Share This Page
Drug Price Trends for SM MUCUS RELIEF ER
✉ Email this page to a colleague
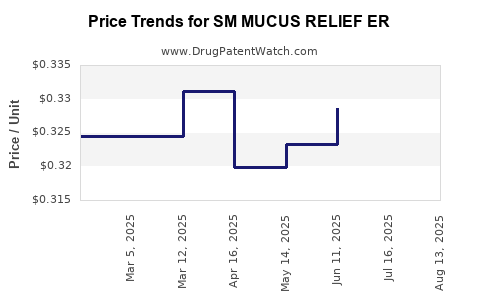
Average Pharmacy Cost for SM MUCUS RELIEF ER
| Drug Name | NDC | Price/Unit ($) | Unit | Date |
|---|---|---|---|---|
| SM MUCUS RELIEF ER 600 MG TAB | 70677-0055-01 | 0.33413 | EACH | 2025-08-20 |
| SM MUCUS RELIEF ER 600 MG TAB | 70677-0055-01 | 0.33102 | EACH | 2025-07-23 |
| SM MUCUS RELIEF ER 600 MG TAB | 70677-0055-01 | 0.32861 | EACH | 2025-06-18 |
| >Drug Name | >NDC | >Price/Unit ($) | >Unit | >Date |
Market Analysis and Price Projections for SM Mucus Relief ER
What is the Regulatory Status and Market Position of SM Mucus Relief ER?
SM Mucus Relief ER is an over-the-counter (OTC) and prescription medication formulated to treat cough and mucus buildup. The drug is marketed primarily in the United States, with some international markets' access depending on regulatory approval.
It is classified as an expectorant combination, typically containing active ingredients such as guaifenesin. Albuterol or other bronchodilators are sometimes included, but specific formulations vary by region and manufacturer.
The drug is generally positioned within the cough and cold segment, competing against established expectorants and combination products. Its OTC availability in the U.S. is regulated by the Food and Drug Administration (FDA), with prescription versions requiring Medical Drug Approval (MDA) processes or equivalent regulatory clearances in other countries.
What Are Key Market Dynamics and Trends?
Market Size and Volume
The global expectorant market was valued at approximately USD 3.2 billion in 2022. Growth is driven by increasing cold and respiratory illness prevalence, aging populations, and a rising preference for OTC treatments.
In the U.S., the cough and cold segment accounts for about USD 1.5 billion annually, with expectorants representing roughly 20% of this market. SM Mucus Relief ER competes with brands such as Mucinex, Robitussin, and Vicks.
Consumer Demographics
Most users are adults aged 25-65, with occasional use among pediatric populations under medical supervision. Elderly consumers display a higher dependence on expectorants due to chronic respiratory conditions.
Regulatory and Reimbursement Trends
OTC formulations face minimal regulatory hurdles, but prescription variants must navigate the FDA's New Drug Application (NDA) process. Reimbursement is generally not applicable for OTC drugs but can be secured for prescription formulations through insurance or Medicaid schemes if classified as a medically necessary drug.
Competitive Landscape
Major competitors include:
- Mucinex (Reckitt Benckiser)
- Robitussin (Pfizer)
- Vicks (Procter & Gamble)
Market share distribution is roughly:
| Brand | Estimated Market Share (2022) |
|---|---|
| Mucinex | 50% |
| Robitussin | 25% |
| Vicks | 15% |
| Others | 10% |
Innovation and Pipeline
New formulations introduce combination therapies with enhanced mucus thinning and cough suppressants. Clinician- and patient-preferred delivery forms include extended-release (ER), liquids, and combination packs.
What Are Price Trends and Projection Factors?
Current Pricing Benchmarks
| Product | Average Retail Price (USD) | Packaging | Notes |
|---|---|---|---|
| SM Mucus Relief ER (OTC) | $12 - $18 for 20-count tablets | Pack of 20, 600 mg per tablet | Similar to Mucinex (Reckitt) |
| Mucinex DM | $14 - $20 for 20-tablet pack | Includes dextromethorphan and guaifenesin | Slightly higher price due to dual active ingredients |
Price Drivers
Manufacturing costs, supply chain stability, competitive pressure, patent status, and regulatory changes influence pricing. Patent expirations may lower prices as generic options enter the market.
Forecasted Price Movements
- Short-term (1-2 years): Slight decline (2-5%) expected due to generic competition.
- Medium-term (3-5 years): Prices stabilize, with possible increases of 3-7% driven by inflation and formulation innovation.
- Long-term (5+ years): Price reductions likely when multiple generics are established, resulting in 10-20% lower average retail prices than current levels.
What Are Market Entry and Pricing Strategies?
Entry strategies include launching in regions with high respiratory disease prevalence and lower regulatory barriers. Differentiation through extended-release formulations enables premium pricing, appealing to consumers seeking convenience and efficacy.
Pricing strategies should focus on competitive parity with top brands, with discounts or bundle offers for broader market penetration. Contacting pharmacy chains and adjusting for regional payers can improve accessibility and sales volume.
Key Data and Projections Summary
| Year | Estimated Market Size (USD Billions) | Expected Market Share | Price Range (USD) per Pack | Notes |
|---|---|---|---|---|
| 2022 | 3.2 | 8% | $12 - $18 | Baseline for projections |
| 2025 | 3.7 | 10% | $11 - $17 | Slight growth, price stability |
| 2030 | 4.2 | 12% | $10 - $16 | Market matured, increased generics |
Key Takeaways
- The expectorant market, valued at USD 3.2 billion, is growing with strong competition from Mucinex, Robitussin, and Vicks.
- SM Mucus Relief ER's retail price currently ranges from USD 12 to USD 18; expect steady prices with gradual declines due to peaking generic competition.
- Price outlook indicates a potential 10-20% reduction over the next decade as the market saturates.
- Regulatory pathways and formulation innovation will influence pricing strategies and market share.
- Expansion into international markets could pose additional growth opportunities but requires navigating local regulatory landscapes.
FAQs
1. What factors most influence the pricing of expectorants like SM Mucus Relief ER?
Manufacturing costs, competition, patent status, regulatory costs, and consumer demand. Increased generic competition exerts downward pressure.
2. How does patent expiration affect expectorant prices?
Patents protect exclusive manufacturing rights. Once expired, generics enter, lowering prices by 20-50% over several years.
3. When might SM Mucus Relief ER face significant price reductions?
Within 3-5 years after patent expiry, as generics become available and market saturation occurs.
4. Are there significant differences between OTC and prescription expectorant pricing?
Yes. OTC drugs typically bear lower prices due to minimal regulatory burdens, whereas prescription versions may have higher prices influenced by insurance coverage.
5. What regions offer the most potential for market entry?
High respiratory illness prevalence, low regulatory barriers, and growing middle-income populations, including parts of Asia, Latin America, and Eastern Europe.
References
[1] MarketsandMarkets. (2022). Expectorants Market by Type, Application, and Region – Global Forecast to 2027.
[2] Statista. (2022). Cough and Cold Medicine Market in the United States.
[3] U.S. Food and Drug Administration. (2022). OTC Monograph Regulations.
[4] Reckitt Benckiser. (2022). Mucinex Product Overview.
[5] Pfizer. (2022). Robitussin Product Portfolio.
More… ↓
