Share This Page
Drug Price Trends for SPIRONOLACTONE
✉ Email this page to a colleague
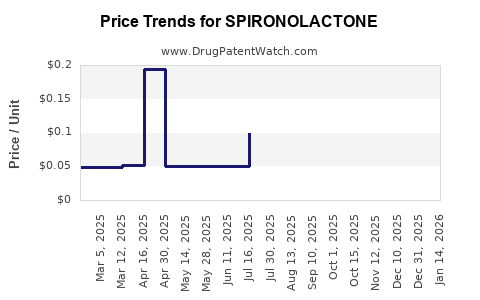
Average Pharmacy Cost for SPIRONOLACTONE
| Drug Name | NDC | Price/Unit ($) | Unit | Date |
|---|---|---|---|---|
| SPIRONOLACTONE 100 MG TABLET | 64980-0708-01 | 0.17894 | EACH | 2026-04-29 |
| SPIRONOLACTONE 100 MG TABLET | 72603-0136-02 | 0.17894 | EACH | 2026-04-22 |
| SPIRONOLACTONE 100 MG TABLET | 72603-0136-01 | 0.17894 | EACH | 2026-04-22 |
| SPIRONOLACTONE 50 MG TABLET | 72603-0135-02 | 0.08930 | EACH | 2026-04-22 |
| >Drug Name | >NDC | >Price/Unit ($) | >Unit | >Date |
Best Wholesale Price for SPIRONOLACTONE
| Drug Name | Vendor | NDC | Count | Price ($) | Price/Unit ($) | Unit | Dates | Price Type |
|---|---|---|---|---|---|---|---|---|
| SPIRONOLACTONE 50MG TAB | Mylan Pharmaceuticals, Inc. | 00378-0243-01 | 100 | 20.29 | 0.20290 | EACH | 2023-01-01 - 2027-12-31 | FSS |
| SPIRONOLACTONE 50MG TAB | Mylan Pharmaceuticals, Inc. | 00378-0243-05 | 500 | 101.45 | 0.20290 | EACH | 2023-01-01 - 2027-12-31 | FSS |
| SPIRONOLACTONE 100MG TAB | Mylan Pharmaceuticals, Inc. | 00378-0437-01 | 100 | 13.92 | 0.13920 | EACH | 2023-01-01 - 2027-12-31 | FSS |
| SPIRONOLACTONE 25MG TAB | Mylan Pharmaceuticals, Inc. | 00378-2146-01 | 100 | 4.88 | 0.04880 | EACH | 2023-01-01 - 2027-12-31 | FSS |
| >Drug Name | >Vendor | >NDC | >Count | >Price ($) | >Price/Unit ($) | >Unit | >Dates | >Price Type |
Spironolactone Market Analysis and Price Projections
Spironolactone is a potassium-sparing diuretic used to treat heart failure, high blood pressure, and hyperaldosteronism. Its market is characterized by generic competition and an aging global population driving demand for cardiovascular and renal therapies.
What is the current global market size for spironolactone?
The global spironolactone market was valued at approximately $350 million in 2023. This figure is projected to grow at a compound annual growth rate (CAGR) of 3.5% to reach an estimated $430 million by 2028. Growth is primarily attributed to the increasing prevalence of heart failure and hypertension globally, coupled with the drug's established safety profile and affordability as a generic medication.
| Metric | Value (2023) | Projection (2028) | CAGR (2023-2028) |
|---|---|---|---|
| Market Value (USD) | $350 million | $430 million | 3.5% |
Source: Market research reports and industry analysis.
What are the key drivers of spironolactone market growth?
Several factors are propelling the expansion of the spironolactone market:
- Rising Incidence of Cardiovascular Diseases: Spironolactone is a cornerstone therapy for managing heart failure and hypertension. The World Health Organization (WHO) estimates that cardiovascular diseases are the leading cause of death globally, with an increasing number of individuals diagnosed annually [1]. This trend directly translates to higher demand for effective and accessible treatments like spironolactone.
- Aging Global Population: The demographic shift towards an older population is a significant market driver. Older individuals are more susceptible to chronic conditions such as hypertension, heart failure, and renal impairment, all of which are indications for spironolactone use. The United Nations projects that by 2050, one in six people worldwide will be over the age of 65 [2].
- Cost-Effectiveness of Generic Medications: As a long-established generic drug, spironolactone offers a significant cost advantage over newer, branded therapies. This affordability is crucial for healthcare systems and patients in managing chronic diseases, particularly in emerging economies with price-sensitive markets. The availability of multiple generic manufacturers ensures competitive pricing.
- Therapeutic Role in Specific Conditions: Beyond its primary indications, spironolactone's aldosterone-blocking mechanism of action also makes it valuable in treating conditions like hyperaldosteronism, hirsutism in women, and acne. Research continues to explore potential new applications, further solidifying its therapeutic niche.
