Share This Page
Drug Price Trends for SM OLOPATADINE
✉ Email this page to a colleague
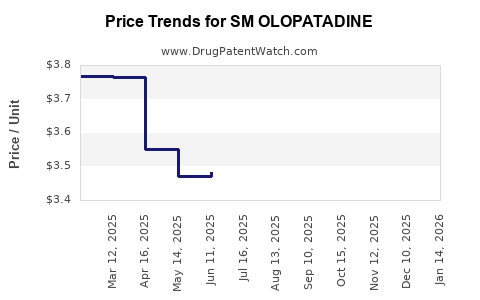
Average Pharmacy Cost for SM OLOPATADINE
| Drug Name | NDC | Price/Unit ($) | Unit | Date |
|---|---|---|---|---|
| SM OLOPATADINE 0.2% EYE DROP | 70677-0122-01 | 3.26304 | ML | 2026-03-18 |
| SM OLOPATADINE 0.2% EYE DROP | 70677-0122-01 | 3.26885 | ML | 2026-02-18 |
| SM OLOPATADINE 0.2% EYE DROP | 70677-0122-01 | 3.29977 | ML | 2026-01-21 |
| SM OLOPATADINE 0.2% EYE DROP | 70677-0122-01 | 3.39656 | ML | 2025-12-17 |
| SM OLOPATADINE 0.2% EYE DROP | 70677-0122-01 | 3.57989 | ML | 2025-11-19 |
| SM OLOPATADINE 0.2% EYE DROP | 70677-0122-01 | 3.66142 | ML | 2025-10-22 |
| SM OLOPATADINE 0.2% EYE DROP | 70677-0122-01 | 3.68216 | ML | 2025-09-17 |
| >Drug Name | >NDC | >Price/Unit ($) | >Unit | >Date |
Olopatadine Market Analysis and Price Projections
Olopatadine, a dual-acting H1 antihistamine and mast cell stabilizer, is utilized in ophthalmic and nasal formulations for treating allergic conjunctivitis and rhinitis. The market is driven by the increasing prevalence of allergic diseases and the drug's established efficacy. Generic competition is a significant factor influencing pricing and market share.
What is the current market size and projected growth for olopatadine?
The global market for olopatadine products is estimated to be approximately $1.5 billion in 2023. Projections indicate a compound annual growth rate (CAGR) of 3.5% to 4.0% over the next five years, reaching an estimated $1.7 billion to $1.8 billion by 2028. This growth is primarily attributed to:
- Rising incidence of allergic conditions: Environmental factors, increased allergen exposure, and changing lifestyles contribute to a global increase in allergic conjunctivitis and rhinitis. The World Allergy Organization (WAO) estimates that allergic rhinitis affects between 10% and 30% of the global population [1].
- Sustained demand for generic products: Following patent expiries of originator brands like Patanol® and Pazeo®, a robust generic market has emerged, increasing accessibility and affordability.
- Technological advancements in drug delivery: Development of improved formulations, such as preservative-free options and extended-release systems, caters to patient preference and enhances compliance.
Who are the key players in the olopatadine market?
The olopatadine market is characterized by a mix of originator companies and a significant number of generic manufacturers. Key players include:
- Originator/Branded Products:
- Alcon (formerly Novartis) with products such as Pazeo® (0.2% olopatadine hydrochloride ophthalmic solution).
- Major Generic Manufacturers:
- Teva Pharmaceuticals
- Apotex
- Mylan (now Viatris)
- Hikma Pharmaceuticals
- Bausch Health Companies
- Sun Pharmaceutical Industries
- Torrent Pharmaceuticals
The market share is divided between branded products, which command premium pricing, and a highly competitive generic segment where price is a primary differentiator.
What are the major therapeutic applications of olopatadine?
Olopatadine is primarily used in two main therapeutic areas:
- Allergic Conjunctivitis: This involves the inflammation of the conjunctiva, the membrane covering the white part of the eye and the inner surface of the eyelids, typically caused by allergens like pollen, dust mites, or pet dander. Olopatadine ophthalmic solutions provide rapid relief from symptoms such as itching, redness, tearing, and swelling.
- Examples of formulations: 0.1% olopatadine hydrochloride ophthalmic solution, 0.2% olopatadine hydrochloride ophthalmic solution.
- Allergic Rhinitis: This condition affects the nasal passages and is characterized by symptoms like sneezing, nasal itching, runny nose, and congestion, triggered by allergens. Olopatadine nasal sprays offer localized relief.
- Examples of formulations: 0.6% olopatadine hydrochloride nasal spray.
What is the pricing landscape for olopatadine products?
