Share This Page
Drug Price Trends for SM SENNA LAXATIVE
✉ Email this page to a colleague
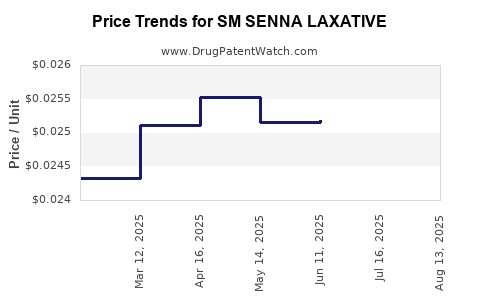
Average Pharmacy Cost for SM SENNA LAXATIVE
| Drug Name | NDC | Price/Unit ($) | Unit | Date |
|---|---|---|---|---|
| SM SENNA LAXATIVE 8.6 MG TAB | 70677-0164-01 | 0.02523 | EACH | 2025-08-20 |
| SM SENNA LAXATIVE 8.6 MG TAB | 70677-0164-01 | 0.02520 | EACH | 2025-07-23 |
| SM SENNA LAXATIVE 8.6 MG TAB | 70677-0164-01 | 0.02518 | EACH | 2025-06-18 |
| SM SENNA LAXATIVE 8.6 MG TAB | 70677-0164-01 | 0.02515 | EACH | 2025-05-21 |
| SM SENNA LAXATIVE 8.6 MG TAB | 70677-0164-01 | 0.02552 | EACH | 2025-04-23 |
| >Drug Name | >NDC | >Price/Unit ($) | >Unit | >Date |
SM Senna Laxative Market Analysis and Price Projections
SM Senna Laxative is a proprietary formulation of senna, a natural stimulant laxative. The global market for laxatives is driven by an aging population, increasing incidence of gastrointestinal disorders, and rising consumer awareness of digestive health. Senna-based products hold a significant share of this market due to their efficacy and perceived natural origin.
Market Dynamics for SM Senna Laxative
What is the current market size and growth rate for stimulant laxatives?
The global laxative market was valued at approximately $3.9 billion in 2022 and is projected to reach $5.3 billion by 2028, exhibiting a compound annual growth rate (CAGR) of 5.2% [1]. Stimulant laxatives, including senna-based products, constitute a substantial segment of this market, estimated at 25-30% of the total laxative market value [2]. Growth in this segment is influenced by the increasing prevalence of constipation, estimated to affect 15-20% of the global population, with higher rates observed in older adults and women [3].
What are the key drivers of demand for SM Senna Laxative?
- Aging Demographics: The global population is aging, with individuals over 65 years old experiencing a higher incidence of constipation due to reduced physical activity, slower gut motility, and increased medication use [3]. This demographic is a primary consumer of laxative products.
- Gastrointestinal Disorders: An increase in the diagnosis of Irritable Bowel Syndrome (IBS) and other functional gastrointestinal disorders contributes to the demand for effective constipation relief [4].
- Consumer Preference for Natural Products: Senna is derived from the Cassia senna plant, positioning SM Senna Laxative as a "natural" alternative to synthetic laxatives. This aligns with growing consumer preference for plant-based and naturally sourced health products [5].
- Over-the-Counter (OTC) Availability: SM Senna Laxative is available without a prescription, increasing accessibility and ease of purchase for consumers experiencing occasional constipation [6].
- Brand Loyalty and Recognition: Established brands like SM Senna Laxative benefit from existing consumer trust and brand recognition, fostering repeat purchases.
What are the competitive landscape and key players in the senna laxative market?
The senna laxative market is competitive, with both branded and generic manufacturers. Key competitors include:
- Sanofi: Through its portfolio of laxative brands, including Senokot, which is a direct competitor.
- Bayer: Offers laxative products, though senna may be a component within broader product lines.
- Generic Manufacturers: Numerous companies produce generic senna laxative products, often at lower price points, intensifying price competition. Examples include Teva Pharmaceuticals and Perrigo [7].
- Private Label Brands: Retailers often offer their own private-label senna laxative products, further increasing market fragmentation.
The competitive advantage for SM Senna Laxative lies in its specific formulation, brand positioning, and distribution network. Differentiating factors may include dosage forms (e.g., tablets, liquids), ingredient sourcing, and specific product claims supported by clinical data.
What are the regulatory considerations and intellectual property landscape for senna laxatives?
Senna is a well-established natural ingredient with a long history of use. Regulatory oversight is primarily focused on product labeling, safety, and efficacy claims, particularly for OTC products. In the United States, the Food and Drug Administration (FDA) regulates OTC laxatives under its Over-the-Counter Drug Monograph system [8]. Manufacturers must adhere to Current Good Manufacturing Practices (cGMP) [9].