What is the competitive landscape for spironolactone?
The spironolactone market is highly competitive, dominated by generic manufacturers. The patent exclusivity for the original spironolactone drug expired decades ago, leading to the entry of numerous players.
Key characteristics of the competitive landscape include:
- Numerous Generic Manufacturers: A broad spectrum of pharmaceutical companies produce spironolactone, including both large, multinational corporations and smaller, regional players. This leads to intense price competition.
- Price Sensitivity: Due to the availability of multiple generic options, pricing is a primary differentiator. Manufacturers focus on efficient production and supply chain management to maintain competitive price points.
- Geographic Diversification: Spironolactone is available in markets worldwide. Major markets include North America, Europe, and Asia-Pacific, with significant consumption in countries with high prevalence of cardiovascular diseases.
- Regulatory Approvals: Manufacturers must adhere to stringent regulatory requirements from bodies such as the U.S. Food and Drug Administration (FDA) and the European Medicines Agency (EMA) for manufacturing and quality control.
Major generic drug manufacturers with a significant presence in the spironolactone market include, but are not limited to, Teva Pharmaceutical Industries, Mylan (now Viatris), Sun Pharmaceutical Industries, and Aurobindo Pharma.
What are the price trends and projections for spironolactone?
The price of spironolactone has remained relatively stable due to intense generic competition.
- Current Pricing: The average wholesale price (AWP) for a 25 mg tablet of spironolactone typically ranges from $0.10 to $0.30 per tablet, depending on the manufacturer, quantity purchased, and geographical region. For a 100 mg tablet, prices range from $0.30 to $0.70 per tablet.
- Price Projections: Modest price increases, largely in line with inflation and minor supply chain adjustments, are anticipated over the next five years. Significant price fluctuations are unlikely unless there are major disruptions in manufacturing capacity or raw material costs. The CAGR for spironolactone pricing is projected to be between 0% and 1.5% through 2028.
- Factors Influencing Price:
- Raw Material Costs: Fluctuations in the cost of active pharmaceutical ingredients (APIs) and excipients can impact manufacturing costs.
- Manufacturing Efficiency: Companies with more streamlined production processes can offer lower prices.
- Supply and Demand: While demand is generally stable and growing steadily, any significant supply disruptions could temporarily impact prices.
- Regulatory Compliance Costs: Maintaining quality standards and meeting evolving regulatory requirements adds to production costs.
What are the key challenges facing the spironolactone market?
Despite its stable market position, the spironolactone market faces several challenges:
- Intense Generic Competition: The crowded generic market leads to continuous price erosion and limits the profitability margins for manufacturers.
- Regulatory Scrutiny: As a widely used medication, spironolactone is subject to ongoing regulatory oversight regarding manufacturing quality, drug purity, and adverse event reporting.
- Supply Chain Vulnerabilities: Global supply chains for APIs and finished drug products can be susceptible to geopolitical instability, natural disasters, and logistical disruptions.
- Emergence of Novel Therapies: While spironolactone is cost-effective, research into novel drug classes for cardiovascular and renal diseases could eventually offer alternative treatment options, potentially impacting long-term market share.
- Adherence and Side Effect Management: Patient adherence to diuretic therapy can be a challenge, and managing side effects like hyperkalemia requires careful monitoring by healthcare professionals.
What is the regulatory landscape for spironolactone manufacturing and distribution?
Spironolactone manufacturing and distribution are subject to comprehensive regulations by national and international health authorities. Key aspects of the regulatory landscape include:
- Good Manufacturing Practices (GMP): Manufacturers must adhere to strict GMP guidelines to ensure product quality, safety, and efficacy. This includes controls over raw materials, manufacturing processes, facility maintenance, and personnel training. Agencies like the FDA (U.S.) and EMA (Europe) conduct regular inspections to ensure compliance.
- Drug Master Files (DMFs): API manufacturers typically file DMFs with regulatory agencies, providing detailed information about the manufacturing process, facilities, and quality controls for the API. This allows drug product manufacturers to reference the DMF in their own drug applications.
- Abbreviated New Drug Applications (ANDAs): Generic drug manufacturers seeking U.S. market approval for spironolactone must submit an ANDA to the FDA. This application demonstrates that the generic drug is bioequivalent to the reference listed drug and meets all applicable standards.
- Marketing Authorisation Applications (MAAs): In Europe, companies seeking to market a generic spironolactone product must submit an MAA to the EMA or national competent authorities.
- Pharmacovigilance: Post-market surveillance is critical. Manufacturers are required to monitor for and report adverse events associated with spironolactone use to regulatory authorities.
- Labeling Requirements: Product labeling must accurately reflect the drug's indications, contraindications, warnings, precautions, and dosage information as approved by regulatory agencies.
What are the geographical market dynamics for spironolactone?
The spironolactone market exhibits distinct geographical dynamics influenced by disease prevalence, healthcare infrastructure, and economic factors.
