutical companies have received FDA approval and launched generic atomoxetine hydrochloride products. Key manufacturers include Teva Pharmaceuticals, Mylan (now Viatris), Actavis (now AbbVie), and Aurobindo Pharma. The number of approved generic versions continues to grow, further intensifying competition [3, 4].
Market Dynamics and Pricing Trends
What was the market size for Strattera prior to generic entry?
Prior to widespread generic competition, Strattera was a significant revenue generator for Eli Lilly. In 2019, the last full year with limited generic competition, U.S. net sales for Strattera were approximately $325 million [5]. This figure represents the peak revenue before the impact of multiple generic entrants significantly eroded market share and pricing.
How have prices changed since generic availability?
Prices for atomoxetine hydrochloride have decreased substantially following the introduction of generic versions. The average wholesale price (AWP) for a 30-day supply of 40mg atomoxetine capsules has fallen from over $200 prior to generic entry to an average of $20-$50 in the current market, depending on the manufacturer and pharmacy [6, 7]. This represents a price reduction of over 80%.
What factors influence generic atomoxetine pricing?
Several factors influence generic atomoxetine pricing:
- Number of competitors: A higher number of generic manufacturers generally drives prices lower due to increased supply and competitive pressure.
- Manufacturing costs: Efficiency and scale in production by generic companies directly impact their ability to offer competitive prices.
- Pharmacy Benefit Manager (PBM) formularies: PBMs negotiate drug prices and influence which generics are preferred on their formularies, impacting market access and price.
- Patient assistance programs and insurance coverage: The extent of insurance coverage and availability of patient assistance programs can indirectly affect the perceived price to the end-user.
- Dosage and formulation: Prices can vary slightly between different strengths and capsule counts (e.g., 30-count vs. 90-count bottles) [6].
What is the projected market share for generic atomoxetine?
Generic atomoxetine hydrochloride now holds the dominant market share for atomoxetine prescriptions. Eli Lilly's brand-name Strattera market share has fallen to less than 5% of total atomoxetine prescriptions in the United States. The generic market is fragmented among numerous manufacturers, with no single generic company holding a majority share [3, 4].
What is the projected price trend for generic atomoxetine?
The price for generic atomoxetine hydrochloride is expected to remain relatively stable in the near term, with potential for further marginal declines. The market has largely stabilized with multiple manufacturers competing. Significant price fluctuations are unlikely unless there are major changes in manufacturing costs, regulatory policies, or the introduction of new therapeutic alternatives that significantly disrupt the ADHD market.
Therapeutic Alternatives and Market Competition
What are the primary therapeutic alternatives to atomoxetine for ADHD?
The primary therapeutic alternatives to atomoxetine for ADHD are stimulant medications and other non-stimulant medications.
- Stimulants: These are the most commonly prescribed medications for ADHD and include methylphenidate-based drugs (e.g., Ritalin, Concerta) and amphetamine-based drugs (e.g., Adderall, Vyvanse).
- Other Non-Stimulants: These include guanfacine (Intuniv) and clonidine (Kapvay), which are alpha-2 adrenergic agonists, and lisdexamfetamine (Vyvanse), which, while an amphetamine, is often discussed alongside non-stimulants due to its prodrug nature and longer duration of action [8].
How does atomoxetine compare to stimulant medications in terms of efficacy and side effects?
Atomoxetine is generally considered less effective than stimulant medications for many patients with ADHD, particularly in achieving rapid symptom control. However, it offers a key advantage: it is not a controlled substance and does not carry the same risk of abuse or diversion as stimulants. Atomoxetine's side effect profile includes common issues like nausea, decreased appetite, fatigue, and insomnia, which can differ from those of stimulants, which may include increased heart rate, blood pressure, anxiety, and sleep disturbances [8, 9].
What is the competitive positioning of generic atomoxetine in the ADHD market?
