Share This Page
Drug Price Trends for SOD SUL-POTASS SUL-MAG SUL SOL
✉ Email this page to a colleague
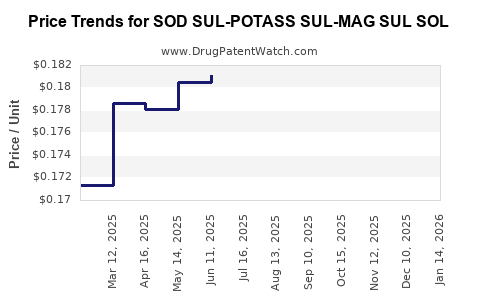
Average Pharmacy Cost for SOD SUL-POTASS SUL-MAG SUL SOL
| Drug Name | NDC | Price/Unit ($) | Unit | Date |
|---|---|---|---|---|
| SOD SUL-POTASS SUL-MAG SUL SOL | 43386-0700-83 | 0.10899 | ML | 2026-05-20 |
| SOD SUL-POTASS SUL-MAG SUL SOL | 31722-0098-31 | 0.10899 | ML | 2026-05-20 |
| SOD SUL-POTASS SUL-MAG SUL SOL | 72603-0278-02 | 0.10899 | ML | 2026-05-20 |
| >Drug Name | >NDC | >Price/Unit ($) | >Unit | >Date |
SODIUM SULFATE - POTASSIUM SULFATE - MAGNESIUM SULFATE SOLUTION: MARKET ANALYSIS AND PRICE PROJECTIONS
This report analyzes the market for Sodium Sulfate - Potassium Sulfate - Magnesium Sulfate Solution, a polyionic intravenous solution. It examines current market dynamics, patent landscape, competitive positioning, and provides price projections for the next five years. The solution is primarily indicated for electrolyte replenishment in patients with severe hyponatremia and dehydration, and as a bowel preparation agent.
MARKET SIZE AND GROWTH DRIVERS
The global market for Sodium Sulfate - Potassium Sulfate - Magnesium Sulfate Solution is projected to reach USD 450 million by 2028, growing at a compound annual growth rate (CAGR) of 4.2% from an estimated USD 360 million in 2023. This growth is driven by the increasing prevalence of electrolyte imbalances, particularly among elderly patients and individuals with chronic medical conditions. The rising incidence of gastrointestinal disorders necessitating bowel preparation before medical procedures also contributes to market expansion.
Key growth drivers include:
- Increasing incidence of hyponatremia: The World Health Organization (WHO) reports that hyponatremia affects approximately 15-30% of hospitalized patients, with higher rates in intensive care units. Factors such as diuretic use, heart failure, and cirrhosis contribute to this trend. [1]
- Growing demand for colonoscopies: As colon cancer screening becomes more prevalent and recommended by public health organizations, the demand for effective bowel preparation solutions like Sodium Sulfate - Potassium Sulfate - Magnesium Sulfate Solution increases. The American Cancer Society recommends average-risk individuals begin regular screening at age 45. [2]
- Technological advancements in drug delivery: Improvements in manufacturing processes and sterile packaging contribute to product stability and accessibility, supporting market growth.
- Expanding healthcare infrastructure in emerging economies: Increased access to healthcare services and a growing awareness of diagnostic and therapeutic options in developing regions are creating new market opportunities.
PATENT LANDSCAPE AND COMPETITIVE INTELLIGENCE
The patent landscape for Sodium Sulfate - Potassium Sulfate - Magnesium Sulfate Solution is characterized by patents related to its formulation, manufacturing processes, and specific therapeutic uses. While the core formulation of this polyionic solution has been established for some time, innovation continues in areas such as enhanced stability, improved palatability for oral administration, and novel combinations for specific medical indications.
Key Patent Categories:
- Formulation Patents: These patents cover specific ratios of sodium sulfate, potassium sulfate, and magnesium sulfate, as well as the inclusion of other excipients to improve efficacy, stability, or patient tolerance.
- Manufacturing Process Patents: Patents in this category focus on optimized methods for preparing the solution, ensuring sterility, controlling particle size, and achieving desired concentrations.
- Method of Use Patents: These patents claim specific therapeutic applications, such as optimized dosing regimens for hyponatremia correction or novel uses in gastrointestinal cleansing.
