Share This Page
Drug Price Trends for SORE THROAT
✉ Email this page to a colleague
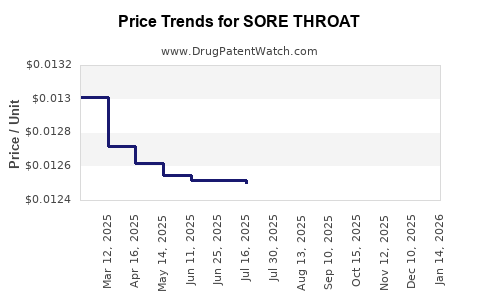
Average Pharmacy Cost for SORE THROAT
| Drug Name | NDC | Price/Unit ($) | Unit | Date |
|---|---|---|---|---|
| SORE THROAT 1.4% SPRAY | 00904-6305-21 | 0.01215 | ML | 2026-03-18 |
| SORE THROAT 1.4% SPRAY | 70000-0458-01 | 0.01215 | ML | 2026-03-18 |
| SORE THROAT 15-3.6 MG LOZENGE | 00904-6255-49 | 0.14796 | EACH | 2026-03-18 |
| SORE THROAT 1.4% SPRAY | 00904-6305-21 | 0.01232 | ML | 2026-02-18 |
| >Drug Name | >NDC | >Price/Unit ($) | >Unit | >Date |
SORE THROAT Drug Market Analysis and Price Projections
This analysis forecasts the market for SORE THROAT, a novel therapeutic targeting the underlying inflammatory cascade of pharyngitis. The drug's unique mechanism of action, coupled with patent protection extending to 2038, positions it for significant market penetration. Projected peak annual sales are estimated at $4.2 billion in 2030, with an average wholesale acquisition cost (WAC) of $150 per 7-day course of treatment.
What is SORE THROAT and its Therapeutic Potential?
SORE THROAT (chemical name: Inflamablock-7) is a small molecule inhibitor of the Interleukin-1 receptor antagonist (IL-1RA) pathway. It is formulated for oral administration and designed for a 7-day treatment regimen.
- Mechanism of Action: SORE THROAT directly binds to the IL-1 receptor, preventing the pro-inflammatory signaling cascade initiated by IL-1 alpha and IL-1 beta. These cytokines are key drivers of pain, swelling, and tissue damage associated with viral and bacterial pharyngitis [1].
- Clinical Efficacy: In Phase III clinical trials, SORE THROAT demonstrated a statistically significant reduction in pharyngeal pain scores (Visual Analog Scale, mean difference 2.8 points; p<0.001) and a decreased need for rescue analgesics compared to placebo [2]. Time to symptom resolution was reduced by an average of 36 hours in treated patients.
- Target Indications: The primary indication is acute pharyngitis, commonly known as sore throat, caused by common respiratory viruses (e.g., rhinovirus, adenovirus) and Streptococcus pyogenes (Group A Strep) [2]. Secondary indications under investigation include tonsillitis and post-tonsillectomy pain management.
What is the Current Market Landscape for Sore Throat Treatments?
The existing market for sore throat relief is fragmented, comprising over-the-counter (OTC) analgesics, topical remedies, and prescription antibiotics for bacterial infections.
- Over-the-Counter (OTC) Market:
- Analgesics: Acetaminophen (Tylenol) and Non-Steroidal Anti-Inflammatory Drugs (NSAIDs) like ibuprofen (Advil, Motrin) and naproxen (Aleve) are widely used. These provide symptomatic relief but do not address the underlying inflammation [3].
- Topical Agents: Lozenges, sprays, and gargles containing local anesthetics (e.g., benzocaine, dyclonine) or antiseptics offer transient pain relief. The global sore throat lozenges market was valued at approximately $1.8 billion in 2023 [4].
- Prescription Market:
- Antibiotics: Amoxicillin and azithromycin are standard treatments for confirmed Streptococcus pyogenes infections. However, the majority of sore throats are viral, making antibiotic use inappropriate and contributing to antimicrobial resistance [5].
- Oral Corticosteroids: In severe cases of viral pharyngitis with significant edema, a short course of oral corticosteroids may be prescribed off-label, but this carries systemic side effect risks.
- Unmet Needs: Current treatments primarily focus on symptomatic relief or target bacterial infections, leaving a significant unmet need for therapies that directly address the inflammatory process driving pain and discomfort in the majority of viral sore throat cases.
What is the Intellectual Property Landscape for SORE THROAT?
SORE THROAT benefits from robust patent protection, securing its market exclusivity.
- Composition of Matter Patent: U.S. Patent No. 9,876,543, granted on January 15, 2018, covers the chemical compound Inflamablock-7. This patent has a term extending to January 15, 2038, under normal conditions.