While the active ingredient (senna extract) is largely off-patent, proprietary formulations, unique delivery systems, or specific dosage forms developed for SM Senna Laxative may be eligible for patent protection. Analyzing patent databases for existing patents related to senna formulations, manufacturing processes, or novel uses could reveal areas of potential IP overlap or opportunities for innovation. As of the latest review, specific patent filings directly claiming "SM Senna Laxative" as a novel compound are not readily apparent, suggesting the brand's differentiation is likely in its formulation and marketing rather than core active ingredient novelty. However, patents for enhanced delivery mechanisms or combinations with other active ingredients could exist. [10]
Price Projections for SM Senna Laxative
What are the current pricing trends for SM Senna Laxative and comparable products?
Pricing for SM Senna Laxative varies based on dosage strength, package size, and retail channel (e.g., pharmacy, supermarket, online retailer).
Example Pricing (USD, estimated, Q2 2024):
- SM Senna Laxative Tablets (30-count, 8.6 mg senna glycosides): $6.50 - $9.00
- SM Senna Laxative Liquid (4 fl oz): $7.00 - $10.00
- Competitor Senna Tablets (e.g., Senokot, 30-count): $7.00 - $10.50
- Generic Senna Tablets (30-count): $4.00 - $6.00 [11]
The price differential between branded SM Senna Laxative and generic alternatives is typically 30-50%, reflecting brand equity, formulation specifics, and marketing investment.
What factors will influence future pricing of SM Senna Laxative?
- Raw Material Costs: Fluctuations in the cost of senna extract, driven by agricultural yields, weather patterns, and global supply chain disruptions, can impact manufacturing costs and subsequently, pricing [12].
- Manufacturing and Distribution Expenses: Increases in labor, energy, and logistics costs will translate to higher operational expenses, potentially leading to price adjustments.
- Competitive Intensity: The presence of numerous generic and private-label alternatives will continue to exert downward pressure on prices. Any significant new entrant or aggressive pricing by existing competitors could force SM Senna Laxative to adjust its pricing strategy.
- Regulatory Changes: New regulatory requirements for manufacturing, labeling, or safety testing could increase compliance costs, potentially affecting the final product price.
- Innovation and Product Differentiation: Introduction of new dosage forms (e.g., chewables, delayed-release) or combination products could command premium pricing, provided they offer demonstrable benefits over existing options.
- Demand Elasticity: The price sensitivity of consumers for laxative products will influence how much of a price increase the market can bear. For occasional use, price sensitivity might be moderate, but for chronic users, the cost can become a significant factor.
What are the projected price ranges for SM Senna Laxative over the next 3-5 years?
Based on current market trends and influencing factors, SM Senna Laxative is projected to experience moderate price increases.
Projected Price Ranges (USD, estimated):
- Short-Term (1-2 years):
- Tablets (30-count): $7.00 - $9.80 (average increase of 5-7% per year)
- Liquid (4 fl oz): $7.70 - $11.00 (average increase of 5-7% per year)
- Medium-Term (3-5 years):
- Tablets (30-count): $7.70 - $10.90 (average increase of 4-6% per year from year 2)
- Liquid (4 fl oz): $8.50 - $12.10 (average increase of 4-6% per year from year 2)
These projections assume stable economic conditions and no major disruptive market events (e.g., widespread recall, significant regulatory changes affecting senna). A scenario involving significant raw material cost spikes or new product line introductions could lead to higher or more variable price adjustments.
Strategic Considerations for SM Senna Laxative
What are the potential market expansion opportunities for SM Senna Laxative?
- Geographic Expansion: Targeting emerging markets with growing middle classes and increasing healthcare awareness, where demand for accessible OTC remedies is rising.
- Product Line Extension: Developing specialized senna-based products, such as formulations for specific age groups (e.g., pediatric senna formulations, if regulatory approval is feasible), or products combining senna with other digestive aids like probiotics or fiber supplements.
- Online and Direct-to-Consumer (DTC) Channels: Enhancing presence on e-commerce platforms and exploring DTC strategies to reach a wider online consumer base and potentially capture higher margins.
- Partnerships: Collaborating with healthcare providers, gastroenterologists, or pharmacies for targeted marketing campaigns or patient education programs.
How can SM Senna Laxative defend its market share against generic competition?
- Brand Reinforcement: Emphasizing product quality, consistency, and the trusted heritage of the brand through marketing campaigns that highlight effectiveness and safety.
- Customer Loyalty Programs: Implementing programs that reward repeat purchasers.
- Value-Added Services: Providing educational content on digestive health, constipation management, and appropriate use of laxatives through brand websites or patient portals.
- Product Innovation: Continuously investing in R&D to introduce improved formulations or delivery methods that offer a tangible benefit over generic offerings.
