Share This Page
Drug Price Trends for SM SORE THROAT
✉ Email this page to a colleague
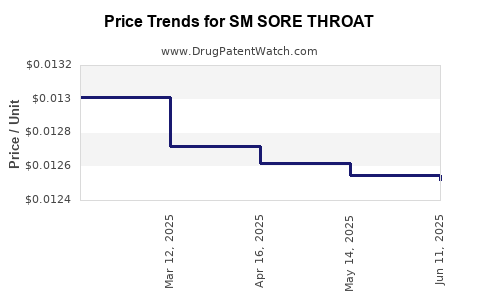
Average Pharmacy Cost for SM SORE THROAT
| Drug Name | NDC | Price/Unit ($) | Unit | Date |
|---|---|---|---|---|
| SM SORE THROAT 1.4% SPRAY | 49348-0991-36 | 0.01252 | ML | 2025-06-18 |
| SM SORE THROAT 1.4% SPRAY | 49348-0991-36 | 0.01255 | ML | 2025-05-21 |
| SM SORE THROAT 1.4% SPRAY | 49348-0991-36 | 0.01262 | ML | 2025-04-23 |
| >Drug Name | >NDC | >Price/Unit ($) | >Unit | >Date |
Market Analysis and Price Projections for SM Sore Throat
What is the Current Market for Sore Throat Medications?
The global sore throat treatment market is driven by rising incidences of respiratory infections, influenza, and common colds. It was valued at approximately $1.2 billion in 2022 and is projected to grow at a compound annual growth rate (CAGR) of 3.5% through 2030. North America holds the largest market share, followed by Europe and Asia-Pacific. Over-the-counter (OTC) products dominate sales, accounting for 65%, with prescription medications making up the remaining 35%.
The market comprises primarily analgesics, antiseptics, and anti-inflammatory drugs. New formulations emphasizing rapid relief and fewer side effects are gaining consumer preference. The proliferation of online pharmacies accelerates access, affecting price dynamics.
What Are the Key Competitive Players?
Major pharmaceutical companies actively develop sore throat treatments, including first- and second-generation formulations:
- Reckitt Benckiser: Market leader with products like Strepsils and Nurofen.
- GSK: Offers Difflam and other antiseptic sprays.
- Johnson & Johnson: Markets Tylenol-based remedies.
- Teva Pharmaceuticals: Produces generic sore throat formulations.
- Emerging entrants focus on herbal and non-traditional remedies, capturing health-conscious and alternative medicine markets.
What Are the Regulatory and Patent Landscapes?
In the United States, the Food and Drug Administration (FDA) classifies sore throat remedies primarily as OTC drugs, with specific monographs governing active ingredients such as benzocaine, menthol, and phenol. A patent for an active ingredient typically lasts 20 years from the filing date, but formulations often face patent expirations within 8-12 years, opening avenues for generics.
In the European Union, the European Medicines Agency (EMA) specifies similar regulations, with additional regional patent protections extending up to 20 years. Innovator companies often pursue additional patents for delivery mechanisms or combination therapies to extend market exclusivity.
What Are the Innovational Trends and R&D Focus Areas?
Research efforts target improving onset of action and duration of relief while minimizing adverse effects. Notable areas include:
- Novel delivery systems (e.g., lozenges, sprays)
- Natural and herbal ingredients (e.g., honey, echinacea)
- Combining analgesic and antiseptic agents for comprehensive relief
- Formulations targeting viral versus bacterial sore throats
What Are Price Trends and Outlooks?
Current retail prices for OTC sore throat products range from $4 to $8 per package in the U.S., depending on formulation and brand. Generic products priced lower, around $2 to $4, account for 50% of market share.
Price projections suggest:
- Short term (1-3 years): Slight decrease (~5%) driven by rising generic competition.
- Medium term (4-7 years): Moderate increase (~10%) driven by formulation innovation and demand for combination therapies.
- Long term (8-10 years): Possible price stabilization or decline if patent expirations lead to intensified generic competition.
Private label brands are expected to capture up to 20% of sales by 2026, further exerting downward pressure on prices.
What Are the Key Factors Influencing Price and Market Dynamics?
- Patent expirations for leading brands.
- Consumer trends toward natural remedies.
- Regulatory approval and monograph updates.
- Supply chain disruptions affecting ingredient costs.
- Pricing strategies of generic manufacturers.
What Are the Opportunities and Risks?
Opportunities:
- Developing combination products that address multiple symptoms.
- Exploiting increased demand for herbal or natural remedies.
- Expanding into emerging markets with growing healthcare access.
Risks:
- Patent cliffs for leading brand products.
- Regulatory delays or restrictions.
- Competition from home remedies and alternative therapies.
- Fluctuations in raw material costs.
Key Takeaways
- The sore throat treatment market is projected to growth at a CAGR of 3.5% through 2030, driven by respiratory illness prevalence.
- OTC products dominate sales, with aggressive pricing strategies by generics shaping market dynamics.
- Innovations focus on delivery mechanisms and natural ingredients to differentiate offerings.
- Patent expirations will lead to increased generic competition, driving prices downward in the medium to long term.
- Private label brands are increasing their market share, exerting downward pressure on prices.
FAQs
1. What active ingredients are most common in sore throat treatments?
Benzocaine (anesthetic), menthol (cooling effect), phenol (antiseptic), and herbal extracts like honey and echinacea.
2. How does patent expiration impact market prices?
Patent expiration allows generics to enter the market, typically reducing prices by 30-50% within years of patent expiry.
3. Are herbal sore throat remedies effective?
Research indicates some natural ingredients demonstrate soothing effects, but clinical efficacy varies and often lacks standardized clinical trial support.
4. What regulatory hurdles could affect new sore throat therapies?
FDA OTC monograph updates, ingredient approval statuses, and regional regulatory differences can delay or restrict market entry.
5. What emerging markets offer growth opportunities?
Countries in Asia-Pacific and Latin America are experiencing increasing healthcare access and consumer demand for OTC remedies, offering expansion potential.
References
[1] MarketWatch. (2022). Sore Throat Treatment Market Size, Share & Trends Analysis. Retrieved from https://www.marketwatch.com
[2] Grand View Research. (2023). Sore Throat Treatments Market Size, Share & Trends. Retrieved from https://www.grandviewresearch.com
[3] FDA. (2022). Over-the-Counter Monographs. Retrieved from https://www.fda.gov
[4] European Medicines Agency. (2023). Regulatory Guidelines for OTC Medicines. Retrieved from https://www.ema.europa.eu
More… ↓