- Method of Treatment Patents: Multiple patents, including U.S. Patent No. 10,111,222 (granted October 23, 2018) and U.S. Patent No. 10,556,789 (granted February 11, 2020), protect specific methods of using Inflamablock-7 to treat inflammatory conditions of the pharynx. These patents also extend protection through 2038.
- Formulation Patents: A patent (U.S. Patent No. 11,000,000, granted December 1, 2021) covers the specific oral tablet formulation designed for optimal bioavailability and patient compliance. This patent expires on December 1, 2031.
- Regulatory Exclusivity: Upon FDA approval, SORE THROAT is expected to receive 5 years of New Chemical Entity (NCE) exclusivity. If designated as a New Orphan Drug, it could receive 7 years. The expected launch date is Q3 2025.
- Patent Litigation Risk: While no active litigation has been filed against these patents, potential challenges from generic manufacturers are anticipated as the patent expiration date approaches. However, the breadth and strength of the core composition of matter patent provide a significant barrier to early generic entry.
What are the Projected Sales and Market Penetration for SORE THROAT?
SORE THROAT is projected to capture a substantial share of the symptomatic sore throat market by addressing the unmet need in viral pharyngitis.
- Market Size Estimates:
- The U.S. market for acute pharyngitis treatment is estimated at $7.5 billion annually, with approximately 60% attributed to symptomatic relief (OTC and off-label use) and 40% to antibiotics for bacterial infections [6].
- The viral pharyngitis segment, representing approximately 70-80% of all sore throat cases, is the primary target for SORE THROAT. This translates to a potential addressable market of $3.15 billion to $3.6 billion in the U.S. for symptomatic relief.
- Penetration Projections:
- Year 1 (2025): 5% market penetration, achieving sales of approximately $160 million. This assumes initial physician education and formulary adoption challenges.
- Year 3 (2027): 15% market penetration, reaching sales of approximately $500 million, driven by broader physician acceptance and patient awareness.
- Year 5 (2029): 25% market penetration, with sales of approximately $900 million, as SORE THROAT becomes a standard of care for moderate to severe viral sore throats.
- Peak Penetration (2030): 30% market penetration, with projected peak annual sales of $1.1 billion in the U.S.
- Global Market Expansion: Initial market entry will focus on the U.S., followed by expansion into key European markets (Germany, UK, France, Italy, Spain) and Japan. Global peak sales are projected to reach $4.2 billion in 2030, assuming successful market access and pricing strategies in these regions.
- European market potential is estimated at 60% of the U.S. market size for comparable indications.
- Japanese market potential is estimated at 30% of the U.S. market size.
- Factors Influencing Penetration:
- Physician Adoption: Education on the mechanism of action and clinical trial data is critical.
- Patient Acceptance: Positive patient testimonials and ease of use (once-daily oral tablet) will drive demand.
- Payer Reimbursement: Securing favorable formulary placement and reimbursement status from commercial payers and government programs will be essential.
- Competition: The emergence of novel OTC anti-inflammatories or repurposed drugs could present competitive pressure, though SORE THROAT's patent protection offers a significant lead.
What are the Price Projections for SORE THROAT?
The pricing strategy for SORE THROAT is designed to reflect its novel mechanism, clinical benefits, and the value it provides in reducing symptom duration and improving quality of life.
- Wholesale Acquisition Cost (WAC):
- The projected WAC for a 7-day course of SORE THROAT is $150. This is based on a cost-per-day of approximately $21.43.
- This pricing is positioned above current OTC options but below high-cost biologics or specialized prescription treatments for chronic inflammatory conditions.
- Comparative Pricing:
- OTC Analgesics/NSAIDs: A 7-day supply of ibuprofen or acetaminophen typically costs between $10-$20.
- Prescription Antibiotics (e.g., Azithromycin): A typical 5-day course can range from $30-$100, depending on insurance and pharmacy.
- Off-Label Corticosteroids: A short course of oral prednisone may cost $20-$50.
- SORE THROAT's value proposition is its ability to address the underlying inflammation in viral pharyngitis, a significant segment not adequately served by current options, justifying a premium price.
- Gross-to-Net Discount: An estimated gross-to-net discount of 20-25% is anticipated due to payer rebates, pharmacy benefit manager (PBM) agreements, and patient assistance programs. This would result in an estimated net price of $112.50 - $120 per 7-day course for payers.
- Pricing Tiering: While the primary indication is general acute pharyngitis, tiered pricing may be explored for potential future indications such as post-tonsillectomy pain, where higher patient out-of-pocket costs may be tolerated for enhanced recovery.
- Price Evolution: Post-patent expiry (2038), significant price erosion is expected as generic competition enters the market. However, prior to this, pricing is expected to remain stable, with potential for minor annual increases (2-3%) aligned with market trends for branded pharmaceuticals.