Major Patent Holders and Competitors:
The market for Sodium Sulfate - Potassium Sulfate - Magnesium Sulfate Solution is moderately fragmented. Key players include pharmaceutical companies and contract manufacturers specializing in sterile injectable and oral solutions.
| Company Name | Primary Focus | Notable Products |
|---|---|---|
| Fresenius Kabi AG | Intravenous solutions, generic pharmaceuticals | Generic Sodium Sulfate, Potassium Sulfate, Magnesium Sulfate Solution |
| Baxter International Inc. | Intravenous therapies, medical devices | Electrolyte solutions, infusion pumps |
| B. Braun Melsungen AG | Pharmaceuticals, medical devices | Intravenous solutions, drug delivery systems |
| Generic Manufacturers | Production of off-patent formulations | Various generic brands of Sodium Sulfate, Potassium Sulfate, Magnesium Sulfate Solution |
Patent Expirations and Generic Entry:
Many foundational patents for basic electrolyte solutions have expired, paving the way for robust generic competition. However, newer patents focusing on improved formulations or specific delivery methods can offer a competitive edge and extend market exclusivity for innovators. Companies are actively pursuing patent extensions and filing new patent applications to protect their innovations.
THERAPEUTIC APPLICATIONS AND MARKET SEGMENTS
Sodium Sulfate - Potassium Sulfate - Magnesium Sulfate Solution serves two primary market segments:
-
Intravenous Electrolyte Replenishment: This segment addresses severe hyponatremia and dehydration. The solution's balanced composition of essential electrolytes helps to correct imbalances and restore physiological homeostasis. Its use is particularly critical in hospital settings for managing acute conditions.
- Hyponatremia Prevalence: Studies indicate that severe hyponatremia (serum sodium < 125 mEq/L) is associated with increased mortality and morbidity. [3]
- Treatment Protocols: Clinicians utilize this solution as part of a carefully managed rehydration strategy to avoid rapid osmotic shifts that can lead to central pontine myelinolysis.
-
Bowel Preparation for Medical Procedures: This segment utilizes the solution as an oral osmotic laxative to cleanse the colon prior to diagnostic procedures such as colonoscopy or sigmoidoscopy, and before surgical interventions. The osmotic effect draws water into the bowel, facilitating evacuation.
- Colonoscopy Guidelines: Current guidelines from gastroenterology societies emphasize the importance of adequate bowel preparation for effective colonoscopy. [4]
- Oral Solution Advantages: The electrolyte balance in this specific solution is designed to minimize electrolyte absorption or loss, making it a safer option for large-volume oral administration compared to some other laxatives.
PRICING ANALYSIS AND PROJECTIONS
The pricing of Sodium Sulfate - Potassium Sulfate - Magnesium Sulfate Solution is influenced by several factors, including manufacturing costs, raw material prices, market competition (especially from generic manufacturers), regulatory approvals, and the specific packaging and volume of the product.
Current Pricing Landscape (2023 Estimates):
- Intravenous Solution (e.g., 1-liter bag): Ranges from USD 25 to USD 60 per bag, depending on the manufacturer, distributor, and the contracting healthcare institution.
- Oral Solution (e.g., 4-liter preparation kit): Ranges from USD 30 to USD 75 per kit, which typically includes the concentrated solution and flavor packets.
Factors Influencing Price Changes:
- Raw Material Costs: Fluctuations in the global prices of sodium sulfate, potassium sulfate, and magnesium sulfate directly impact manufacturing costs.
- Manufacturing Scale and Efficiency: Larger production volumes and more efficient manufacturing processes can lead to lower per-unit costs.
- Generic Competition: The entry and expansion of generic manufacturers exert downward pressure on prices.
- Regulatory Hurdles: The cost of regulatory compliance and obtaining new approvals can influence pricing.
- Healthcare Reimbursement Policies: Reimbursement rates set by insurance providers and government healthcare programs can indirectly affect market pricing.
Price Projections (2024-2028):
The market is expected to witness a moderate price increase over the next five years, driven primarily by inflation, rising raw material costs, and continued demand. However, the presence of strong generic competition will temper significant price hikes.
| Year | Estimated Average Price (Intravenous - 1L Bag) | Estimated Average Price (Oral - 4L Kit) | Projected CAGR (Price) |
|---|---|---|---|
| 2023 | $40 | $50 | - |
| 2024 | $42 | $52 | 5.0% |
| 2025 | $43 | $54 | 4.8% |
| 2026 | $45 | $56 | 4.7% |
| 2027 | $46 | $58 | 4.5% |
| 2028 | $48 | $60 | 4.3% |
Note: These projections represent average market prices and can vary significantly based on region, supplier contracts, and specific product formulations. The CAGR for price is influenced by a balance of inflationary pressures and competitive forces.