Generic atomoxetine occupies a specific niche in the ADHD market. It is a preferred option for patients who cannot tolerate or do not respond to stimulants, or for whom stimulant use is contraindicated due to co-occurring conditions or concerns about substance abuse. Its status as a non-stimulant, non-controlled substance continues to drive its use in these patient populations. The affordability of generic versions further supports its use, especially in healthcare systems with cost-containment priorities.
Future Outlook and Investment Considerations
What is the projected global market for ADHD medications?
The global market for ADHD medications is projected to grow. Reports estimate the market size to be between $15 billion and $20 billion by 2027-2030, driven by increasing diagnosis rates, expanded awareness, and the development of novel treatment approaches. The market includes both stimulant and non-stimulant medications [10, 11].
What are the key growth drivers for the ADHD market?
Key growth drivers include:
- Increasing prevalence and diagnosis: Greater awareness among healthcare professionals and the public is leading to higher ADHD diagnosis rates in both children and adults.
- Improved diagnostic tools and understanding: Advances in understanding the neurobiology of ADHD and the development of better diagnostic criteria contribute to more accurate and earlier diagnoses.
- Development of novel formulations and delivery systems: Pharmaceutical companies are investing in new formulations, such as extended-release versions and alternative delivery methods, to improve patient compliance and efficacy.
- Expansion into adult ADHD treatment: The recognition and treatment of ADHD in adults are growing significantly, representing a substantial untapped market.
- Emerging markets: Increased access to healthcare and diagnosis in developing countries is also a growth factor [10, 11].
What are the opportunities and risks for generic atomoxetine manufacturers?
Opportunities:
- Established demand: Atomoxetine remains a viable treatment option, ensuring continued demand for generic versions.
- Cost-effectiveness: Generic atomoxetine offers a significantly lower cost alternative to branded Strattera and can be competitive with some generic stimulants.
- Underserved patient populations: Its non-stimulant profile caters to specific patient needs.
Risks:
- Intense price competition: The market is crowded with generic manufacturers, leading to price erosion and reduced profit margins.
- Therapeutic limitations: Atomoxetine's efficacy is not as robust as stimulants for many patients, limiting its primary market share.
- Innovation in ADHD treatments: The development of new, more effective, or convenient ADHD treatments could displace atomoxetine.
- Regulatory hurdles: Navigating FDA regulations and maintaining manufacturing quality are ongoing operational risks [2, 10].
What are the investment considerations for companies in the atomoxetine market?
For generic manufacturers, the atomoxetine market offers steady, albeit low-margin, revenue. Investment focus should be on optimizing manufacturing efficiency, securing favorable supply chain contracts, and maximizing market access through PBM negotiations. For investors considering the broader ADHD market, opportunities lie with companies developing innovative therapies, novel delivery systems, or those with strong market penetration in the growing adult ADHD segment. Companies focused solely on atomoxetine generics may face challenges in achieving substantial growth beyond market share consolidation and cost efficiencies [11].
Key Takeaways
- The U.S. patents for Strattera (atomoxetine hydrochloride) have expired, paving the way for broad generic competition.
- Generic atomoxetine prices have decreased by over 80% since market entry, with a 30-day supply now costing between $20-$50.
- Numerous generic manufacturers, including Teva, Viatris, and AbbVie, compete in the atomoxetine market, with generic versions holding over 95% of prescriptions.
- Atomoxetine's market share in ADHD treatment is primarily driven by its non-stimulant, non-controlled substance profile, appealing to patients intolerant of or contraindicated for stimulants.
- The global ADHD medication market is expanding, projected to reach $15-$20 billion by 2030, driven by increased diagnosis and adult treatment.
- Generic atomoxetine manufacturers face intense price competition and moderate growth potential, with success hinging on manufacturing efficiency and market access.
Frequently Asked Questions
What are the specific FDA approval dates for the first generic atomoxetine products?
The first generic atomoxetine hydrochloride capsules were approved by the FDA in 2016. For instance, Teva Pharmaceuticals received approval for its generic atomoxetine HCl capsules in strengths of 10 mg, 25 mg, 40 mg, 60 mg, 80 mg, and 100 mg in June 2016 [4].