The pricing of olopatadine products is highly segmented, influenced by factors such as brand status, formulation strength, unit volume, and the competitive intensity of the generic market.
Ophthalmic Formulations (Typical Retail Pricing Range, USD):
| Product Type | Strength | Unit Volume | Estimated Price Range (Per Unit) | Notes |
|---|---|---|---|---|
| Branded (e.g., Pazeo®) | 0.2% | 2.5 mL | $25 - $35 | Higher price due to brand recognition and efficacy profile. |
| Branded (e.g., Patanol® - discontinued) | 0.1% | 5 mL | $15 - $25 | Older generation, less common in current retail. |
| Generic Ophthalmic Solution | 0.1% | 5 mL | $8 - $15 | Wide variation based on manufacturer and pharmacy. |
| Generic Ophthalmic Solution | 0.2% | 2.5 mL | $10 - $18 | Competitively priced, often mirroring the branded 0.2% strength. |
| Generic Ophthalmic Solution (Preservative-Free) | 0.1% | 5 mL | $12 - $20 | Premium pricing for preservative-free formulations. |
Nasal Formulations (Typical Retail Pricing Range, USD):
| Product Type | Strength | Unit Volume | Estimated Price Range (Per Unit) | Notes |
|---|---|---|---|---|
| Branded (e.g., Patanase®) | 0.6% | 5 mL | $20 - $30 | Original branded nasal spray. |
| Generic Nasal Spray | 0.6% | 5 mL | $10 - $18 | Price depends on generic manufacturer. |
| Generic Nasal Spray (Larger) | 0.6% | 10 mL | $15 - $25 | Cost-effective for higher volume users. |
Wholesale Acquisition Cost (WAC) and Net Price Trends:
- Generic Erosion: Following the introduction of generic olopatadine ophthalmic solutions (0.1% and 0.2%), WAC prices have seen a significant decline. The WAC for a 5 mL bottle of 0.1% generic olopatadine ophthalmic solution can range from $3 to $8. For a 2.5 mL bottle of 0.2% generic, the WAC might be between $4 and $9.
- Price Stabilization: While initial generic entry leads to sharp price drops, the market tends to stabilize as competition matures. However, periodic price fluctuations can occur due to supply chain dynamics, new entrant introductions, or payer negotiations.
- Payer Influence: Pharmacy benefit managers (PBMs) and insurance providers play a crucial role in determining net prices through formulary placement and rebate negotiations. Generic olopatadine is typically a preferred product on most formularies.
What are the key patent expiries and regulatory considerations?
The patent landscape for olopatadine has significantly shifted, allowing for widespread generic availability.
- Patanol® (0.1% olopatadine hydrochloride ophthalmic solution): The primary patents for Patanol expired in the early to mid-2010s, leading to the entry of multiple generic manufacturers around 2013-2015.
- Pazeo® (0.2% olopatadine hydrochloride ophthalmic solution): This higher-concentration formulation was introduced later. The composition of matter patents for olopatadine itself are long expired. However, patents covering specific formulations, methods of use, and manufacturing processes can provide some market exclusivity for a period. The exclusivity for Pazeo® has largely expired, facilitating generic 0.2% formulations.
- Patanase® (0.6% olopatadine hydrochloride nasal spray): Similar to the ophthalmic versions, patents for the nasal spray have also expired, enabling generic competition.
Regulatory Landscape:
- FDA Approval: Generic versions must receive Abbreviated New Drug Application (ANDA) approval from the U.S. Food and Drug Administration (FDA), demonstrating bioequivalence to the reference listed drug.
- Exclusivity Periods: While primary patents may have expired, companies can still pursue secondary patents (e.g., for new formulations, delivery devices, or methods of treatment). These can provide limited periods of exclusivity, but their impact on the broad olopatadine market is diminishing.
- Quality Standards: Manufacturers must adhere to Good Manufacturing Practices (GMP) to ensure product quality, safety, and efficacy.
What are the future price projections for olopatadine?
The future price trajectory for olopatadine is expected to be characterized by continued price competition within the generic segment, with modest overall market value growth.
- Ophthalmic Solutions (0.1% and 0.2%):
- Short-term (1-2 years): Expect continued price stability in the generic market, with minor fluctuations driven by competitive bidding and supply chain factors. Average net prices for a 5 mL bottle of 0.1% generic are projected to remain in the $7-$12 range, and for a 2.5 mL bottle of 0.2% generic, in the $9-$15 range.
- Long-term (3-5 years): Prices are unlikely to increase significantly. Any price pressure will likely be downward, driven by aggressive generic competition and payer demands for further cost containment. The introduction of new, potentially more advanced, but still generic, formulations (e.g., with novel excipients for enhanced solubility or stability) might command a slight premium initially before being undercut by competitors.