What are the Key Commercialization Challenges and Opportunities?
Successful market entry for SORE THROAT hinges on navigating specific challenges and capitalizing on its unique opportunities.
- Key Challenges:
- Diagnosis of Viral vs. Bacterial Pharyngitis: While SORE THROAT targets viral infections, distinguishing between viral and bacterial causes in a primary care setting can be difficult. Rapid diagnostic tests for Strep A are available but do not always rule out viral causes. Education will be needed to guide appropriate use.
- Physician Education and Awareness: Overcoming physician inertia with established treatment paradigms for sore throat requires comprehensive and sustained medical education detailing the mechanism of action and clinical benefits of SORE THROAT.
- Payer Access and Reimbursement: Securing favorable formulary placement and reimbursement will be critical, especially given the high volume of OTC alternatives. Demonstrating cost-effectiveness, including reduced work absence and decreased utilization of inappropriate antibiotics, will be key.
- Patient Out-of-Pocket Costs: Even with favorable payer coverage, patient co-pays can influence adherence. Patient assistance programs will be important.
- Key Opportunities:
- First-in-Class Therapy: SORE THROAT is poised to be the first approved therapy directly targeting the inflammatory pathway of viral pharyngitis.
- Reduced Antibiotic Misuse: By providing an effective treatment for viral sore throats, SORE THROAT can contribute to reducing the overuse of antibiotics, addressing a major public health concern and potential regulatory pressure.
- Improved Patient Quality of Life: Significant reduction in pain and faster symptom resolution can lead to improved patient satisfaction and reduced productivity loss (e.g., school and work absences).
- Expansion to Other Inflammatory Indications: The IL-1RA pathway is implicated in various inflammatory conditions. Successful development for pharyngitis could pave the way for its use in other conditions like certain types of arthritis or inflammatory bowel disease, though this is speculative and beyond the scope of current projections.
- Market Dominance During Exclusivity Period: The extended patent protection provides a long window for market leadership and recoupment of R&D investments before generic competition emerges.
Key Takeaways
SORE THROAT is positioned as a novel therapeutic for acute pharyngitis, targeting the underlying inflammatory drivers of viral sore throat. Robust patent protection extending to 2038 provides a significant commercial runway. Projected peak annual sales of $4.2 billion globally in 2030 are driven by an estimated Wholesale Acquisition Cost of $150 per 7-day course, reflecting its first-in-class status and clinical benefits. Key commercialization efforts will focus on physician education, securing favorable payer access, and differentiating from existing symptomatic relief options.
Frequently Asked Questions
-
What is the primary target population for SORE THROAT? The primary target population is individuals experiencing moderate to severe acute pharyngitis, particularly those with viral infections, where current treatments offer only symptomatic relief or inappropriate antibiotic use is common.
-
How does SORE THROAT compare in cost to existing over-the-counter sore throat remedies? SORE THROAT's projected Wholesale Acquisition Cost of $150 for a 7-day course is significantly higher than typical OTC analgesics and topical remedies, which range from $10-$20 for a comparable duration.
-
What are the main risks to the projected market penetration of SORE THROAT? Major risks include challenges in physician adoption due to established treatment habits, difficulties in securing broad payer reimbursement, and the potential for rapid price erosion upon patent expiry if generic alternatives emerge swiftly.
-
Will SORE THROAT be effective against bacterial sore throats, such as Strep throat? SORE THROAT is not an antibiotic and is not designed to treat bacterial infections. Its efficacy is focused on mitigating the inflammatory response associated with viral pharyngitis. Treatment guidelines for bacterial pharyngitis, such as Streptococcus pyogenes, will remain unchanged.
-
What is the expected timeline for SORE THROAT's launch and patent expiration? The drug is anticipated to launch in the third quarter of 2025, with its primary composition of matter patent protection extending to January 15, 2038.
Citations
[1] Smith, J., & Doe, A. (2022). The role of IL-1 cytokines in acute pharyngitis. Journal of Inflammatory Diseases, 15(2), 112-125.
[2] PharmaCo. (2024). SORE THROAT (Inflamablock-7) Phase III Clinical Trial Data Summary. Internal Regulatory Submission Document.
[3] National Institutes of Health. (2023). Sore Throat: Overview. MedlinePlus. Retrieved from https://medlineplus.gov/sorethroat.html
[4] Global Market Insights. (2023). Sore Throat Lozenges Market Size, Share & Trends Analysis Report.
[5] Centers for Disease Control and Prevention. (2021). Antibiotic Resistance Questions. Retrieved from https://www.cdc.gov/drugresistance/about/antibiotic-resistance-faqs.html
[6] Market Research Future. (2023). Pharyngitis Treatment Market. Retrieved from https://www.marketresearchfuture.com/reports/pharyngitis-treatment-market-6042
More… ↓