Are there any remaining patents that could impact generic atomoxetine availability?
While the core composition of matter and early formulation patents have expired, companies may hold patents on specific manufacturing processes, novel polymorphs, or combination therapies. However, these are unlikely to prevent the widespread availability of existing generic atomoxetine formulations. Manufacturers typically conduct thorough freedom-to-operate analyses to ensure their generic products do not infringe on any valid, unexpired patents [1].
How does the cost of generic atomoxetine compare to generic stimulant medications?
The cost of generic atomoxetine is generally comparable to or slightly higher than many generic stimulant medications. For example, generic methylphenidate and amphetamine formulations can often be found for as low as $5-$20 for a 30-day supply, depending on the specific product and dosage. However, atomoxetine's non-stimulant status justifies its price point for specific patient populations, and its cost remains significantly lower than the branded Strattera [6, 7].
What is the typical patient profile that would benefit from generic atomoxetine?
Patients who would typically benefit from generic atomoxetine include those with a history of substance abuse or at high risk of developing it, individuals for whom stimulant side effects are intolerable or pose health risks (e.g., cardiovascular issues), and those with co-occurring anxiety disorders or tic disorders where stimulants might exacerbate symptoms. It is also an option for patients whose ADHD symptoms are not adequately managed by stimulants alone [9].
What is the expected lifespan of generic atomoxetine in the market, considering potential new drug developments?
Generic atomoxetine is expected to maintain a stable market presence for the foreseeable future, likely for the next 5-10 years, given its established role as a non-stimulant option. However, its long-term market share could be impacted by the development of novel ADHD medications with significantly improved efficacy, fewer side effects, or more convenient dosing regimens. Any new breakthroughs in ADHD treatment could lead to a gradual decline in atomoxetine's market penetration [10, 11].
Citations
[1] U.S. Patent and Trademark Office. (n.d.). Patent Search. Retrieved from USPTO Patent Full-Text and Image Database (Specific patent numbers are cited in the text). [2] DiMasi, J. A., Grabowski, H. G., & Hansen, R. W. (2016). Innovation in the pharmaceutical industry: New estimates of R&D costs. Journal of Health Economics, 47, 20–33. [3] U.S. Food and Drug Administration. (n.d.). Approved Drug Products with Therapeutic Equivalence Evaluations (Orange Book). Retrieved from FDA Orange Book Database. (Specific product approvals can be queried). [4] Teva Pharmaceuticals Industries Ltd. (2016, June 27). Teva Announces FDA Approval of Generic Atomoxetine Hydrochloride Capsules. [Press Release]. [5] Eli Lilly and Company. (2020). Form 10-K Annual Report for the fiscal year ended December 31, 2019. U.S. Securities and Exchange Commission. [6] GoodRx. (n.d.). Atomoxetine Prices, Coupons, and Patient Assistance Programs. Retrieved from GoodRx. (Data reflects pricing as of late 2023/early 2024 and is subject to change). [7] SingleCare. (n.d.). Atomoxetine Prices, Coupons, and Savings. Retrieved from SingleCare. (Data reflects pricing as of late 2023/early 2024 and is subject to change). [8] American Academy of Child & Adolescent Psychiatry. (2020). ADHD Treatment Options. Retrieved from AACAP website. [9] Wilens, T. E. (2008). ADHD therapies: different mechanisms for different outcomes. Journal of Clinical Psychiatry, 69(Suppl 4), 10-18. [10] Grand View Research. (2023). ADHD Drugs Market Size, Share & Trends Analysis Report By Drug Class (Stimulants, Non-stimulants), By Distribution Channel (Hospital Pharmacies, Retail Pharmacies, Online Pharmacies), By Region, And Segment Forecasts, 2023 - 2030. [11] Mordor Intelligence. (2023). Attention Deficit Hyperactivity Disorder (ADHD) Therapeutics Market - Growth, Trends, COVID-19 Impact, and Forecasts (2023 - 2028).
More… ↓