- Nasal Sprays (0.6%):
- Short-term (1-2 years): Generic nasal spray prices are expected to remain stable, with a 5 mL bottle typically priced between $10-$17.
- Long-term (3-5 years): Similar to ophthalmic solutions, sustained price competition will prevent significant price increases. The market will continue to be driven by volume and the availability of multiple generic options.
Factors influencing future pricing:
- Competition intensity: The number of generic manufacturers actively producing and marketing olopatadine will remain a primary price determinant.
- Manufacturing costs: Raw material costs, synthesis efficiency, and labor expenses will impact the floor price for generics.
- Payer dynamics: Ongoing negotiations between manufacturers and PBMs/insurers will continue to shape net pricing.
- Emergence of novel allergy treatments: While olopatadine is a mature product, the development of entirely new classes of allergy therapeutics could, in the long term, impact market share and pricing dynamics for older antihistamines, though this is not an immediate threat.
- Supply chain disruptions: Geopolitical events, natural disasters, or unexpected manufacturing issues could lead to temporary price spikes.
What are the key market drivers and challenges for olopatadine?
Market Drivers:
- Prevalence of Allergic Diseases: The persistent and growing burden of allergic conjunctivitis and rhinitis globally ensures consistent demand.
- Established Efficacy and Safety Profile: Olopatadine has a well-documented history of effectiveness and a generally favorable safety profile, making it a trusted choice for healthcare providers and patients.
- Cost-Effectiveness of Generics: The availability of affordable generic options makes olopatadine a primary treatment choice, particularly for self-pay patients and healthcare systems focused on cost containment.
- Expanding Indications and Formulations: While primary indications are established, ongoing research into potential new uses or the development of improved delivery systems can sustain interest. Preservative-free formulations cater to a growing segment of the population with sensitivities.
Market Challenges:
- Intense Generic Competition: The large number of generic manufacturers leads to significant price erosion and thin profit margins, particularly for ophthalmic solutions.
- Development of Newer Antihistamines: Newer generations of antihistamines, some with different mechanisms of action or perceived advantages (e.g., fewer systemic side effects), can pose competitive threats.
- Market Saturation: The ophthalmic and nasal allergy markets are mature, with many established treatment options. Gaining significant market share often requires aggressive pricing or product differentiation.
- Reimbursement Pressures: Payers continuously seek to reduce healthcare costs, which can lead to stricter formulary management and lower reimbursement rates for established generics.
- Supply Chain Vulnerabilities: Reliance on global supply chains for active pharmaceutical ingredients (APIs) and finished products can create risks of shortages or price volatility.
Key Takeaways
The olopatadine market is stable, driven by the high prevalence of allergic diseases and the accessibility of generic products. Originator brands hold a premium segment, but the vast majority of volume and value is in generics, characterized by intense price competition. Future price projections indicate continued stability with a downward bias due to ongoing generic pressures. While the market faces challenges from newer therapies and reimbursement pressures, the established efficacy and cost-effectiveness of olopatadine ensure its sustained demand.
Frequently Asked Questions
-
Will the price of generic olopatadine ophthalmic solutions increase in the next two years? Significant price increases for generic olopatadine ophthalmic solutions are not anticipated. The market is highly competitive, and pricing is expected to remain stable or experience minor downward pressure.
-
Are there any new olopatadine formulations expected to enter the market that could significantly alter pricing? While incremental improvements in formulations or delivery methods are possible, major disruptive new olopatadine formulations are unlikely to drastically alter the pricing landscape in the short to medium term, given the drug's mature lifecycle.
-
What is the primary driver for the growth in the olopatadine market? The primary driver for market growth is the increasing global incidence of allergic conjunctivitis and allergic rhinitis, coupled with the widespread availability and affordability of generic olopatadine products.
-
How does the price of a 0.2% generic olopatadine ophthalmic solution compare to a 0.1% generic? Typically, a 0.2% generic olopatadine ophthalmic solution is priced slightly higher per unit volume than a 0.1% generic, reflecting the higher concentration of the active ingredient and potentially different manufacturing efficiencies. However, when comparing cost per dose or cost per milliliter, the difference may be less pronounced.
-
What is the regulatory status of olopatadine in major global markets? Olopatadine has received regulatory approval in numerous major global markets, including the United States (FDA), European Union (EMA), Japan (PMDA), and Canada (Health Canada), for its indicated uses. Generic approvals vary by region.
Citations
[1] World Allergy Organization. (n.d.). Allergic Rhinitis. Retrieved from https://www.worldallergy.org/education-and-resources/allergic-disease-resource-center/allergic-rhinitis
More… ↓
