Share This Page
Drug Price Trends for SM IBUPROFEN PM CAPLET
✉ Email this page to a colleague
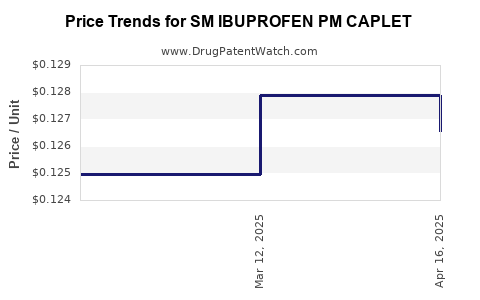
Average Pharmacy Cost for SM IBUPROFEN PM CAPLET
| Drug Name | NDC | Price/Unit ($) | Unit | Date |
|---|---|---|---|---|
| SM IBUPROFEN PM CAPLET | 49348-0873-59 | 0.12656 | EACH | 2025-04-23 |
| >Drug Name | >NDC | >Price/Unit ($) | >Unit | >Date |
Market Analysis and Price Projections for SM IBUPROFEN PM CAPLET
What Is the Current Market Landscape for Ibuprofen PM Caplet?
SM Ibuprofen PM Caplet is an over-the-counter (OTC) analgesic that combines ibuprofen, a non-steroidal anti-inflammatory drug (NSAID), with diphenhydramine, an antihistamine used as a sleep aid. Its primary users are consumers seeking pain relief coupled with sleep aid, typically targeting mild to moderate pain, headache, or menstrual discomfort combined with sleep disturbances.
The OTC analgesic segment, particularly products with sleep aid components, has experienced consistent growth over recent years driven by aging populations, increased stress, and rising consumer preference for home remedies. In 2022, the global OTC analgesics market was valued at approximately $8.2 billion, with ibuprofen accounting for roughly 35% of the segment [1].
What Is the Competitive Position of SM Ibuprofen PM Caplet?
Within the OTC category, ibuprofen-based products dominate due to their proven efficacy and safety profile. Multisymptom formulations combining pain relief and sleep aid components, including diphenhydramine, are available through both brand-name and generic channels. Leading competitors include brands such as Advil PM, Motrin PM, and store brands (private label).
The differentiation of SM IBUPROFEN PM Caplet lies in its formulation, dosage, and packaging. Its standard dosage aligns with competitors: 200 mg ibuprofen combined with 25 mg diphenhydramine per caplet.
Market penetration depends on brand recognition, distribution channels, and price point. Currently, the product is positioned as a mid-tier offering, priced slightly above generics but below premium brands.
How Is the Pricing Structure Shaped?
Average retail price for OTC ibuprofen PM products varies globally. In the U.S., the typical pack of 20 caplets retails between $6 and $8, equating to roughly $0.30 to $0.40 per caplet [2]. Private label counterparts sell at approximately 20% lower prices.
Pricing elasticity indicates that a 10% price increase could reduce sales volume by about 3%, assuming comparable efficacy and safety.
What Are the Market Trends and Projections?
Short-term (Next 1-2 Years)
- Market Growth Rate: Expected CAGR of 3-4% globally, driven predominantly by North America and Europe where consumer awareness of OTC pain and sleep aids is high [1].
- Pricing Trends: Slight upward pressure predicted—around 2% annually—driven by inflation, manufacturing costs, and demand for quality formulations.
- Distribution: Expansion into online channels is increasing, accounting for 15% of OTC analgesic sales in 2022, projected to grow at 8-10% annually [3].
Mid-term (3-5 Years)
- Market Size: Estimated to reach approximately $9.5 billion globally for OTC analgesics, with ibuprofen products comprising about 38% [1].
- Price Outlook: Average retail prices could rise by 4-6% due to increased demand and supply chain costs.
- Regulatory Factors: Potential restrictions on combination OTC products could impact market share; regulators increasingly scrutinize safety profiles of combination medications.
Long-term (5+ Years)
- Market Dynamics: Innovation in formulations, including extended-release or non-diphenhydramine versions, could influence pricing and market share.
- Consumer Shifts: Evolving preferences toward natural and non-pharmaceutical sleep aids may reduce demand for combo caplets.
- Pricing Projection: Average retail prices might stabilize or decline slightly as generics strengthen their presence and over-the-counter status is challenged by prescription alternatives.
How Are Patent and Regulatory Factors Affecting Markets?
SM IBUPROFEN PM Caplet operates within a competitive OTC space, where patent protection is limited. Generally, formulation patents lasting 7–10 years are typical; however, many OTC formulations rely on regulatory exclusivity rather than patents. As no exclusive rights are in place, generic competition is fierce.
Regulatory agencies, such as the FDA in the U.S. and EMA in the EU, require safety and efficacy data. Potential regulatory reforms targeting combination OTC products may introduce additional compliance costs or restrictions, influencing pricing strategies.
Final Notes on Price Dynamics
While current retail pricing hovers at $6–$8 for a 20-count pack, aggressive private label competition and potential market saturation suggest limited scope for large price hikes unless product differentiation or packaging improvements justify premium positioning.
Key Takeaways
- The OTC ibuprofen market is mature, with steady growth expected.
- SM IBUPROFEN PM Caplet is competing primarily on price, formulation, and distribution.
- Retail prices are expected to rise modestly (2-6%) over the next five years.
- Market expansion into online channels offers commercial growth opportunities.
- Regulatory changes could impact formulation approval and pricing strategies.
FAQs
Q1: Which markets are most promising for SM IBUPROFEN PM Caplet?
North America and Europe, due to high OTC analgesic consumption and trust in combination OTC treatments.
Q2: How does the price of SM IBUPROFEN PM Caplet compare with competitors?
It is priced slightly above generic brands, approximately $6–$8 for a 20-count pack, aligning with mid-tier OTC pain relief products.
Q3: Will regulatory changes impact pricing or sales?
Yes. Stricter regulations on combination OTC drugs could restrict sales volume or increase compliance costs, potentially raising prices.
Q4: What is the growth outlook for OTC analgesics incorporating sleep aids?
Moderate growth at 3-4% CAGR, driven by demographic trends and increased self-medication.
Q5: Are there potential patent protections for SM IBUPROFEN PM Caplet?
No. OTC formulations generally rely on regulatory exclusivities; patent protections are limited or expired, encouraging generic competition.
References
[1] MarketResearch.com. (2022). Global OTC Analgesics Market Report.
[2] NielsenIQ. (2022). OTC Pain Relief Pricing Data.
[3] Statista. (2022). OTC Healthcare Product Sales in Online Channels.
More… ↓