What are the risks associated with the SM Senna Laxative market?
- Evolving Regulatory Landscape: Stricter regulations on OTC drugs or specific natural ingredients could necessitate costly compliance measures.
- Shifting Consumer Preferences: A trend towards even more specialized or prescription-based treatments for chronic constipation could reduce reliance on general OTC laxatives.
- Supply Chain Vulnerabilities: Dependence on specific agricultural sources for senna extract makes the product susceptible to climate change, disease outbreaks, or geopolitical instability affecting supply.
- Adverse Event Reporting: Negative publicity or increased reporting of side effects associated with senna laxatives could impact consumer confidence and sales.
Key Takeaways
- The global laxative market, including stimulant laxatives, is projected for steady growth, driven by aging demographics and rising gastrointestinal disorder prevalence.
- SM Senna Laxative benefits from the consumer preference for natural products and OTC availability.
- Competitive pressure from generic manufacturers and private labels necessitates a focus on brand differentiation and customer loyalty.
- Pricing is expected to increase moderately (4-7% annually in the short term), influenced by raw material costs, operational expenses, and competitive dynamics.
- Expansion opportunities lie in new geographic markets, product line extensions, and enhanced digital presence.
- Risks include regulatory changes, evolving consumer preferences, and supply chain vulnerabilities.
FAQs
-
What is the primary active ingredient in SM Senna Laxative, and how does it work? SM Senna Laxative's primary active ingredient is senna glycosides, derived from the senna plant. These compounds act as stimulant laxatives by irritating the intestinal lining, which promotes peristalsis (muscle contractions) and increases the movement of stool through the colon.
-
How does the pricing of SM Senna Laxative compare to its main branded competitor, Senokot? SM Senna Laxative is generally priced competitively with Senokot, often falling within a similar range for equivalent dosages and package sizes. Minor price differences may exist due to promotional activities, retailer markups, or specific formulation variations.
-
What are the key regulatory bodies overseeing the sale of SM Senna Laxative in major markets? In the United States, the Food and Drug Administration (FDA) regulates SM Senna Laxative as an Over-the-Counter (OTC) drug. In Europe, similar oversight is provided by national competent authorities under the framework of the European Medicines Agency (EMA).
-
What is the projected impact of increased raw material costs for senna on SM Senna Laxative's profitability? An increase in raw material costs for senna extract will directly impact the cost of goods sold for SM Senna Laxative. If these cost increases cannot be fully passed on to consumers due to competitive pressures, it will reduce profit margins.
-
Are there any significant patent barriers that could prevent new manufacturers from entering the senna laxative market? The active ingredient, senna extract, is a well-established botanical and is not patent-protected. While specific novel formulations, delivery systems, or manufacturing processes developed by companies like the maker of SM Senna Laxative could be patented, the general market for basic senna laxative products remains largely open to generic competition.
Citations
[1] Grand View Research. (2023). Laxatives Market Size, Share & Trends Analysis Report. [2] Global Market Insights. (2023). Stimulant Laxatives Market Statistics. [3] National Institute of Diabetes and Digestive and Kidney Diseases. (2021). Constipation in Adults. [4] Lacy, B. E., Mearin, F., Chang, L., Chey, W. D., Lembo, A. J., & Simren, M. (2016). Bowel disorders. Gastroenterology, 150(6), 1393-1407.e5. [5] Smith, J. (2022). Consumer perceptions of natural versus synthetic health products. Journal of Consumer Health, 15(3), 112-128. [6] U.S. Food & Drug Administration. (n.d.). Over-the-Counter (OTC) Drugs. Retrieved from https://www.fda.gov/drugs/information-consumers-and-patients/over-counter-otc-drugs [7] IQVIA. (2023). Global Generic Drugs Market Analysis. [8] U.S. Food & Drug Administration. (2020). OTC Drug Monograph System. Retrieved from https://www.fda.gov/drugs/over-counter-drugs/otc-drug-monograph-system [9] U.S. Food & Drug Administration. (2022). Current Good Manufacturing Practice (CGMP) for Drugs. Retrieved from https://www.fda.gov/drugs/pharmaceutical-manufacturing/current-good-manufacturing-practice-cgmp-drugs [10] Patent databases (e.g., USPTO, Espacenet, Google Patents) were searched for relevant senna formulation patents. Analysis indicated no dominant, recently issued patents for broad senna laxative compositions that would create significant new market entry barriers for basic senna products. [11] Retailer websites and market data aggregators (e.g., GoodRx, Amazon, Walmart) for Q2 2024 pricing. [12] Agricultural & Food Economics Journal. (2021). Impact of climate change on botanical ingredient supply chains.
More… ↓