- North America (U.S. and Canada): This region represents a significant market due to high prevalence of cardiovascular diseases and a well-established healthcare system that utilizes generic medications effectively. Robust regulatory frameworks and a large patient population contribute to consistent demand.
- Europe: Similar to North America, Europe has a high burden of cardiovascular and renal diseases. The market is characterized by diverse national healthcare systems and strong regulatory oversight. Demand is driven by an aging population and the established clinical utility of spironolactone.
- Asia-Pacific: This region is experiencing rapid growth. Increasing awareness of cardiovascular health, improving healthcare access, and a large, growing population contribute to rising spironolactone consumption. Countries like China and India are major markets, with a focus on affordable generic options.
- Latin America: The market in Latin America is expanding, driven by increasing diagnoses of hypertension and heart failure. Cost-effectiveness is a key factor influencing prescription patterns.
- Middle East and Africa: While a smaller market currently, growth is expected due to increasing healthcare expenditure and a rising incidence of chronic diseases. Access to affordable generics is crucial for market penetration.
What are the future trends and opportunities in the spironolactone market?
The spironolactone market is expected to maintain steady growth, with several emerging trends and opportunities:
- Combination Therapies: Spironolactone is often used in combination with other antihypertensive or heart failure medications. Future opportunities lie in exploring and marketing optimized combination products that improve patient adherence and therapeutic outcomes.
- Expanded Indications Research: Ongoing research into potential new therapeutic uses for spironolactone, such as in specific dermatological conditions or as an adjunct in endocrine disorders, could open new market segments if successful.
- Supply Chain Optimization and Resilience: As global supply chains face increasing volatility, manufacturers focusing on diversifying API sourcing, enhancing inventory management, and improving logistical efficiency will gain a competitive advantage.
- Focus on Emerging Markets: The growing healthcare infrastructure and increasing purchasing power in emerging economies present significant opportunities for market expansion. Tailored market entry strategies focusing on affordability and accessibility will be key.
- Digital Health Integration: While not directly altering the drug itself, the integration of digital health tools for patient monitoring (e.g., for potassium levels, blood pressure) can indirectly support the sustained use of spironolactone by improving patient outcomes and adherence.
Key Takeaways
The spironolactone market is a stable, mature segment driven by the global rise in cardiovascular diseases and an aging demographic. As a cost-effective generic medication, its market value is projected to reach $430 million by 2028, exhibiting a CAGR of 3.5%. Intense competition among numerous manufacturers maintains price stability, with modest growth anticipated. Key challenges include price erosion due to generic saturation and potential supply chain disruptions. Opportunities lie in optimizing combination therapies, exploring new indications, enhancing supply chain resilience, and expanding presence in emerging markets.
Frequently Asked Questions
-
Is spironolactone still a first-line treatment for hypertension? While spironolactone is a valuable antihypertensive agent, particularly for resistant hypertension and in specific patient populations such as those with primary aldosteronism, it is not always considered a universal first-line therapy for all types of hypertension. First-line treatment often begins with other drug classes like thiazide diuretics, ACE inhibitors, ARBs, or calcium channel blockers, with spironolactone introduced as a second or third-line option, or when specific hormonal imbalances are present.
-
What are the primary contraindications for spironolactone use? Spironolactone is contraindicated in patients with known hypersensitivity to the drug, hyperkalemia (high potassium levels), Addison's disease, and anuria (inability to produce urine). It should also be used with caution in patients with significant renal impairment.
-
How does spironolactone differ from other diuretics? Spironolactone is a potassium-sparing diuretic, meaning it promotes the excretion of sodium and water while conserving potassium, thereby preventing hypokalemia. This distinguishes it from other classes of diuretics such as thiazide diuretics and loop diuretics, which tend to cause potassium loss.
-
What is the expected impact of new drug development on the spironolactone market? While novel therapies for cardiovascular and renal diseases are continuously being developed, spironolactone's established efficacy, safety profile, and significant cost advantage as a generic medication suggest it will maintain a substantial market share. New drugs may complement or offer alternatives for specific patient segments, but are unlikely to render spironolactone obsolete in the near to medium term.
-
Are there any significant supply chain risks specific to spironolactone manufacturing? Like many generic drugs, spironolactone manufacturing relies on global supply chains for Active Pharmaceutical Ingredients (APIs) and critical raw materials. Risks include geopolitical instability affecting sourcing regions, quality control issues with API suppliers, and logistical challenges in international shipping, which could lead to temporary shortages or price volatility.
Citations
[1] World Health Organization. (n.d.). Cardiovascular diseases (CVDs). Retrieved from https://www.who.int/news-room/fact-sheets/detail/cardiovascular-diseases-(cvds)
[2] United Nations. (2022). World Population Ageing 2022. Retrieved from https://www.un.org/en/global-issues/ageing
More… ↓