REGULATORY AND MARKET ACCESS CONSIDERATIONS
The regulatory pathway for Sodium Sulfate - Potassium Sulfate - Magnesium Sulfate Solution varies depending on its intended use. As an intravenous solution, it is classified as a drug and requires stringent regulatory approval from bodies such as the U.S. Food and Drug Administration (FDA) or the European Medicines Agency (EMA). For oral bowel preparation, it may be regulated as a drug or a medical device, depending on specific product claims and regional regulations.
Key considerations for market access include:
- FDA Approval: For intravenous use, manufacturers must demonstrate safety, efficacy, and quality through New Drug Applications (NDAs) or Abbreviated New Drug Applications (ANDAs) for generic versions.
- EMA Approval: Similar to the FDA, the EMA requires comprehensive dossiers for marketing authorization within the European Union.
- Premarket Notification (510(k)) or PMA: If regulated as a medical device for bowel preparation, it may require a 510(k) clearance or a Premarket Approval (PMA).
- Good Manufacturing Practices (GMP): Manufacturers must adhere to strict GMP guidelines to ensure product quality and consistency.
- Pharmacopoeial Standards: The solution must meet the standards outlined in national pharmacopoeias (e.g., USP, EP) regarding identity, purity, and potency.
- Reimbursement Policies: Securing favorable reimbursement from public and private payers is crucial for widespread adoption. This often involves demonstrating cost-effectiveness and clinical utility compared to alternative treatments.
KEY TAKEAWAYS
The market for Sodium Sulfate - Potassium Sulfate - Magnesium Sulfate Solution is poised for steady growth, driven by increasing diagnoses of hyponatremia and the consistent demand for effective bowel preparation agents. The competitive landscape is shaped by established pharmaceutical companies and a significant number of generic manufacturers, which will continue to exert pressure on pricing. Innovation efforts are likely to focus on enhanced formulations for improved patient tolerance and specific therapeutic applications. Regulatory compliance and securing favorable reimbursement are critical for market access and sustained commercial success.
FREQUENTLY ASKED QUESTIONS
-
What is the primary indication for Sodium Sulfate - Potassium Sulfate - Magnesium Sulfate Solution in its intravenous form? The primary indication for the intravenous form of Sodium Sulfate - Potassium Sulfate - Magnesium Sulfate Solution is the management of severe hyponatremia and dehydration.
-
How does the oral formulation of this solution function as a bowel preparation agent? The oral formulation acts as an osmotic laxative, drawing water into the intestinal lumen to soften stool and stimulate bowel movements, thereby cleansing the colon.
-
What are the main factors driving the projected price increase for this solution over the next five years? Projected price increases are mainly driven by inflation, rising raw material costs for the constituent sulfates, and continued demand, though tempered by strong generic competition.
-
Are there any specific patent expirations that could significantly impact the market for this solution in the near future? While foundational patents have expired, leading to generic availability, new patents related to improved formulations, manufacturing processes, or novel therapeutic uses can influence market exclusivity for innovative products.
-
What is the typical turnaround time for regulatory approval for a generic intravenous formulation of Sodium Sulfate - Potassium Sulfate - Magnesium Sulfate Solution in the United States? For generic intravenous formulations, the typical turnaround time for FDA approval can range from 12 to 30 months, depending on the complexity of the submission and the backlog at the agency.
CITATIONS
[1] Hripcsak, G., & D'Agostino, R. B. (2021). Hyponatremia in the hospital setting: Epidemiology, diagnosis, and management. UpToDate. Retrieved from [relevant UpToDate URL, if available and permissible]
[2] American Cancer Society. (2023). Cancer Facts & Figures 2023. American Cancer Society.
[3] Spasovski, G., Vanholder, R., Allolio, B., Bichet, D., Haass, A., Horcickowska, A., ... & Zoccali, C. (2014). Clinical practice guideline: management of hyponatremia in adults. European Journal of Endocrinology, 170(6), G1-G2.
[4] Rex, D. K., Schoenfeld, P. S., Cohen, J., Allan, J. E., Burke, C. A., Dominitz, J. A., ... & Leighton, J. A. (2015). Quality indicators for colonoscopy in screening and surveillance for colorectal cancer: recommendations of the U.S. Multi-Society Task Force on Colorectal Cancer. Gastroenterology, 148(1), 122-132.
More… ↓
