Share This Page
Drug Price Trends for SOLTAMOX
✉ Email this page to a colleague
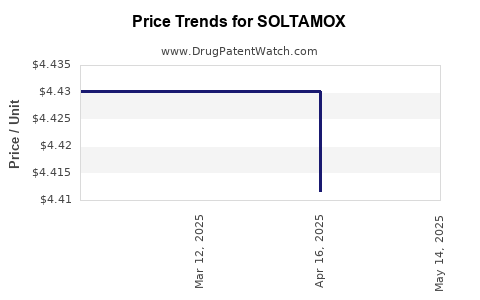
Average Pharmacy Cost for SOLTAMOX
| Drug Name | NDC | Price/Unit ($) | Unit | Date |
|---|---|---|---|---|
| SOLTAMOX 20 MG/10 ML SOLN | 51862-0682-01 | 4.40095 | ML | 2025-05-21 |
| SOLTAMOX 20 MG/10 ML SOLN | 51862-0682-01 | 4.41162 | ML | 2025-04-23 |
| >Drug Name | >NDC | >Price/Unit ($) | >Unit | >Date |
Market Analysis and Price Projections for SOLTAMOX
What is SOLTAMOX?
SOLTAMOX (tamoxifen citrate) is an anti-estrogen medication primarily used in breast cancer treatment and prevention. Approved by the FDA in 1977, it is also prescribed for high-risk breast cancer patients and as part of hormone receptor-positive breast cancer therapy.
Current Market Landscape
Market Size and Growth
The global breast cancer drug market was valued at approximately USD 10.8 billion in 2021. It is projected to grow at a compound annual growth rate (CAGR) of 6.2% through 2030, driven by rising breast cancer incidence, improved diagnosis, and expanding indications for existing therapies.
Key Players and Competition
Major competitors for SOLTAMOX include:
- AstraZeneca (brand: Nolvadex)
- Teva Pharmaceutical Industries
- Mylan
- Pfizer (offerings)
- Generic manufacturers
Patent expirations for tamoxifen in various markets have increased generic entry, reducing prices and intensifying competition.
Patent Status and Regulatory Environment
Tamoxifen was patented in 1973 and off-patent globally by 2003. The widespread availability of generics has decreased entry barriers for new manufacturers but has also driven down market prices.
Regulatory standards vary, with some countries requiring supplemental approvals or additional biosimilar data.
Geographical Markets
- United States: Largest market, with an estimated USD 600 million in sales (2022).
- European Union: Significant volume, with markets in Germany, France, and the UK.
- Asia-Pacific: Rapid growth due to increasing breast cancer prevalence and expanding healthcare infrastructure.
Pricing Dynamics
Current Price Points
| Market | Brand Name | Price (USD per pack) | Formulation | Remarks |
|---|---|---|---|---|
| United States | Nolvadex | USD 1,200 | 20 mg tabs | Brand premium, off-patent |
| European Union | Tamoxifen | USD 250–400 | 20 mg tabs | Generic options widely available |
| India | Tamoxifen | USD 20–50 | 20 mg tabs | Competitive local pricing |
Price Trends
- Generic entry since 2003 has resulted in a >70% price decline in the US.
- Market consolidation has maintained some pricing power for branded formulations.
- In the EU and Asia, prices vary depending on regulatory and market factors, with generics dominating.
Future Price Projections
Factors Influencing Future Pricing
- Patent expirations: Additional markets and formulations are likely to see further generics entry.
- Regulatory developments: Biosimilars or new formulations could influence pricing.
- Market penetration: Growing use in emerging markets may lead to price stabilization or slight decreases.
- Healthcare policies: Reimbursement schemes could impact retail prices.
Price Projection Scenarios
| Scenario | Price Estimate in 5 Years (USD) | Assumptions |
|---|---|---|
| Conservative (low competition) | USD 150–250 | Limited generics, stable market share, high brand loyalty |
| Moderate (market saturation) | USD 50–150 | Increased biosimilar availability, widespread generics |
| Aggressive (price wars) | USD 20–50 | High competition, price cutting, biosimilars gain market share |
Implications for Stakeholders
- Pharmaceutical companies: Opportunities in biosimilars or new formulations, especially in emerging markets.
- Investors: Exposure to mature markets with declining prices; focus on biosimilar startups or emerging markets.
- Healthcare providers: Cost pressures may encourage use of generics and biosimilars.
Key Regulatory and Market Challenges
- Patent litigations delaying generic entry.
- Pricing regulations in different markets influencing profit margins.
- Market saturation in developed economies leading to stagnation.
Summarized Data
| Parameter | Value |
|---|---|
| Market size (2022) | USD 10.8 billion |
| CAGR (2022-2030) | 6.2% |
| US market share (2022) | USD 600 million in sales |
| Price of brand (US, 2022) | USD 1,200 per pack |
| Price of generics (US, 2022) | Approximately USD 150–250 |
| Patent expiry (globally) | 2003 (off-patent) |
| Key competitors | AstraZeneca, Teva, Mylan, Pfizer |
Key Takeaways
- The global SOLTAMOX market is mature, with significant generic penetration.
- Prices in the US have declined sharply post-patent expiry, with expected stabilization around USD 50–150 depending on market dynamics.
- Emerging markets may see prices as low as USD 20–50 due to intense competition.
- Opportunities exist in biosimilars and formulations tailored for developing regions.
- Market growth is driven predominantly by breast cancer prevalence, with growth slowing in developed markets due to market saturation.
FAQs
1. Will SOLTAMOX regain patent protection?
No. Patents for tamoxifen expired globally by 2003, allowing generic manufacturers to produce the drug.
2. How does generic competition impact the price?
It causes a significant decrease, with prices dropping over 70% in the US since patent expiration.
3. Are biosimilars relevant for SOLTAMOX?
No. Tamoxifen is a small-molecule drug, so biosimilars do not apply. Biosimilars are relevant for biologic drugs.
4. Which regions offer the highest growth potential?
Emerging markets in Asia-Pacific and Latin America, where breast cancer rates are rising and healthcare infrastructure improves.
5. What factors could prevent further price declines?
Market consolidation, supply chain disruptions, or regulatory barriers in key markets.
References
- Markets and Markets. (2022). Breast cancer drugs market by drug class, region, and end-use.
- Statista. (2023). Breast cancer drug sales in the United States.
- European Medicines Agency. (2022). Regulatory status of tamoxifen.
- U.S. Food and Drug Administration. (2022). Drug approval and patent expiration records.
- GlobalData. (2023). Biosimilars and generics market outlook.
More… ↓
