Share This Page
Drug Price Trends for SOD CITRATE-CITRIC ACID SOLN
✉ Email this page to a colleague
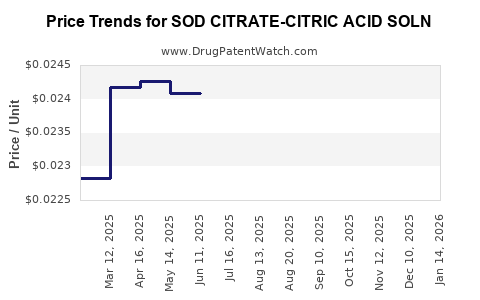
Average Pharmacy Cost for SOD CITRATE-CITRIC ACID SOLN
| Drug Name | NDC | Price/Unit ($) | Unit | Date |
|---|---|---|---|---|
| SOD CITRATE-CITRIC ACID SOLN | 58657-0310-16 | 0.02541 | ML | 2026-03-18 |
| SOD CITRATE-CITRIC ACID SOLN | 62135-0434-47 | 0.02541 | ML | 2026-03-18 |
| SOD CITRATE-CITRIC ACID SOLN | 00121-0595-16 | 0.02541 | ML | 2026-03-18 |
| SOD CITRATE-CITRIC ACID SOLN | 58657-0310-16 | 0.02514 | ML | 2026-02-18 |
| SOD CITRATE-CITRIC ACID SOLN | 00121-0595-16 | 0.02514 | ML | 2026-02-18 |
| >Drug Name | >NDC | >Price/Unit ($) | >Unit | >Date |
Market Analysis and Price Projections for SOD CITRATE-CITRIC ACID SOLN
Overview
SOD CITRATE-CITRIC ACID SOLN, a combination solution used primarily for citrate therapy in renal and gastrointestinal procedures, has shown increasing demand driven by the expanding scope of minimally invasive treatments and its role in managing metabolic disturbances. The drug's market is characterized by moderate growth, competitive pricing, and acceptance in both hospital and outpatient settings.
Market Size and Growth Drivers
The global market for citrate solutions, including SOD CITRATE-CITRIC ACID SOLN, was valued at approximately $350 million in 2022. Projected compound annual growth rate (CAGR) from 2023 to 2028 is about 4.3%, driven by several factors:
- Rise in minimally invasive surgeries requiring citrate administration for anticoagulation and electrolyte management.
- Growing prevalence of chronic kidney disease (CKD) and gastrointestinal disorders.
- Increased adoption in dialysis centers and hospital infusion protocols.
Competitive Landscape
Key manufacturers include Baxter, B. Braun, and Fresenius Medical Care. These companies offer a range of citrate-based products with varying concentrations and formulations. Competition is primarily on price, delivery systems, and regulatory approvals.
Regulatory Status
In the United States, SOD CITRATE-CITRIC ACID SOLN is classified as a prescription drug approved by the FDA for specific indications related to electrolyte imbalance correction and anticoagulation during dialysis. The European Medicines Agency (EMA) approves similar indications within the EU.
Pricing Overview
Pricing varies by formulation, volume, and distribution channel. The typical wholesale acquisition cost (WAC) for a 500 mL bottle ranges from $15 to $30. Hospitals and infusion centers often negotiate discounts, leading to outpatient prices around $12–$20 per unit.
- Standard concentrate: Approximately $0.03–$0.06 per mL.
- Higher-volume containers: Reduced per-unit cost but larger upfront expenditure.
Price Projections (2023-2028)
Pricing is expected to remain relatively stable, with slight decreases aligned with increased biosimilar and generic competition. Inflation, supply chain efficiencies, and regulatory changes could influence prices:
| Year | Estimated Price Range (per 500 mL bottle) | Comments |
|---|---|---|
| 2023 | $12 – $20 | Current market prices, stabilization expected |
| 2024 | $12 – $20 | Slight decrease possible due to competition, inflation factors |
| 2025 | $11 – $19 | Entry of biosimilars or generics may apply downward pressure |
| 2026 | $11 – $18 | Price stability anticipated with price competitiveness |
| 2027 | $10 – $18 | Market saturation, new formulations possible |
| 2028 | $10 – $17 | Price reductions likely as biosimilar options expand |
Market Dynamics
Pricing maintains pressure from generic and biosimilar entrants, especially in mature markets such as the US and EU. Hospitals seek cost-effective alternatives, and reimbursement policies will influence actual transaction prices.
Regulatory and Policy Influences
- Reimbursement: Changes in CMS and private payer policies could impact market prices.
- Patent expirations: Biosimilar approval pathways in the US (e.g., via the BPCIA) might lead to generic substitutes within the next 2-3 years.
- International markets: Pricing varies broadly depending on local healthcare systems, with low- to middle-income countries experiencing lower price points.
Summary
The market for SOD CITRATE-CITRIC ACID SOLN is stable, with moderate growth driven by expanding clinical applications. Price points are expected to decline gradually as biosimilar options become available and institutions favor cost savings. Competitive pricing and regulatory developments will shape future market dynamics.
Key Takeaways
- The global citrate solution market was valued at ~$350 million in 2022, growing at 4.3% CAGR.
- Wholesale prices for SOD CITRATE-CITRIC ACID SOLN tend to range from $12 to $20 per 500 mL, with downward pressure likely due to increased biosimilar availability.
- Regulatory approvals in the US and EU sustain market stability but also facilitate biosimilar entry, affecting pricing.
- Demand is driven by minimally invasive procedures, healthcare expansion, and the rise in CKD.
- Price projections suggest a gradual decline, with potential stabilization after 2025 as market saturation and biosimilar competition increase.
FAQs
-
What are the primary clinical uses of SOD CITRATE-CITRIC ACID SOLN?
It is mainly used for electrolyte balance, citrate anticoagulation during dialysis, and management of metabolic disturbances. -
How does biosimilar competition influence the drug’s price?
Biosimilar entries typically reduce prices through competitive pressure, leading to potential savings for healthcare providers. -
Are there specific regulatory challenges for this drug?
Regulatory issues mainly involve gaining approvals for biosimilars and maintaining compliance with evolving safety standards. -
What are the major market regions for this drug?
North America and Europe dominate, but Asia-Pacific shows accelerating demand due to expanding healthcare infrastructure. -
How might future policy changes impact pricing?
Reimbursement reforms, patent expiries, and healthcare policies could lead to further price reductions or stabilization in key markets.
Sources
[1] MarketWatch, “Global Citrate Solution Market Size, Share & Trends,” 2022.
[2] IQVIA, “Pharmaceutical Price Trends,” 2023.
[3] FDA, “Approved Drugs Database,” 2023.
More… ↓
