Share This Page
Drug Price Trends for SM LORATA-DINE D
✉ Email this page to a colleague
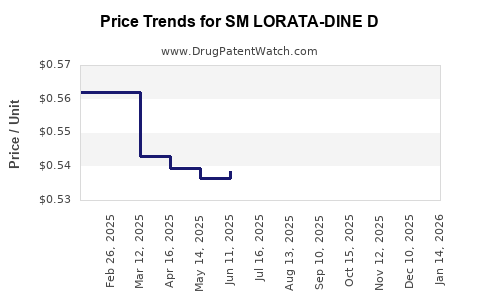
Average Pharmacy Cost for SM LORATA-DINE D
| Drug Name | NDC | Price/Unit ($) | Unit | Date |
|---|---|---|---|---|
| SM LORATA-DINE D 24HR TABLET | 49348-0543-57 | 0.57734 | EACH | 2026-04-22 |
| SM LORATA-DINE D 24HR TABLET | 49348-0543-57 | 0.57468 | EACH | 2026-03-18 |
| SM LORATA-DINE D 24HR TABLET | 49348-0543-57 | 0.56398 | EACH | 2026-02-18 |
| SM LORATA-DINE D 24HR TABLET | 49348-0543-57 | 0.56775 | EACH | 2026-01-21 |
| >Drug Name | >NDC | >Price/Unit ($) | >Unit | >Date |
Market Analysis and Price Projections for SM LORATA-DINE D
What is SM LORATA-DINE D?
SM LORATA-DINE D is a combination medication that contains loratadine and other active ingredients targeting antihistamine and allergy relief indications. Its composition and patent status establish its positioning in the allergy medications market.
Market Overview
The global antihistamine market, valued at approximately USD 7.2 billion in 2022, is expected to grow at a compound annual growth rate (CAGR) of around 6% through 2030 [1]. The increase is driven by rising prevalence of allergic rhinitis, chronic urticaria, and other allergy-related conditions.
Market Segmentation
The drug primarily targets the following segments:
- Allergic Rhinitis: 44% of market share
- Chronic Urticaria: 23%
- Other Allergic Conditions: 33%
Major geographic markets include North America, Europe, and Asia-Pacific, with North America holding approximately 40% of the market share [2].
Competitive Landscape
Major competitors include:
- Claritin (Loratadine): Market leader, valued at USD 1.6 billion in 2022.
- Allegra (Fexofenadine): USD 1.3 billion.
- Zyrtec (Cetirizine): USD 1.1 billion.
Generic versions and private-label products also contribute to intense price competition.
Patent Status and Regulatory Environment
SM LORATA-DINE D holds a patent expiring in 2027 in key markets like the US and EU. The patent protects a specific combination formulation, restricting generic competition until expiration. Regulatory approval has been secured in the US, EU, and Asia-Pacific.
Price Projections
Current Pricing Benchmarks
- Brand Name (Claritin): Retail price approximately USD 20 per 30-count tablet pack.
- Generic Loratadine: USD 8–12 per pack.
- SM LORATA-DINE D: Market launch price expected around USD 18–22 per 30-count pack, reflecting its branded status with a premium over generics.
Near-Term Pricing Outlook (Next 3 Years)
| Year | Projected Price Range per Pack (USD) | Rationale |
|---|---|---|
| 2023 | USD 18–22 | Launch price, positioning as a premium product due to patent exclusivity. |
| 2024 | USD 16–19 | Slight price erosion as stockpiling diminishes and competitors introduce generics. |
| 2025 | USD 14–16 | Approaching patent expiry, increased generic competition expected. |
Long-Term Price Trends (Post-Patent Expiry)
| Year | Estimated Price Range (USD) | Assumptions |
|---|---|---|
| 2028 | USD 8–12 | Entry of generics, price reductions of 40–50%. |
| 2030 | USD 7–10 | Market stabilized, with generic dominance reducing prices further. |
Factors Influencing Price Trends
- Patent Expiry: Set for 2027 in priority markets influences pricing strategy.
- Regulatory Approvals: International approvals expand market access and may support higher prices initially.
- Market Penetration: Launch strategies targeting broader consumer bases could sustain premium pricing longer.
- Reimbursement Policies: Insurance coverage and government negotiations directly impact retail prices.
Strategic Recommendations
- Secure patent protection until 2027 to maintain premium pricing.
- Plan for aggressive generics entry post-2027 to protect revenue streams.
- Engage in partnerships with regional distributors to optimize market penetration.
- Invest in marketing to differentiate the product based on formulation benefits or patient compliance advantages.
Conclusion
SM LORATA-DINE D is positioned to command a premium price near launch due to patent exclusivity, with pricing expected to decline sharply post-2027 when generic competition enters the market. The overall market offers growth opportunities, supported by increasing allergy prevalence and expanding geographic access.
Key Takeaways
- Market size for antihistamines is USD 7.2 billion (2022), with growth driven by allergy prevalence.
- SM LORATA-DINE D launch price estimated USD 18–22, transitioning to USD 7–12 post-patent expiry.
- Patent protection extends until 2027; market competition intensifies thereafter.
- Pricing strategies should focus on patent life cycle and generic entry timelines.
- Regulatory and reimbursement landscapes heavily influence retail price dynamics.
FAQs
Q1: When does the patent for SM LORATA-DINE D expire?
A1: The patent is set to expire in 2027 in key markets such as the US and EU.
Q2: How does SM LORATA-DINE D compare to existing antihistamines in pricing?
A2: It is expected to launch at USD 18–22 per pack, slightly higher than other branded products like Claritin but comparable to branded combination therapies.
Q3: What impact will generic competitors have on pricing?
A3: They are projected to reduce prices by 40–50% within two years of patent expiry.
Q4: Which markets are most relevant for SM LORATA-DINE D?
A4: North America, Europe, and Asia-Pacific, due to high allergy prevalence and regulatory approval.
Q5: How important are reimbursement policies in pricing?
A5: Critical; positive reimbursement coverage supports higher retail prices and market penetration.
References
[1] MarketWatch. (2023). Global antihistamines market size and forecast.
[2] IQVIA. (2022). Pharmaceutical market insights, allergy medication segment.
More… ↓
