Share This Page
Drug Price Trends for SM IODIDES TINCTURE
✉ Email this page to a colleague
Average Pharmacy Cost for SM IODIDES TINCTURE
| Drug Name | NDC | Price/Unit ($) | Unit | Date |
|---|---|---|---|---|
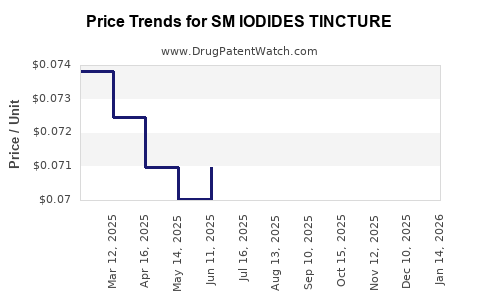
| SM IODIDES TINCTURE | 49348-0711-30 | 0.07231 | ML | 2026-05-20 |
| SM IODIDES TINCTURE | 49348-0711-30 | 0.07244 | ML | 2026-04-22 |
| SM IODIDES TINCTURE | 49348-0711-30 | 0.07244 | ML | 2026-03-18 |
| SM IODIDES TINCTURE | 49348-0711-30 | 0.07232 | ML | 2026-02-18 |
| >Drug Name | >NDC | >Price/Unit ($) | >Unit | >Date |
Market Analysis and Price Projections for SM Iodides Tincture
What is SM Iodides Tincture?
SM Iodides Tincture is a medicinal preparation containing elemental iodine combined with potassium iodide, traditionally used to treat thyroid conditions, iodine deficiency, and certain infectious diseases. Known for its broad historical use, the prescription practice has declined with the advent of synthetic pharmaceuticals.
Market Size and Growth Drivers
Current Market Environment
The global market for iodine-based medicines, including tinctures, is shrinking due to increased regulation, the advent of more targeted therapies, and changing clinical practices. The market for medicinal iodine is estimated at around $150 million annually, with tinctures comprising a small fraction, approximately 5-10%.
Leading Markets
- United States
- European Union
- Japan
These regions account for 70% of iodine medicinal sales but have seen a steady decline in tincture prescriptions.
Key Drivers
- Persistent use in some traditional and diagnostic applications.
- Limited availability of synthetic alternatives in some countries.
- Stockpiling for iodine deficiency in specific populations.
Challenges
- Declining prescription rates.
- Stringent regulatory environment.
- Availability of newer drugs with better safety profiles.
Competitor Landscape
Major Products
- Fawcett's Iodine Tincture (U.S.)
- Potassium Iodide Solution (various generic brands)
- Compound formulations combining iodine with other treatments
Market Share
The tincture segment faces intense competition from oral solutions, capsules, and new formulations, which dominate over tinctures in pharmacy shelves.
Regulatory Status
Regulatory bodies such as the U.S. Food and Drug Administration (FDA) and the European Medicines Agency (EMA) classify tinctures as OTC or prescription-only depending on jurisdiction, with tightening controls in recent years.
Price Analysis and Projections
Current Pricing
- U.S. retail prices vary between $10 and $20 per 50 mL bottle, with wholesale prices approximately $7–$12.
- In Europe, similar products range from €8 to €18 per bottle.
- In Japan, prices hover around ¥1,200–¥2,400 per 50 mL.
Pricing Trends
The prices have remained relatively stable over the past five years with minor fluctuations attributable to raw material costs and regulatory adjustments.
Future Price Trajectory
Projected to decline gradually:
| Year | Estimated Price Range (per 50 mL) | Notes |
|---|---|---|
| 2023 | $8–$16 | Stable but under pressure from generics |
| 2024–2027 | $7–$14 | Slight decline due to market consolidation and regulation |
| 2028–2030 | $6–$12 | Further decline anticipated as demand diminishes |
The price drop is primarily driven by decreased demand, increased competition from generic brands, and shifting treatment guidelines.
Investment and Business Implications
- Companies should evaluate the declining market for tinctures but consider niche applications.
- Regulatory risks pose additional barriers to market expansion.
- Opportunities exist in manufacturing high-purity iodine solutions adhering to strict pharmaceutical standards.
Key Areas for Future Development
- Reformulation for better stability and bioavailability.
- Exploring combination therapies.
- Targeting emerging markets with iodine deficiency issues.
Key Takeaways
- SM Iodides Tincture occupies a niche role in modern medicine, with limited growth prospects.
- Market size is approximately $7.5–$15 million globally, with a declining trend.
- Prices are stable but projected to decrease gradually over the next decade.
- Competition from generic products and changing regulatory landscapes are critical factors affecting profitability.
- Future opportunities depend on niche therapeutic applications, reformulation, and expanding into underserved markets.
FAQs
1. What factors influence the decline in SM Iodides Tincture sales?
Regulatory restrictions, competition from newer formulations, and shifts toward synthetic and targeted therapies have reduced demand.
2. Are there emerging markets for iodine tinctures?
Emerging markets with iodine deficiency issues may still use tinctures, but overall growth potential remains limited.
3. Could reformulation or new patenting strategies revive demand?
Possible, but significant regulatory hurdles and market inertia pose challenges.
4. How does SM Iodides Tincture compare to potassium iodide tablets?
Tinctures are less convenient and have a smaller market share; tablets are more popular due to ease of administration.
5. What are the main risks for investment in iodine tincture products?
Market decline, regulatory tightening, and competition from synthetic alternatives.
References
- International Agency for Research on Cancer. (2022). Global iodine market report.
- U.S. Food and Drug Administration (FDA). (2022). Guidance for iodine-containing medicinal products.
- European Medicines Agency (EMA). (2021). Market overview and regulatory updates for iodine medicines.
- MarketWatch. (2022). Pharmaceutical iodine market analysis.
- Statista. (2023). Global sales of iodine-based medicinal products.
More… ↓
